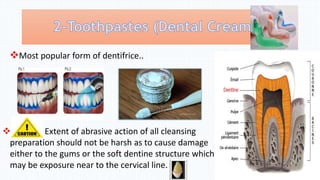
This document defines dentifrices and lists their main components. It states that dentifrices are substances used with toothbrushes to clean teeth and help the mechanical cleaning action. The most common form is toothpaste. It then describes the main components of toothpaste which include cleansing and polishing materials to remove stains, detergents and foaming agents to clean teeth, humectants to prevent drying, binding agents to maintain stability, sweetening agents for taste, and flavors - usually mint, to provide a fresh sensation. Minor ingredients can include abrasives, pH regulators, and therapeutic agents.