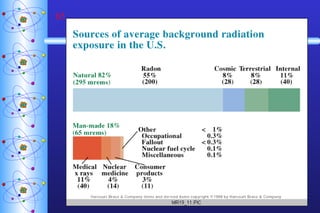
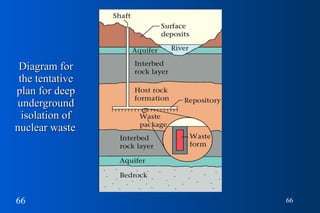
This document provides a summary of key concepts in nuclear chemistry, including:
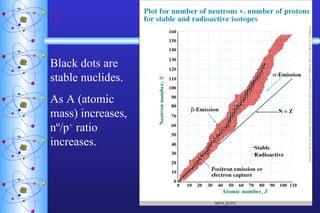
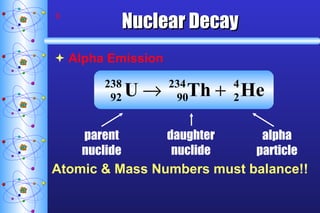
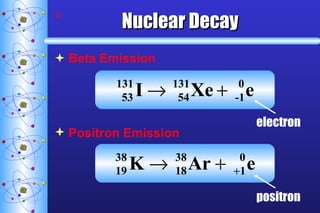
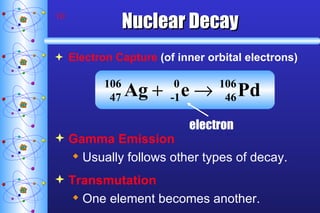
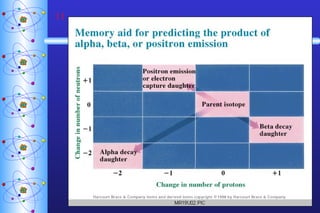
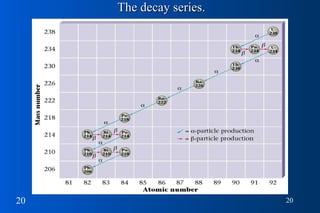
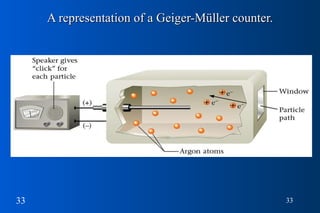
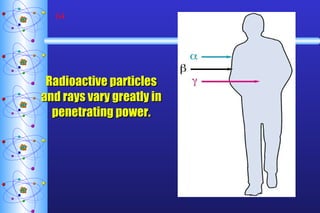
1) Nuclear stability and radioactive decay involve the emission of particles like alpha and beta from unstable nuclei. Different types of radiation (alpha, beta, gamma) require different shielding methods.
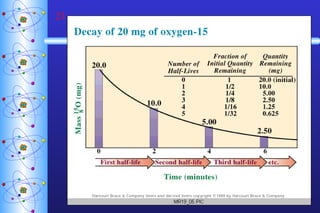
2) Radioactive decay follows first-order kinetics and half-life is used to describe the rate of decay. Carbon-14 dating and lead-uranium dating use radioactive half-lives to determine the age of materials.
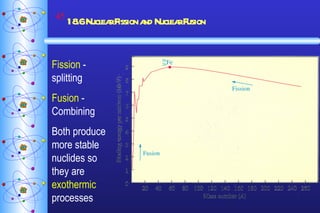
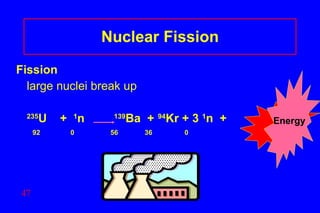
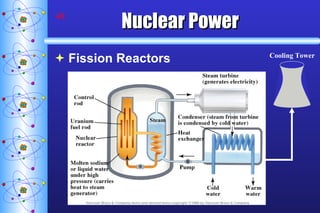
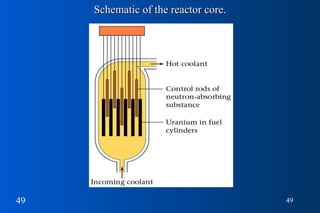
3) Nuclear reactions like fission and fusion release large amounts of energy. Fission is the splitting of heavy nuclei like uranium-235 and is used in nuclear power reactors. Fusion combines light nuclei and occurs in