The document provides an overview of Ebola virus disease (EVD), including its origins, transmission, signs and symptoms, diagnosis, treatment and recovery. Some key points:
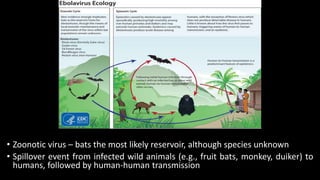
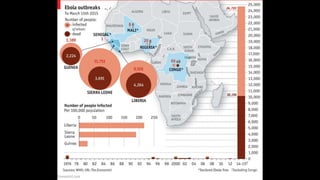
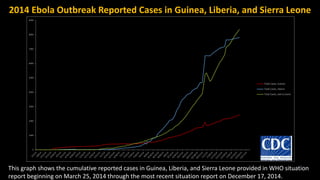
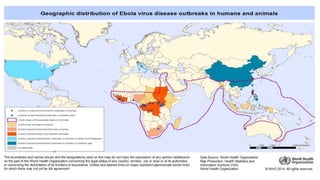
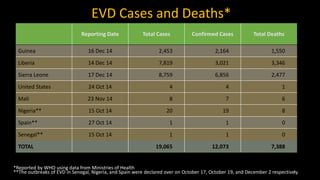
- EVD first appeared in 1976 in simultaneous outbreaks in Sudan and Democratic Republic of Congo. The current 2014 outbreak in West Africa is the largest on record.
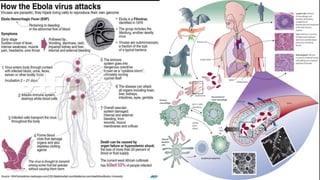
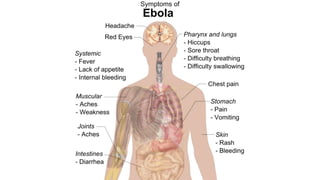
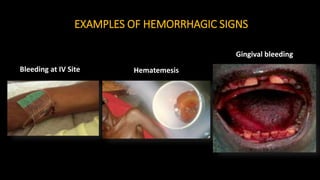
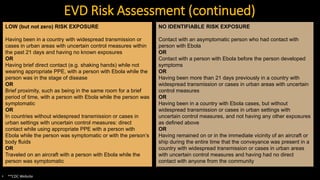
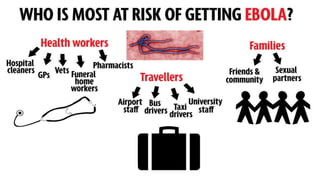
- The virus is transmitted through direct contact with body fluids of infected humans or animals. Early symptoms are nonspecific but progress to hemorrhagic fever, vomiting, diarrhea and organ failure.
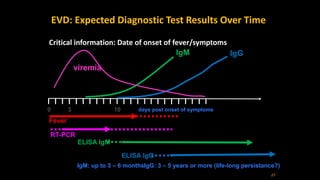
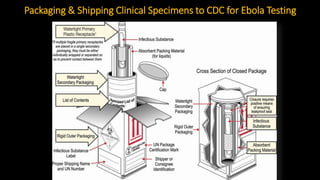
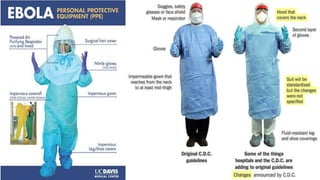
- Diagnosis involves detecting the virus or antibodies in blood, with RT-PCR being the most sensitive test. There is no approved vaccine or treatment, so care is largely supportive