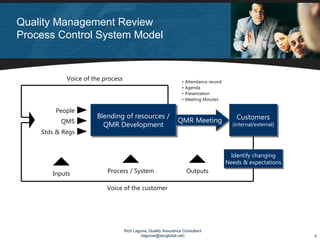
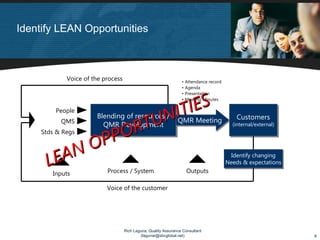
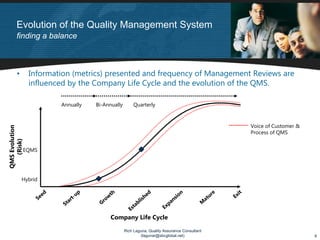
The document discusses applying lean principles to quality management reviews to make them more efficient and effective. It proposes a process-based model for quality management reviews and identifies opportunities to eliminate waste, such as streamlining documentation and approval processes. It also emphasizes the importance of presenting both positive and negative quality system performance data to facilitate continuous improvement and compliance with regulations. The goal is to conduct quality management reviews that are both lean, by reducing waste, and compliant, by fully evaluating the quality system's effectiveness.









![Compliant
Quality Management Reviews
Compliance Statements
Fully Implemented QMS
• The [company name] Quality Management System continues to be suitable
and effective with the identified Quality Plan, goals, and resource needs.
Start-Up QMS (pre-commercialization)
• The [company name] Quality Management System (QMS) continues to
evolve into a fully implemented system. The Management Review has
identified sub-processes status, implementation, and monitoring plans.
Therefore, the QMS is found to be suitable and effective with the Quality
Plan, goals, and resource needs.
Rich Laguna, Quality Assurance Consultant
(lagunar@sbcglobal.net) 13](https://image.slidesharecdn.com/asqleanmgmtrvwrlaguna-1264574390937-phpapp01/85/ASQ-Presentation-Lean-Mgt-Reviews-13-320.jpg)

