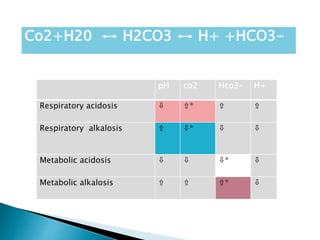
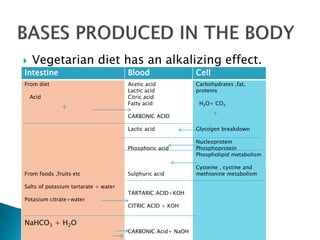
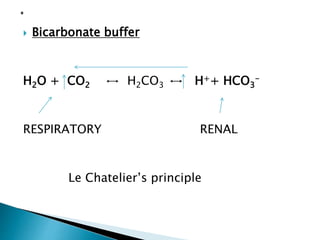
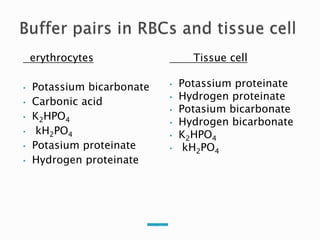
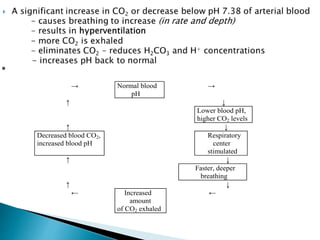
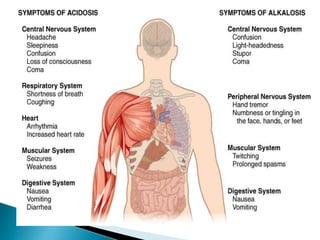
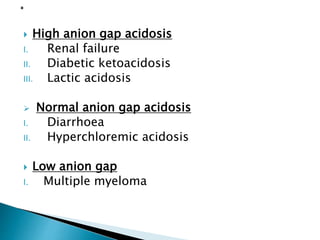
This document discusses acid-base balance and disorders. It covers 3 key mechanisms to maintain blood pH: 1) blood buffers, 2) respiratory regulation, and 3) renal regulation. The blood's bicarbonate buffer system uses carbonic acid, while tissues also use phosphate and protein buffers. Respiration controls pH by regulating CO2 exhalation. The kidneys compensate for acid-base imbalances over hours by regulating bicarbonate reabsorption and acid excretion. Acid-base disorders include respiratory and metabolic acidosis and alkalosis.








![ Acid add [ H+ ] therefore ↑ [ H+ ] - ↓ pH
Base bind [ H+ ] therefore ↓ [ H+ ] - ↑ pH .
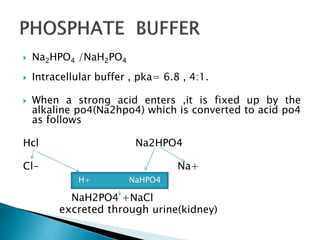
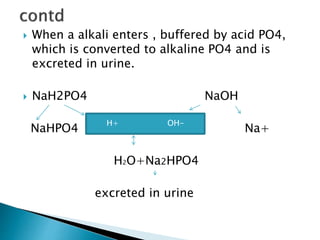
Phosphate buffer
H2PO4 ↔ HPO4
2- + H+](https://image.slidesharecdn.com/acidbasebalance-150216071333-conversion-gate02/85/Acid-base-balance-10-320.jpg)




![ pH = - log [H+] , dimensionless quantity.
[H+] means gm of hydrated H+ ion present as
H3O+ per litre of fluid .
E.g. H2O contains 1/1000000gm of hydrogen ion
in 1 litre ,means [H+]=10-7.
Decrease of one pH unit represents a ten fold
increase in the H+ activity.
The pH 7.40 corresponds to a hydrogen ion
concentration of 40 nmol/L ( European centre).](https://image.slidesharecdn.com/acidbasebalance-150216071333-conversion-gate02/85/Acid-base-balance-17-320.jpg)




![ Extracellular buffer system of the body
NaHCO3/H2CO3= [ SALT ] / [ACID]
NORMAL RATIO= 20 : 1
Base constituent (HCO3 ) - regulated by the kidney
(Metabolic component)
Acid (H2CO3) - respiratory regulation
( Respiratory component ).](https://image.slidesharecdn.com/acidbasebalance-150216071333-conversion-gate02/85/Acid-base-balance-22-320.jpg)
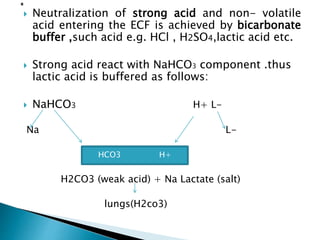
![ H2CO3 H+ + HCO3-
By the law of mass action, at equilibrium
Ka = [H+] [HCO3] --------(1)
[H2CO3 ]
[H+] =Ka [H2CO3 ] -------------(2)
HCO3-
By taking reciprocal and logarithm
Log 1/[H+] = log 1/Ka +log [HCO3] --------(3)
[ H2CO3 ]
Ka = dissociation constant
Ka=log 1/[H+]](https://image.slidesharecdn.com/acidbasebalance-150216071333-conversion-gate02/85/Acid-base-balance-24-320.jpg)
![pH = pka + log [ HCO3 ] -------(4)
[H2CO3 ]
USES;
1. It determines the pH of blood.
2. Serve as an index to understand the
disturbance in acid base balance of the
body.](https://image.slidesharecdn.com/acidbasebalance-150216071333-conversion-gate02/85/Acid-base-balance-25-320.jpg)



























![parameter value
[H+] 35-43 mmol/l
pH 7.35-7.45
pco2 4.5-6.0 kpa
34-45 mmHg
1.02-1.35 mmol/l
po2 10.5-13.5 kpa
Bicarbonate 24-30 mmol/l](https://image.slidesharecdn.com/acidbasebalance-150216071333-conversion-gate02/85/Acid-base-balance-62-320.jpg)