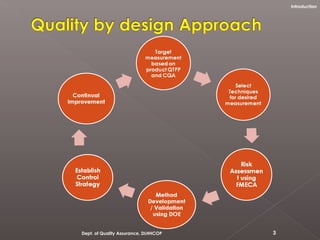
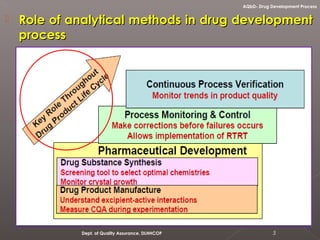
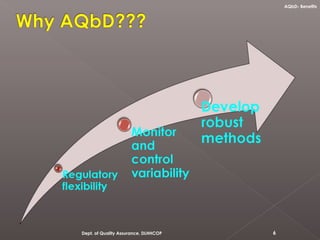
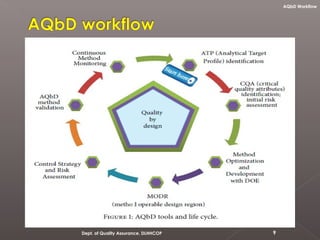
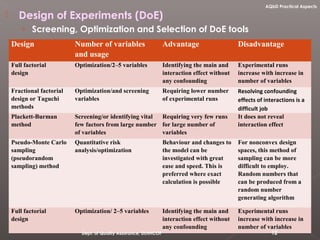
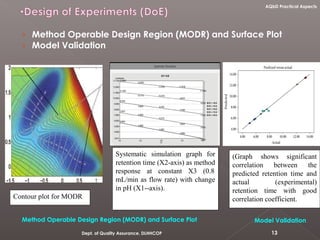
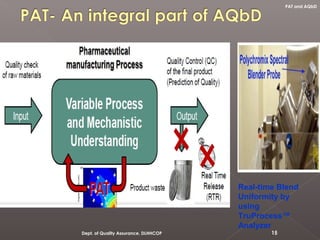
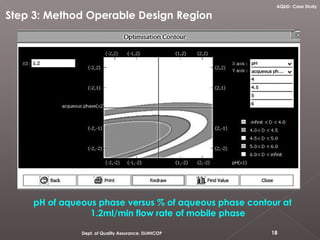
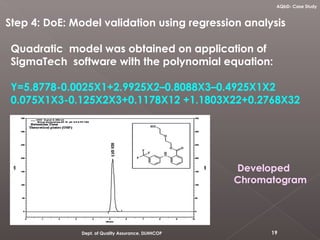
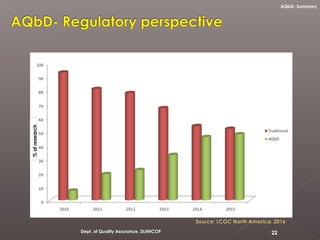
The document discusses analytical quality by design (AQbD) and its implementation. It compares traditional analytical methods to AQbD methods. AQbD uses a systematic approach including risk assessment, design of experiments, and establishing a method operable design region. A case study demonstrates developing an HPLC method for assay using an AQbD approach including target measurement, design of experiment, method validation, and establishing a method operable design region. The conclusion states AQbD requires defining the right analytical target profile and using appropriate tools to ensure the right analytics are performed at the right time.