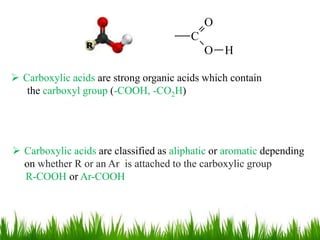
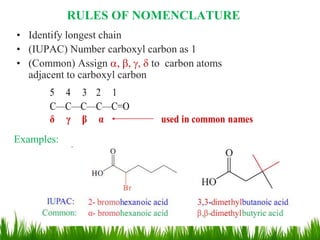
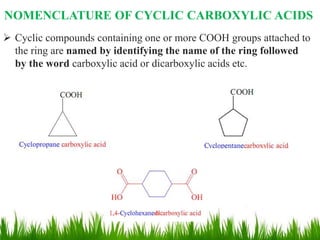
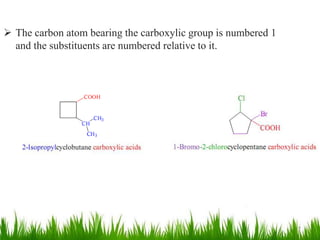
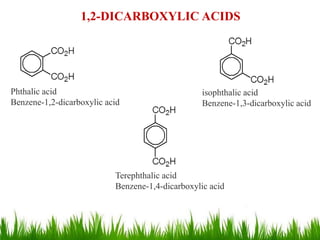
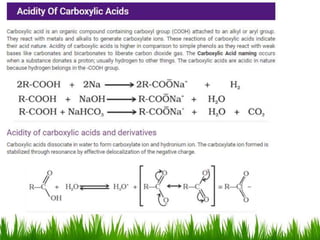
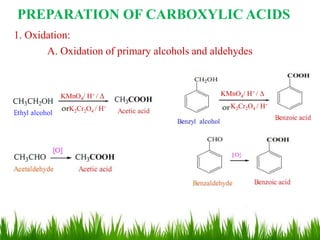
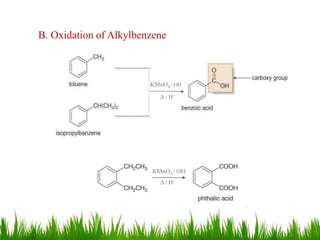
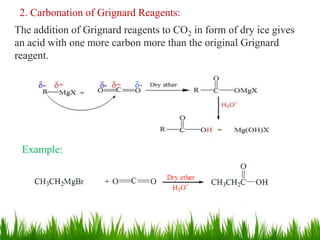
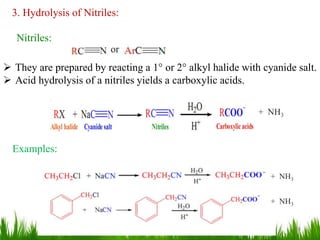
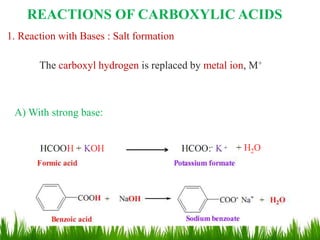
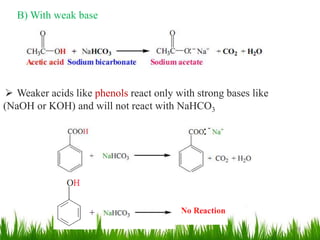
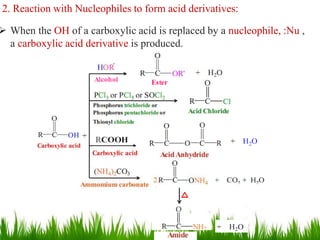
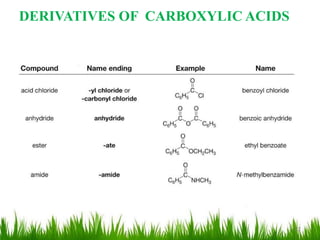
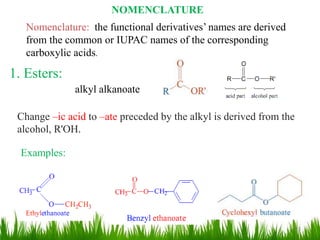
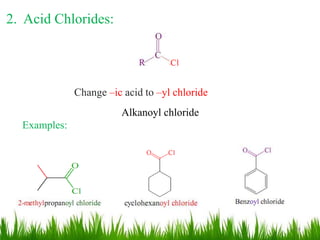
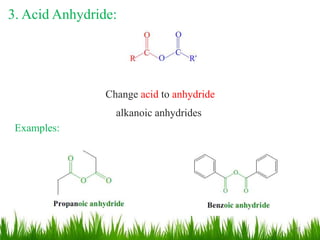
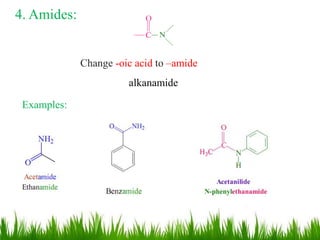
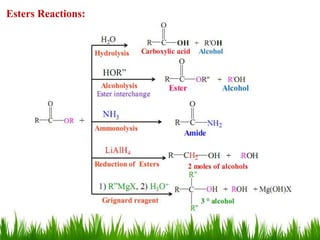
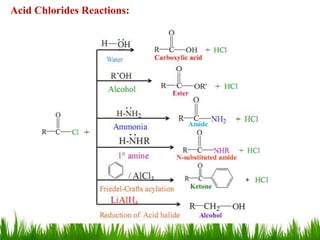
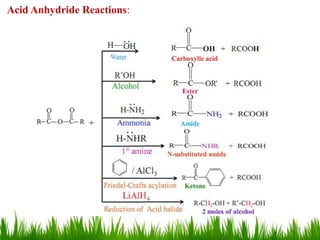
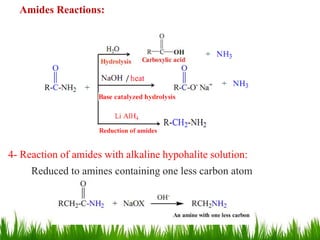
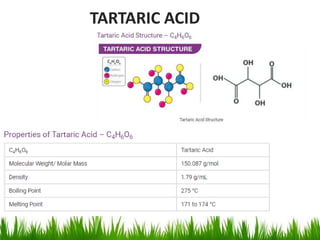
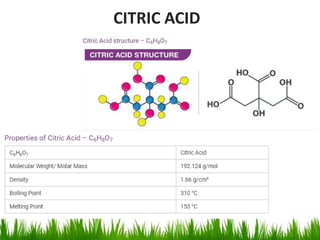
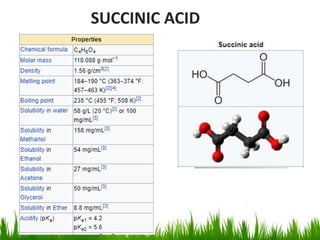
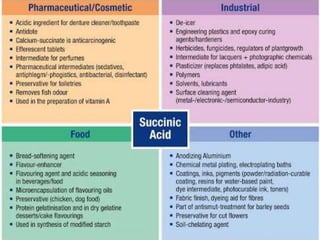
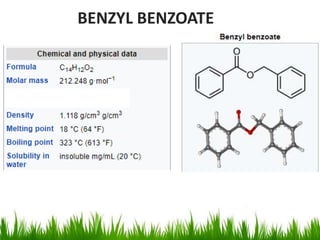
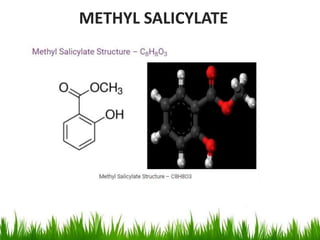
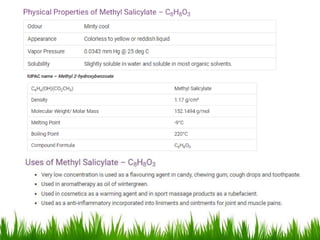
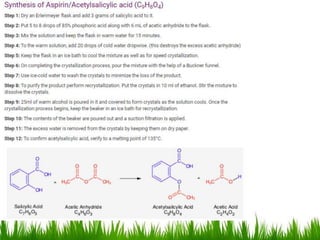
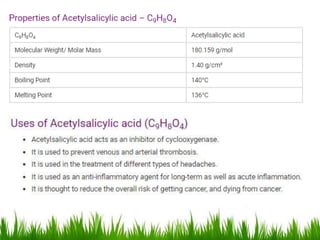
This document discusses the nomenclature, physical properties, preparation, and reactions of carboxylic acids. It begins by defining carboxylic acids and how they are classified. Rules of IUPAC nomenclature for aliphatic, cyclic, and aromatic carboxylic acids are provided. Key physical properties like solubility and boiling point are attributed to hydrogen bonding. Carboxylic acids are described as stronger acids than alcohols or phenols due to resonance stabilization of the conjugate base. Common methods for preparing carboxylic acids include oxidation reactions and hydrolysis of nitriles. Characteristic reactions include forming salts with bases, and generating acid derivatives like esters, acid chlorides, anhydrides, and am