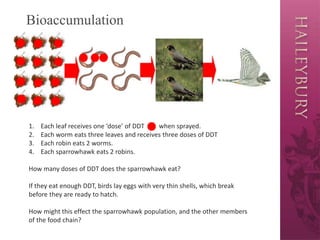
The document discusses bioaccumulation and biomagnification in ecosystems, illustrating how pesticides like DDT accumulate in food chains and their detrimental effects on organisms, particularly birds. It highlights the concept of biomagnification, where toxic substances increase in concentration at higher trophic levels, demonstrating this with the example of a sparrowhawk consuming worms and robins. Additionally, the document suggests biological controls as sustainable alternatives to chemical pesticides, emphasizing their lower environmental impact.