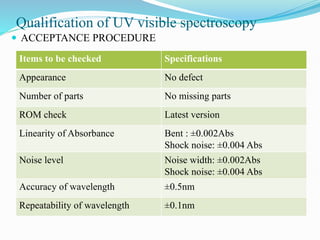
The document provides an in-depth overview of calibration and validation in the pharmaceutical industry, emphasizing the importance of validation as a critical quality assurance element to ensure consistent and compliant production processes. It outlines definitions, stages, types of validation—including process, cleaning, and analytical method validation—and the roles different departments play in this context. Furthermore, it details the necessity of both regulatory compliance and optimization of processes to achieve cost reduction and enhanced product quality.