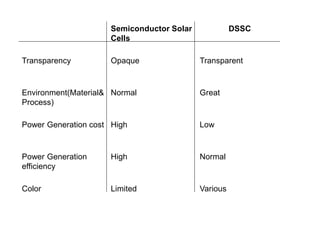
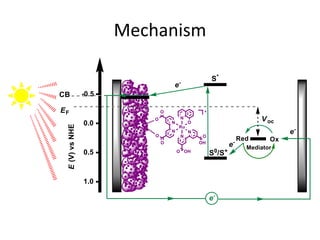
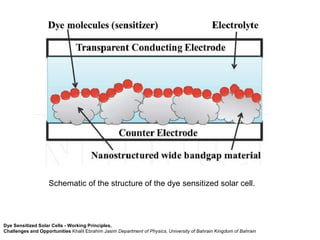
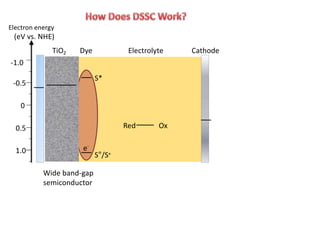
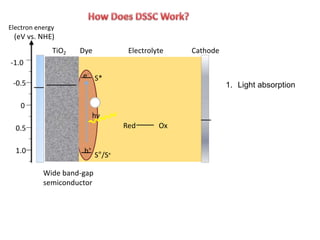
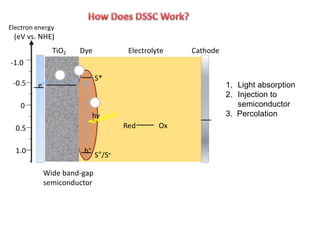
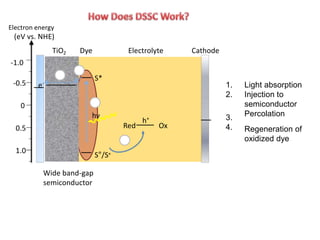
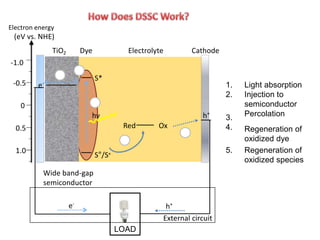
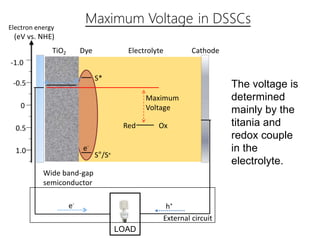
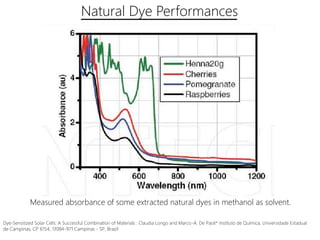
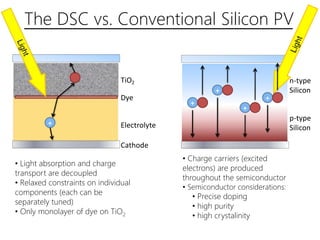
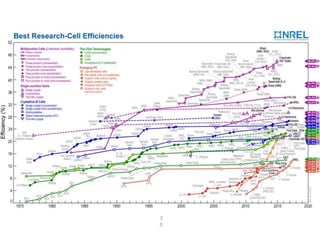
Dye-sensitized solar cells (DSSCs) are a low-cost alternative to conventional solar cells, utilizing natural dyes for light absorption and showing potential for applications in building-integrated photovoltaics. They consist of components like a semiconducting electrode, dye sensitizer, redox electrolyte, and counter electrode, offering advantages such as high performance-price ratio and operation in low light conditions. However, they face challenges like lower efficiencies and issues with dye stability and liquid electrolyte leakage.