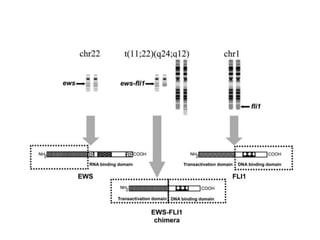
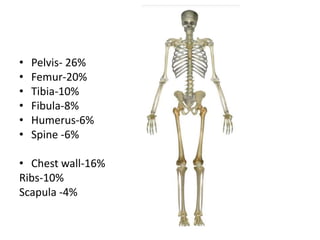
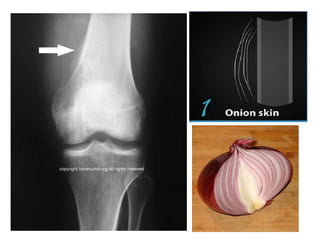
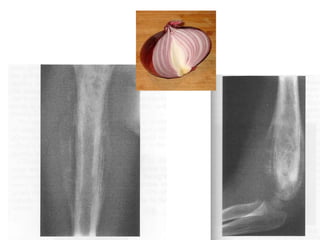
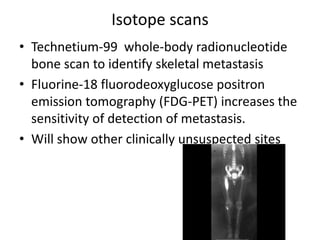
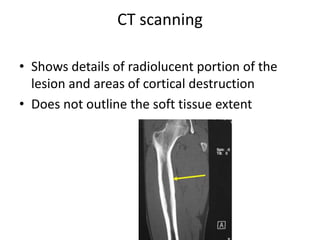
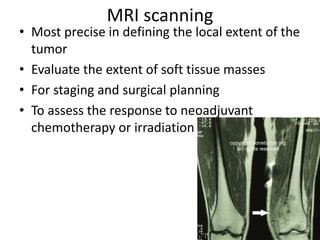
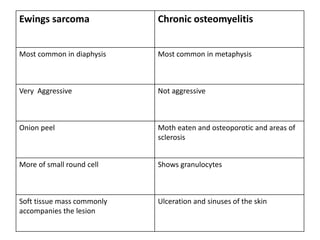
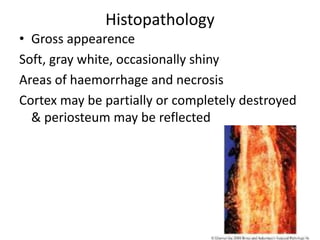
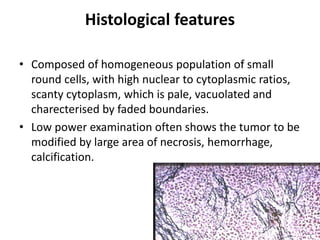
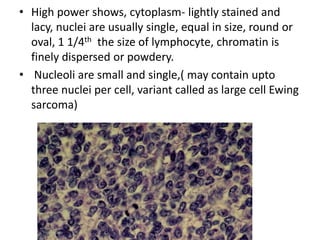
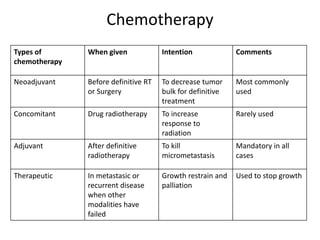
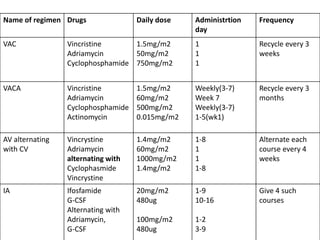
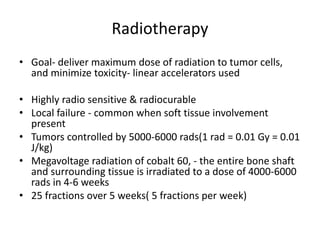
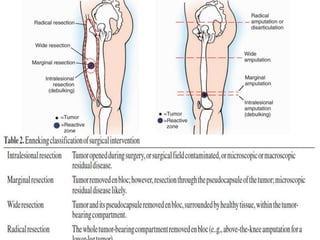
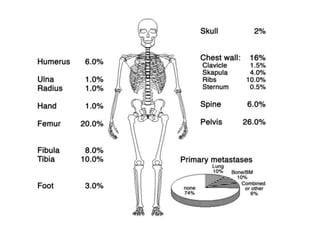
Ewing sarcoma is a rare cancer that develops in bone or soft tissue. It is most common in children and young adults between ages 10-20. The cancer cells are small, round cells and are usually found in the bones of the legs, pelvis, ribs, or spine. Ewing sarcoma is treated with a combination of chemotherapy, radiation therapy, and surgery. Prognosis depends on factors like tumor size, location, and whether the cancer has spread. With current multi-agent chemotherapy regimens, long-term survival rates are around 60-70%.