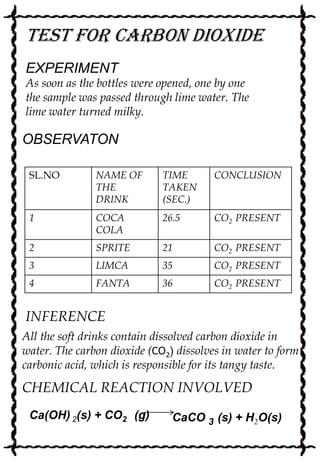
Rajkira conducted a chemistry project to determine the contents of various cold drinks. Through a series of qualitative tests, Rajkira found that all the drinks contained glucose, alcohol, sucrose, phosphate, and carbon dioxide. The drinks varied in their acidity levels and amount of dissolved carbon dioxide. While cold drinks provide refreshment, they can also be harmful due to their sugar content and ability to damage teeth and bones over long-term consumption.