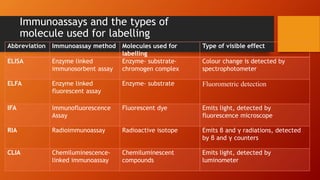
This document provides an overview of antigen-antibody reactions including their general properties, types of reactions, and evaluation of immunoassays. It describes various conventional techniques such as precipitation reactions, agglutination reactions, and complement fixation tests. It also summarizes newer techniques like enzyme-linked immunosorbent assay (ELISA), enzyme-linked fluorescent assay (ELFA), radioimmunoassay (RIA), and chemiluminescence-linked immunoassay (CLIA). The document aims to help students understand antigen-antibody reactions and their applications in diagnostic testing.













































































![Applications of ELISA
• ELISA used for antigen detection -hepatitis B [hepatitis B
surface antigen (HBsAg) and pre-core antigen (HBeAg)], NS1
antigen for dengue etc.
• ELISA can also be used for antibody detection against
hepatitis B, hepatitis C, HIV, dengue, EBV, HSV,
toxoplasmosis, leishmaniasis, etc.](https://image.slidesharecdn.com/ag-ab2-230422073546-c3a48b72/85/AG-AB-2-pptx-82-320.jpg)





