Embed presentation
Downloaded 583 times



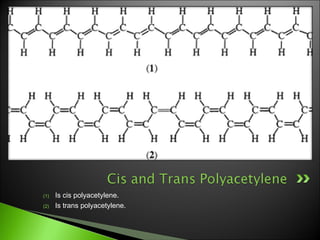







This document discusses polyacetylene, an organic polymer that can conduct electricity. It describes how polyacetylene was first synthesized in 1970 using a Ziegler-Natta catalyst. Polyacetylene exists in two forms, cis and trans. The document also explains how doping polyacetylene with oxidizing or reducing agents can increase its conductivity, and some applications of conductive polymers including rechargeable batteries, sensors, transistors, LEDs, solar cells, and electromagnetic shielding.









