This document provides information on acute eye irritation testing methods. It discusses using topical anesthetics and systemic analgesics to minimize animal pain and distress during testing. Procedures are outlined for applying the test substance to a rabbit's eye while maintaining anesthesia. Lesions are then scored at intervals to evaluate irritation effects. The test aims to assess reversibility of eye damage in a humane manner.

![UNIT 2
TOXICOLOGICAL SCREENING METHODS
OBJECTIVE
1 Acute eye irritation and skin sensitization.
Irritation,
in biology and physiology, is a state of inflammation or painful reaction to allergy or cell-
lining damage. A stimulus or agent which induces the state of irritation is an
irritant.
Irritants are typically thought of as chemical agents (for example phenol and capsaicin) but
mechanical, thermal (heat), and radiative stimuli (for example ultraviolet light or ionising
radiations) can also be irritants. Irritation also has non-clinical usages referring to bothersome
physical or psychological pain or discomfort.
Irritation can also be induced by some allergic response due to exposure of some allergens for
example contact dermatitis, irritation of mucousal membranes and pruritus. Mucosal
membrane is most common site of irritation because it contains secretory glands that release
mucous which attracts the allergens due to its sticky nature.
Chronic irritation is a medical term signifying that afflictive health conditions have been
present for a while. There are many disorders that can cause chronic irritation, the majority
involves the skin, vagina, eyes and lungs.
Types of irritation
1 eye irritation
2 skin irriatation
3vaginal irritation
4 stomach irritation
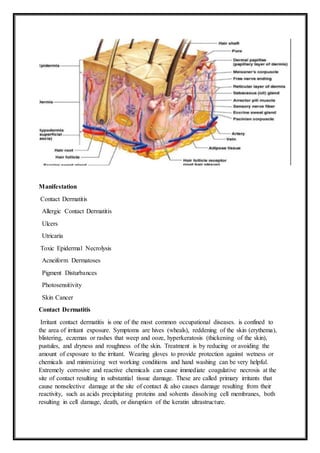
Eye irritation
Infant with blepharitis on the right eye
Modern office work with use of office equipment has raised concerns about possible adverse
health effects.[1] Since the 1970s, reports have linked mucosal, skin, and general symptoms to
work with self-copying paper. Emission of various particulate and volatile substances has
been suggested as specific causes. These symptoms have been related to Sick Building](https://image.slidesharecdn.com/acuteeyeirritation1-201225045831/85/Acute-eye-irritation-1-2-320.jpg)
![Syndrome, which involves symptoms such as irritation to the eyes, skin, and upper airways,
headache and fatigue.[2]
The eye is also a source of chronic irritation. Disorders like Sjögren's syndrome, where one
does not make tears, can cause a dry eye sensation which feels very unpleasant. The
condition is difficult to treat and is lifelong. Besides artificial tears, there is a drug called
Restasis which may help.[3]
Blepharitis is dryness and itching on the upper eyelids. This condition is often seeing in
young people and can lead to reddish dry eye and scaly eyebrows. To relieve the itching
sensation, one may need to apply warm compresses and use topical corticosteroid creams.
Acute eye irritation/corrosion- 405
OECD Guidelines for Testing of Chemicals are periodically reviewed to ensure that they
reflect the best available science. The latest update mainly focused on the use of analgesics
and anesthetics without impacting the basic concept and structure of the Test Guideline.
ICCVAM and an independent international scientific peer review panel reviewed the
usefulness and limitations of routinely using topical anesthetics, systemic analgesics, and
humane endpoints during in vivo ocular irritation safety testing.The review concluded that
the use of topical anesthetics and systemic analgesics could avoid most or all pain and
distress without affecting the outcome of the test, and recommended that these substances
should always be used. Topical anesthetics, systemic analgesics, and humane endpoints
should be routinely used during acute eye irritation and corrosion in vivo testing. Exceptions
to their use should be justified. The refinements described in this proposal will substantially
reduce or avoid animal pain and distress in most testing situations where in vivo ocular safety
testing is still necessary.
BALANCED PREEMPTIVE PAIN MANAGEMENT SHOULD INCLUDE:
Routine pretreatment with a topical anesthetic (e.g., proparacaine or tetracaine) and a
systemic analgesic (e.g. buprenorphine).
Routine post-treatment schedule of systemic analgesia (e.g., buprenorphine and
meloxicam). Scheduled observation, monitoring, and recording of animals for clinical
signs of pain and/or distress, and
Scheduled observation, monitoring, and recording of the nature, severity, and
progression of all eye injuries.
No additional topical anesthetics or analgesics should be applied in order to avoid
interference with the study.
Analgesics with anti-inflammatory activity (e.g., meloxicam) should not be applied
topically, and doses used systemically should not interfere with ocular effects.
PRINCIPLE OF THE IN VIVO TEST
Following pretreatment with a systemic analgesic and induction of appropriate topical
anesthesia, the substance to be tested is applied in a single dose to one of the eyes of the
experimental animal; the untreated eye serves as the control. The degree of eye
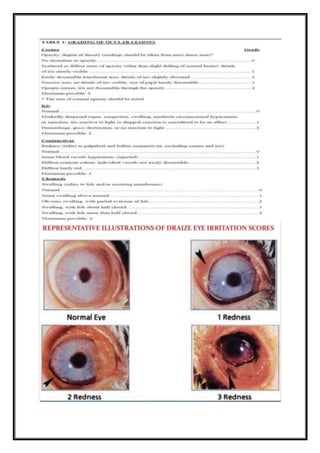
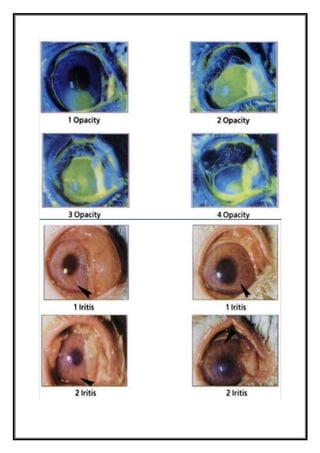
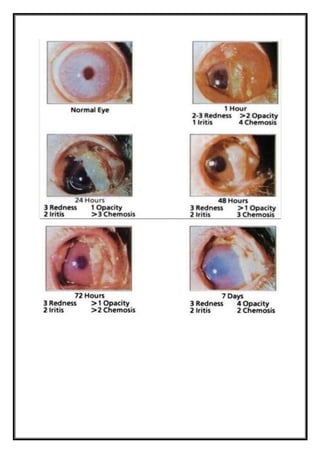
irritation/corrosion is evaluated by scoring lesions of conjunctiva, cornea, and iris, at
specific intervals. Other effects in the eye and adverse systemic effects are also described](https://image.slidesharecdn.com/acuteeyeirritation1-201225045831/85/Acute-eye-irritation-1-3-320.jpg)