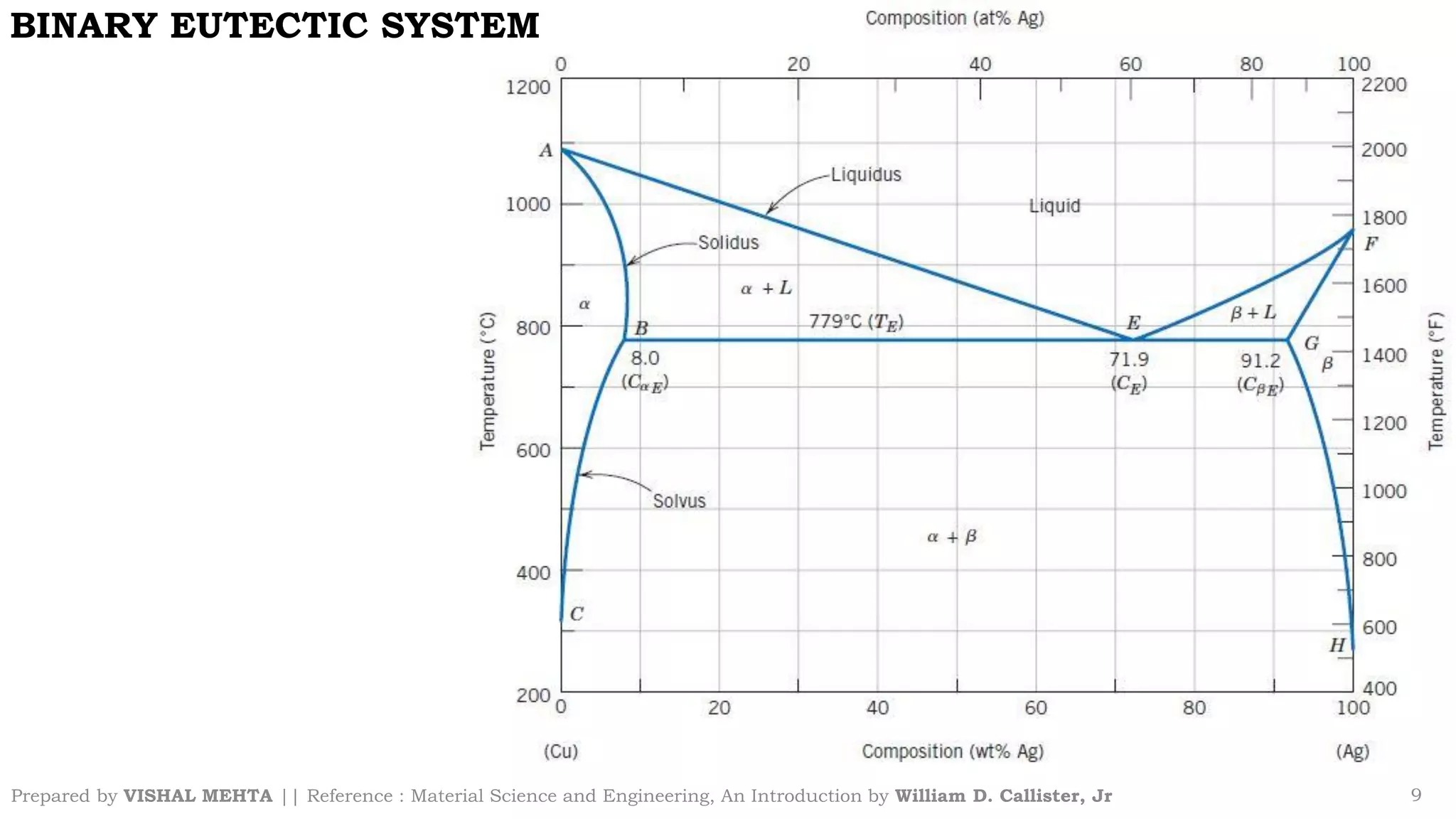
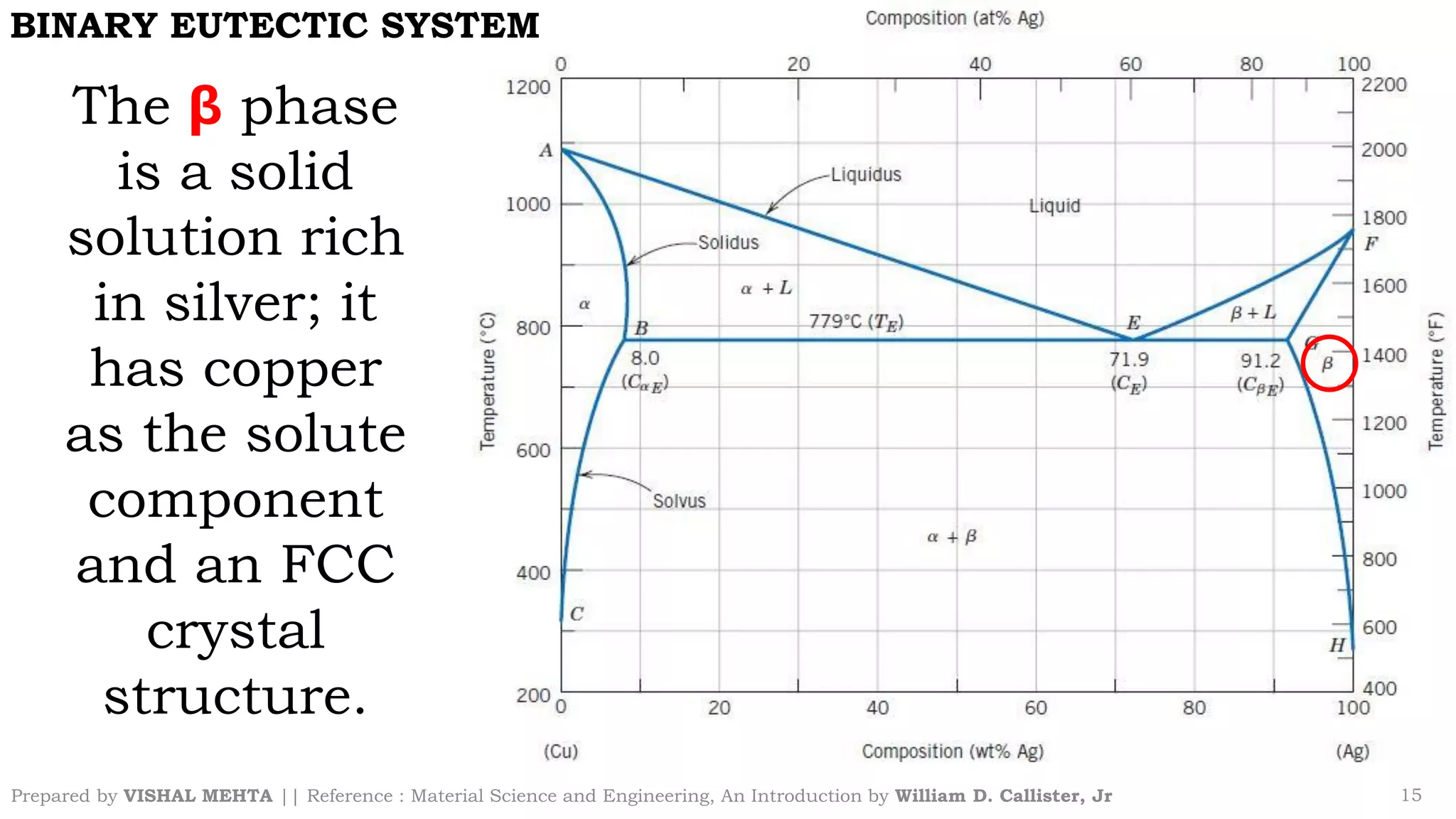
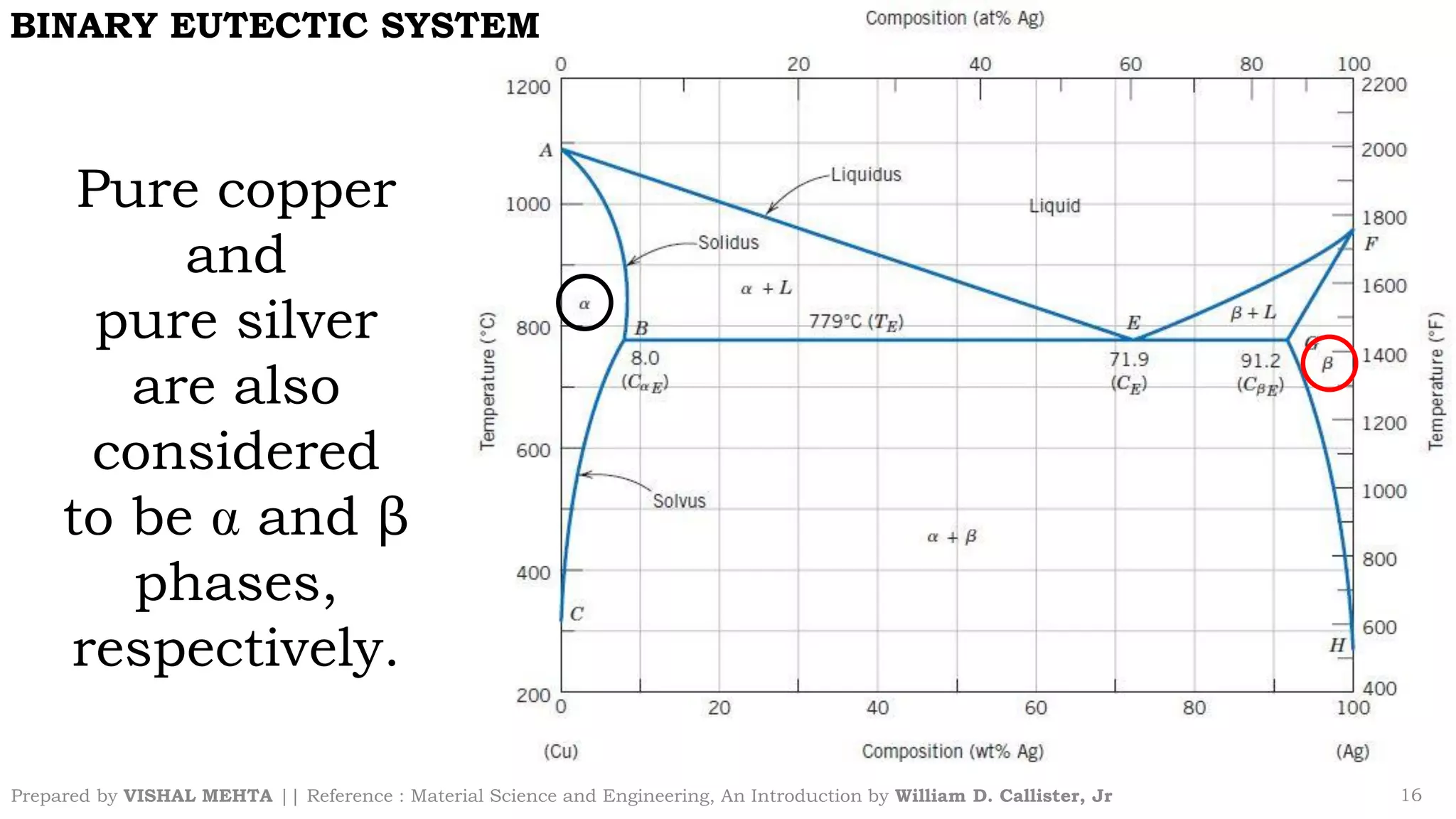
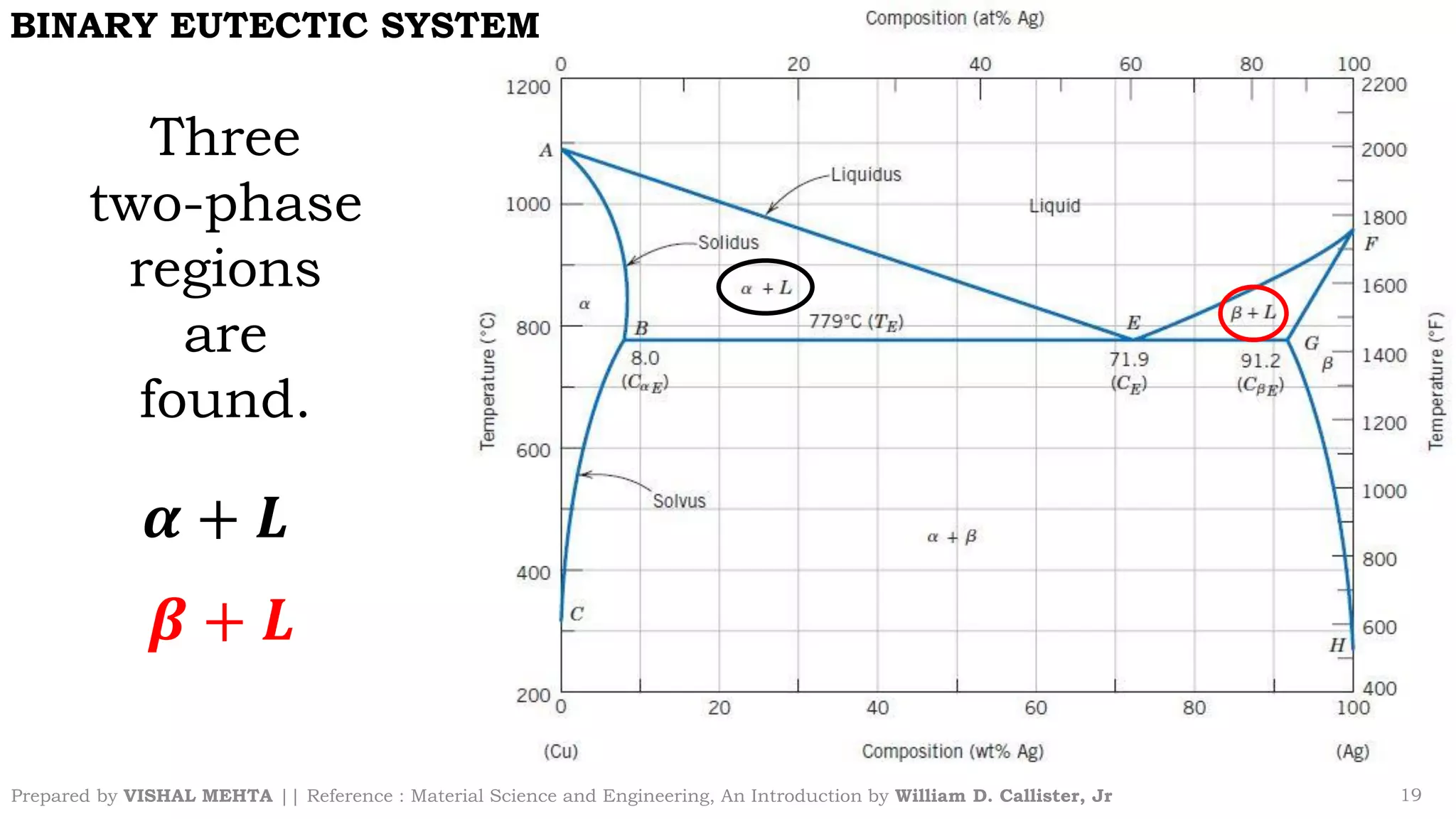
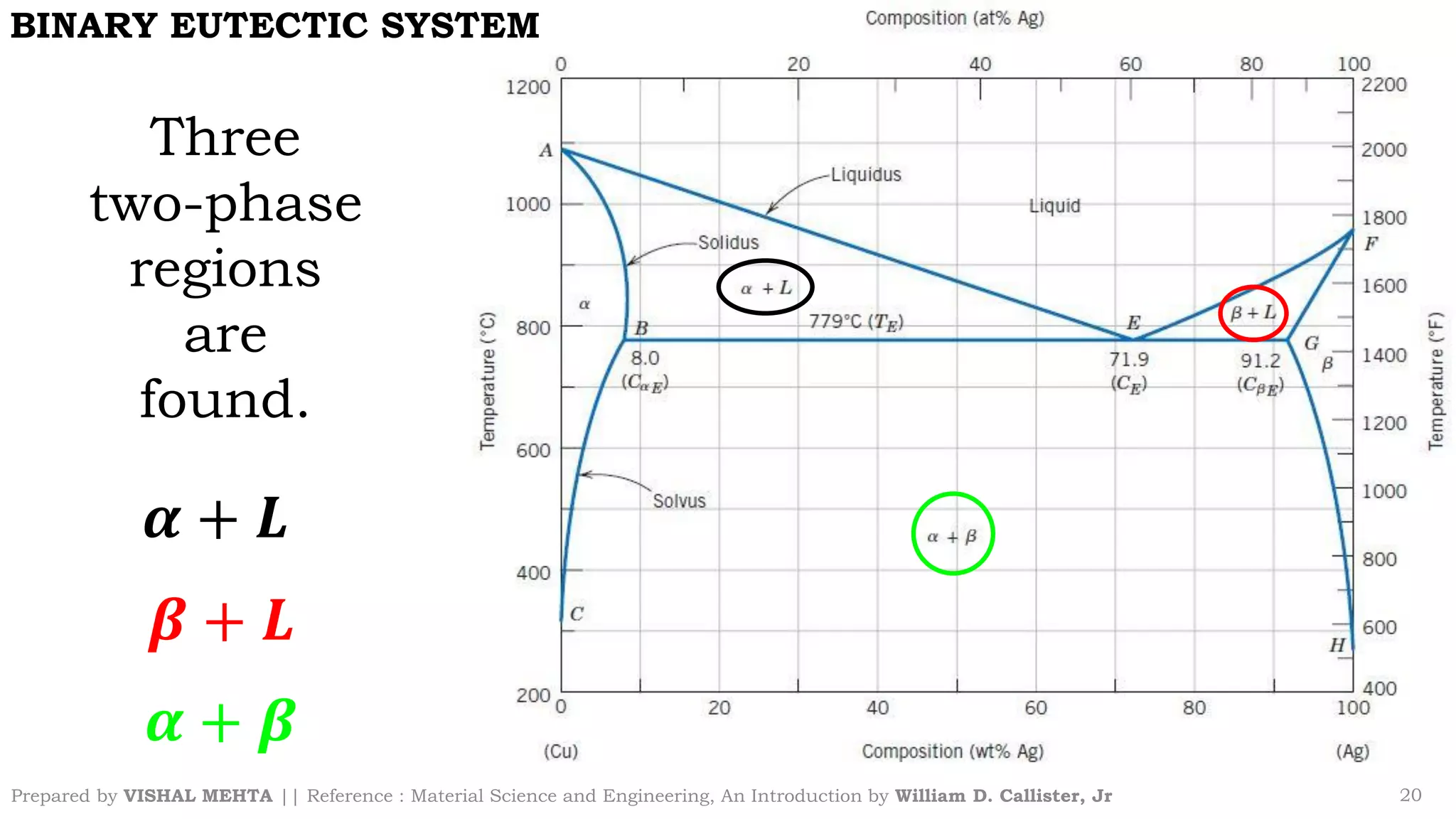
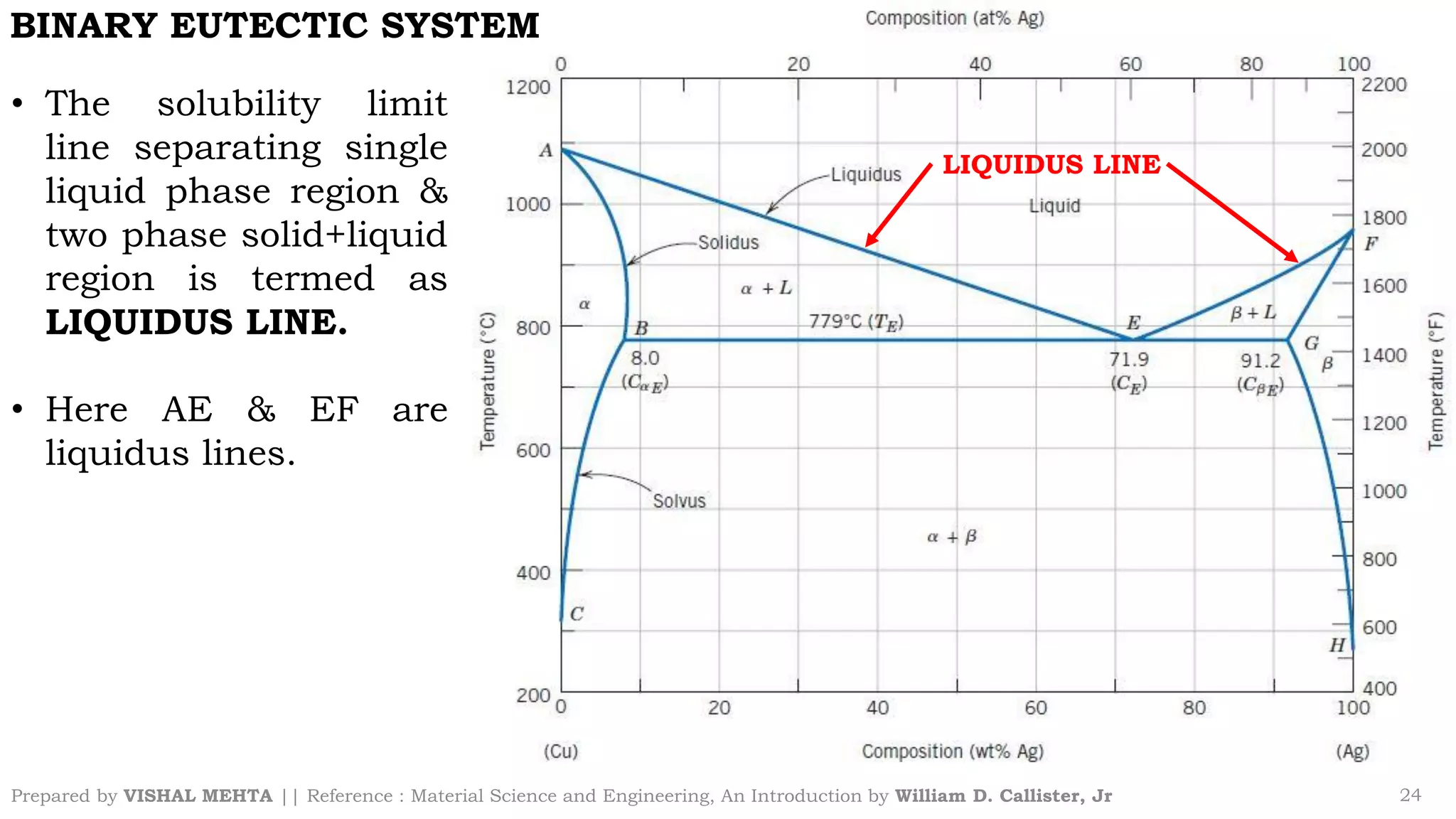
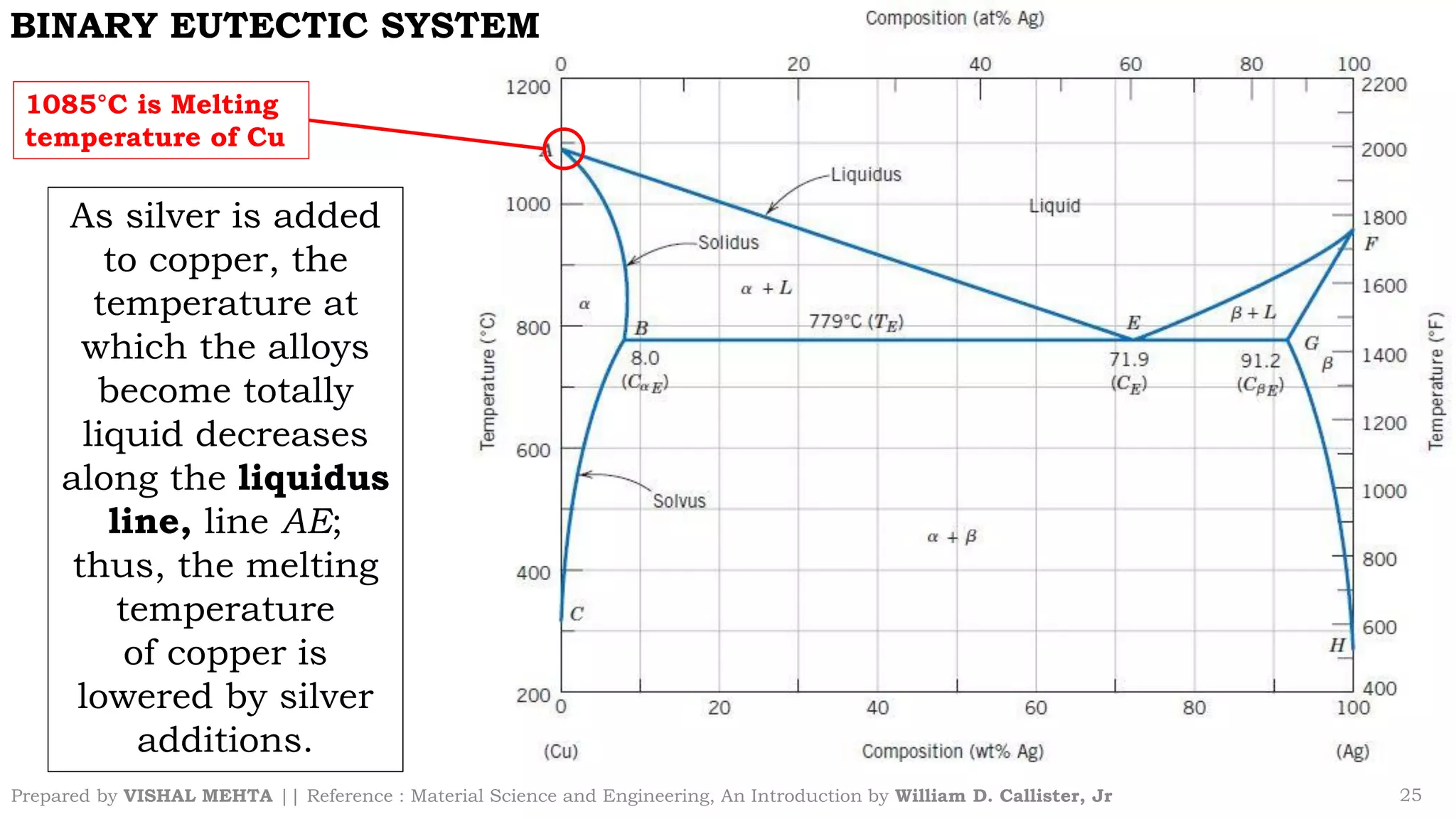
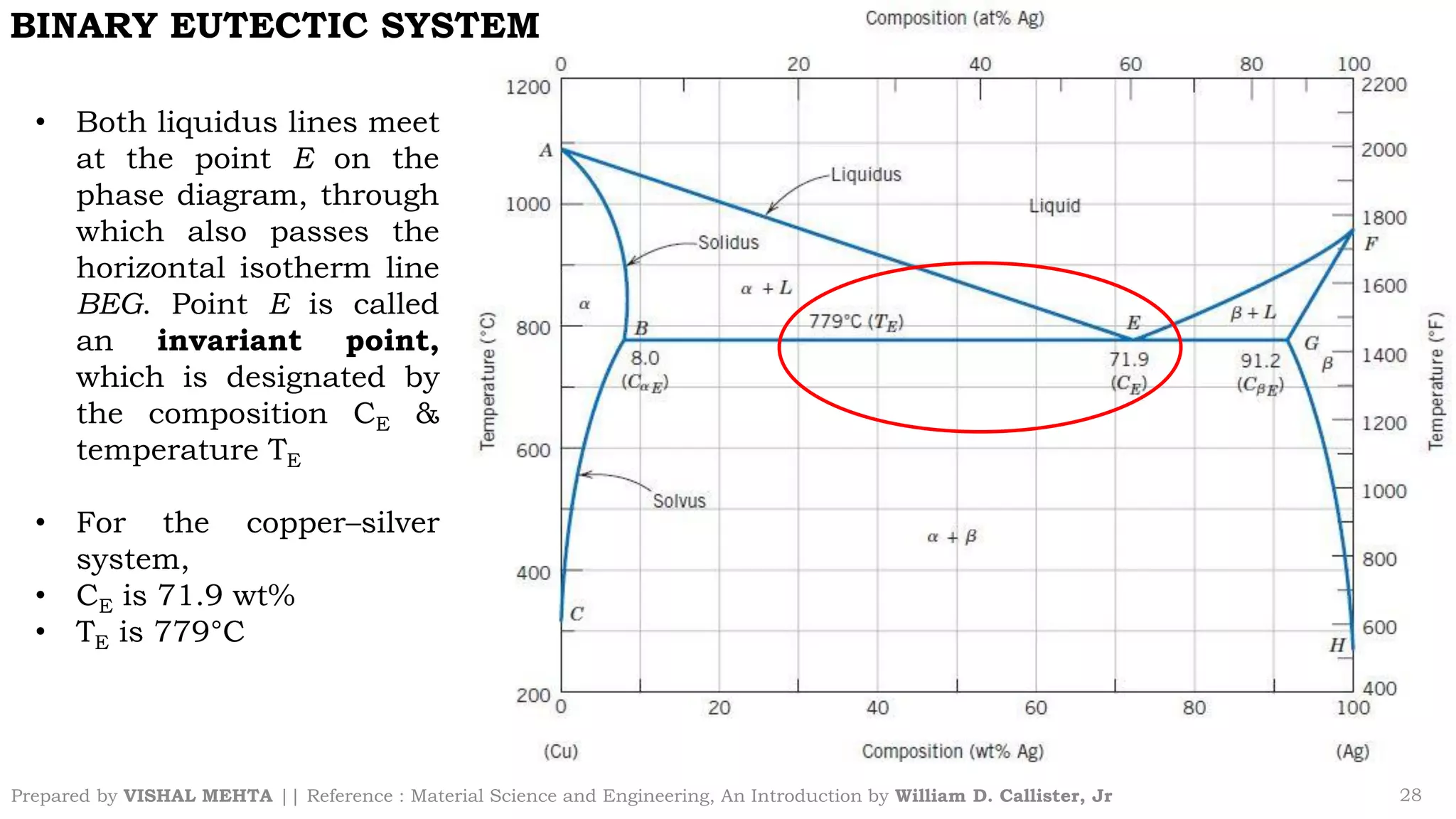
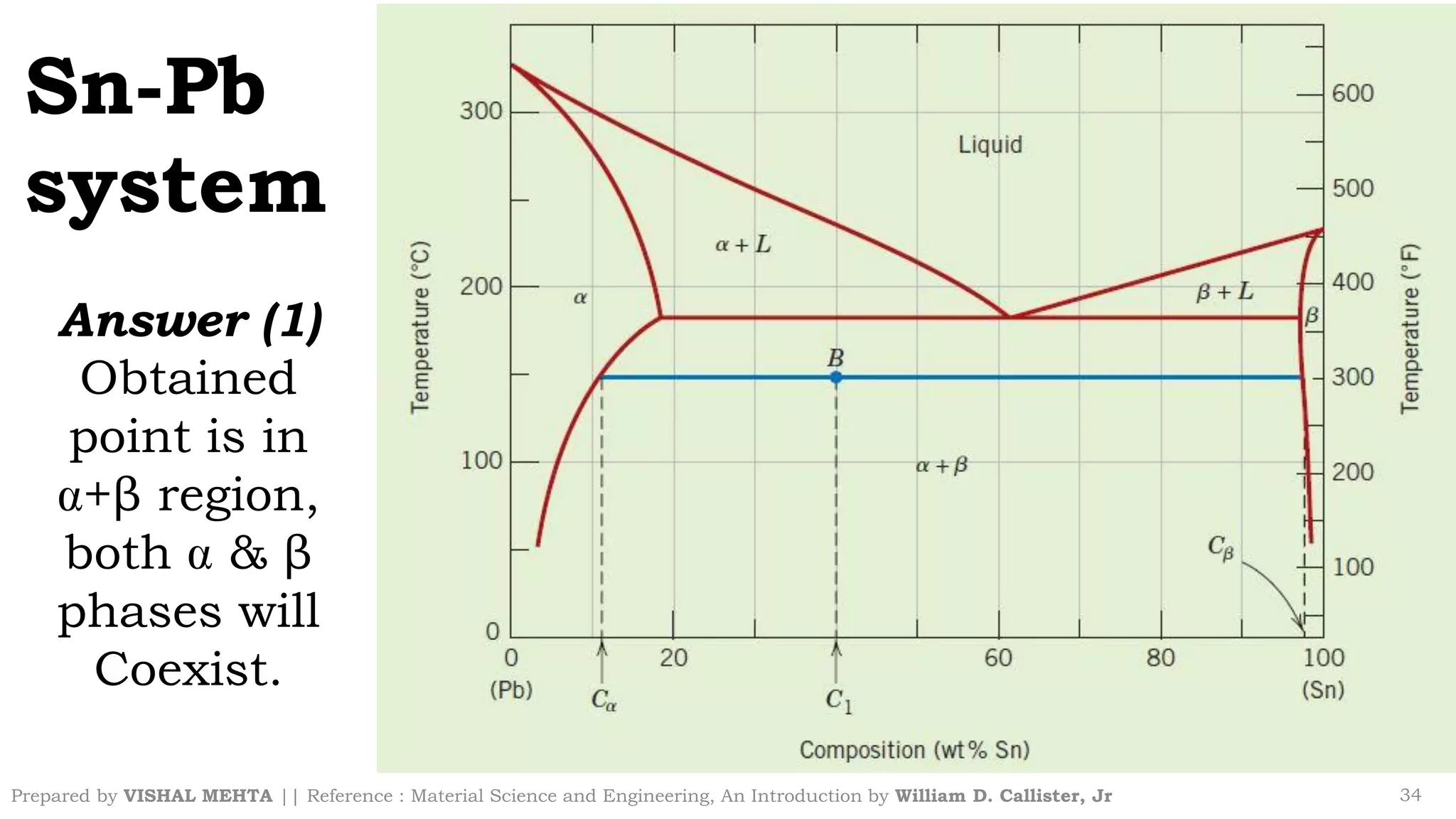
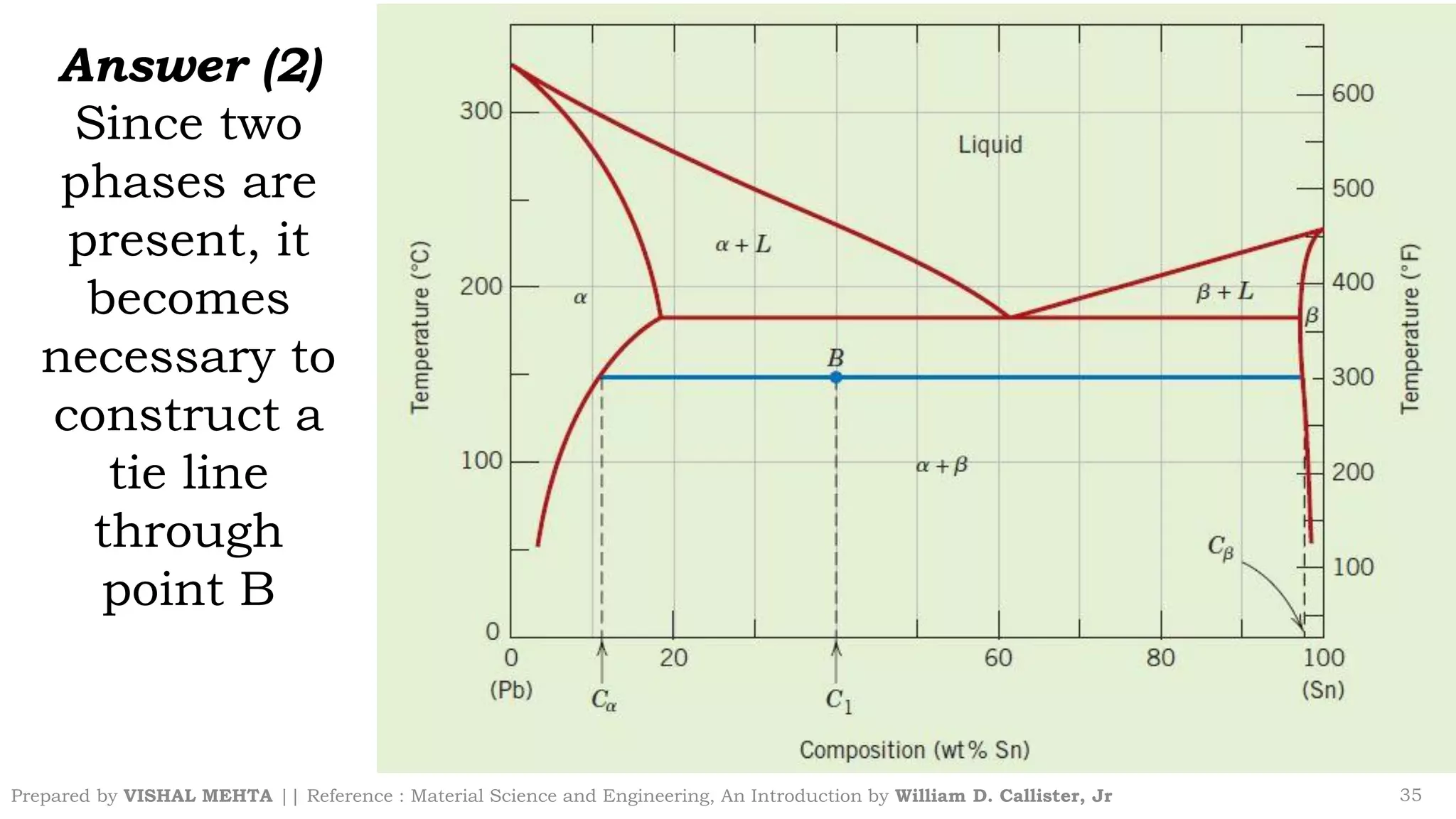
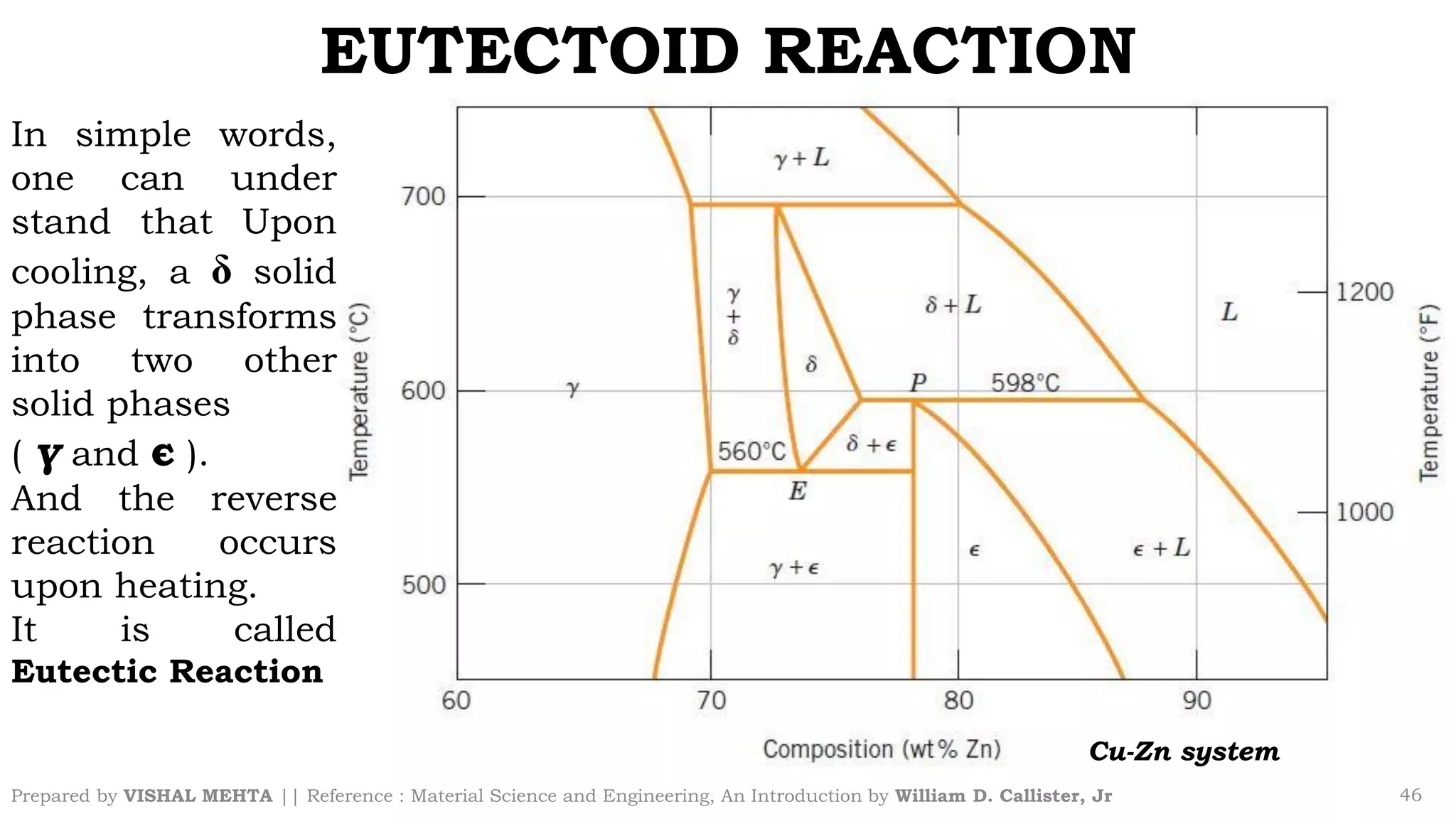
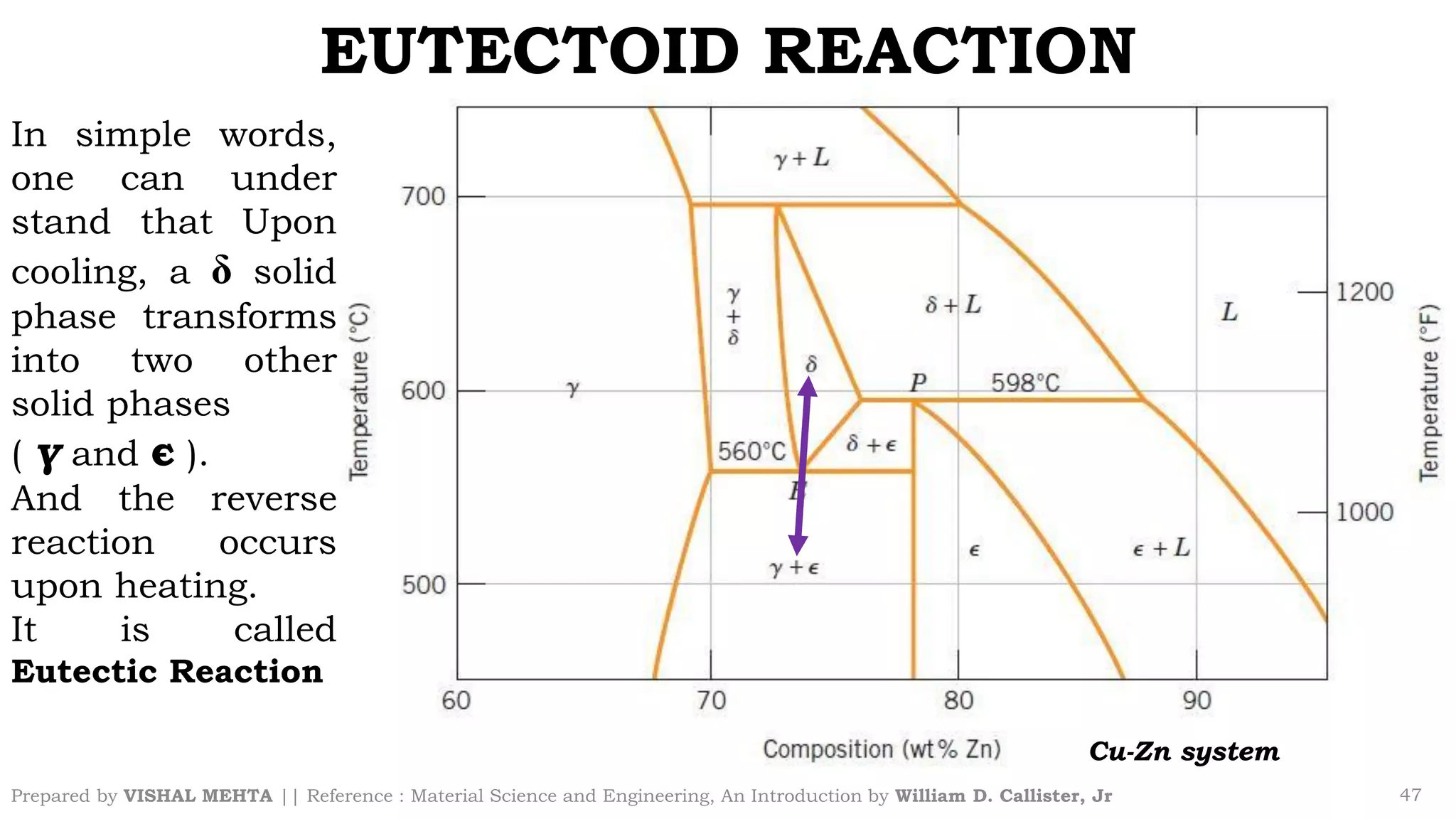
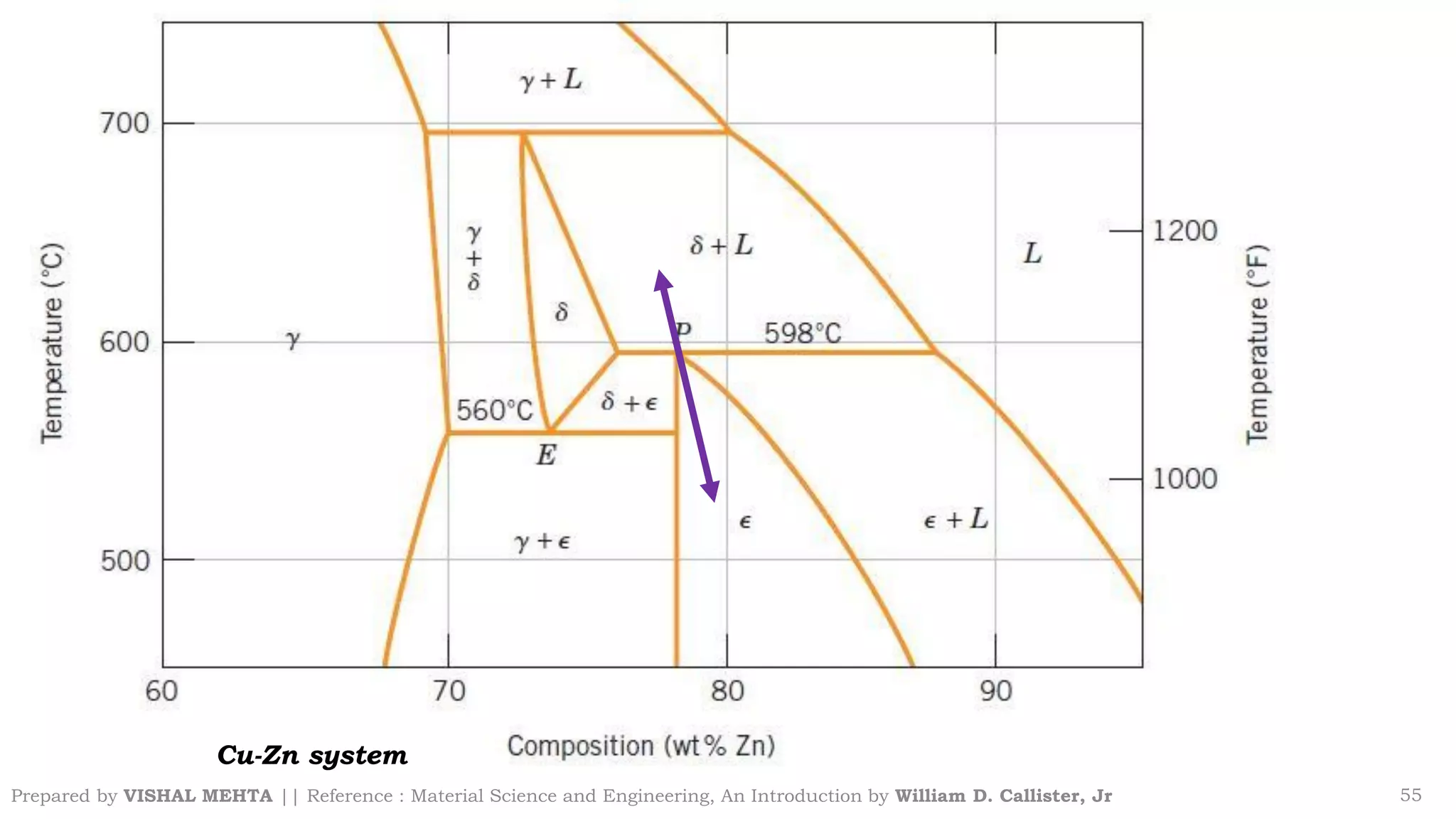
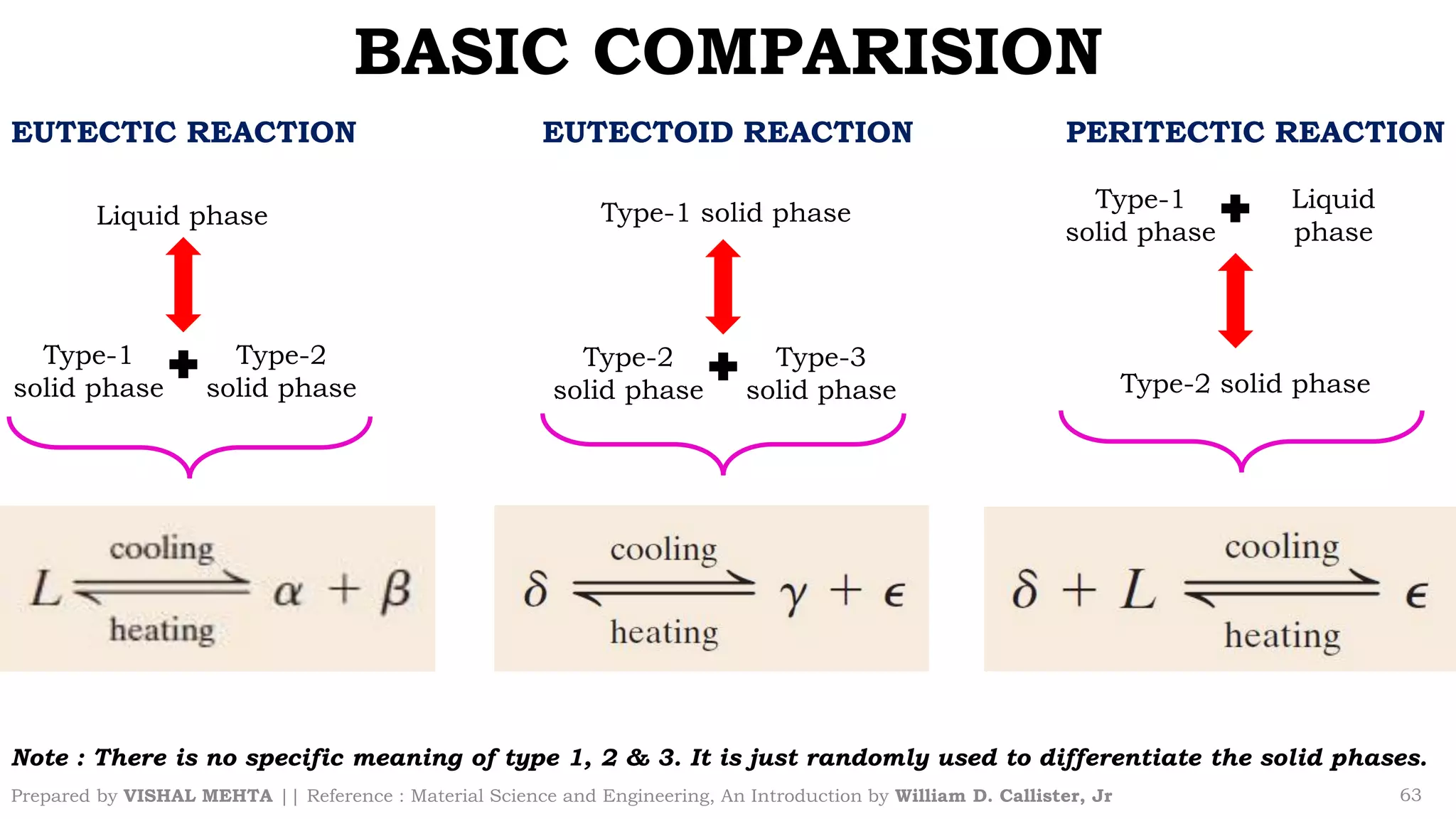
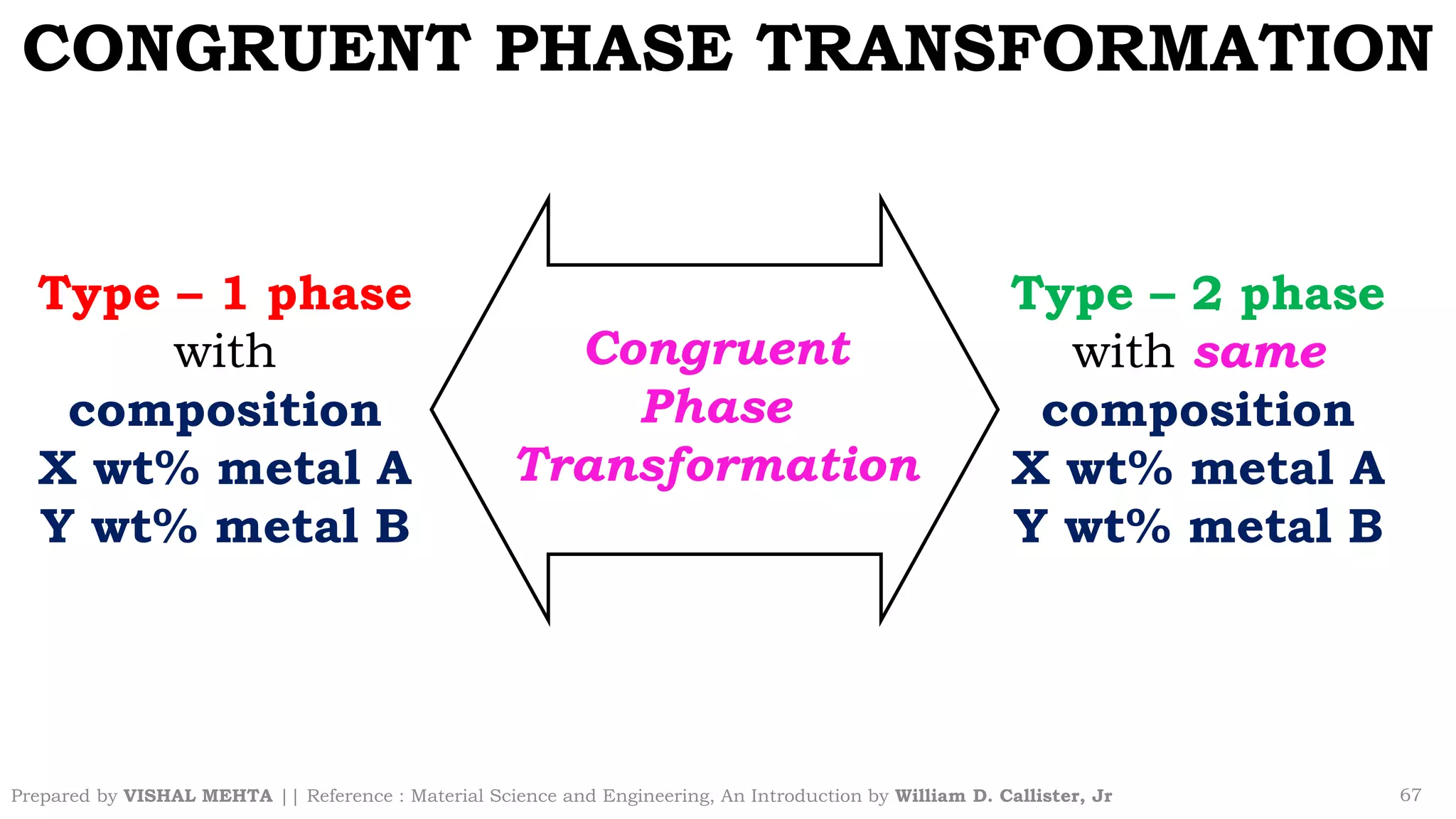
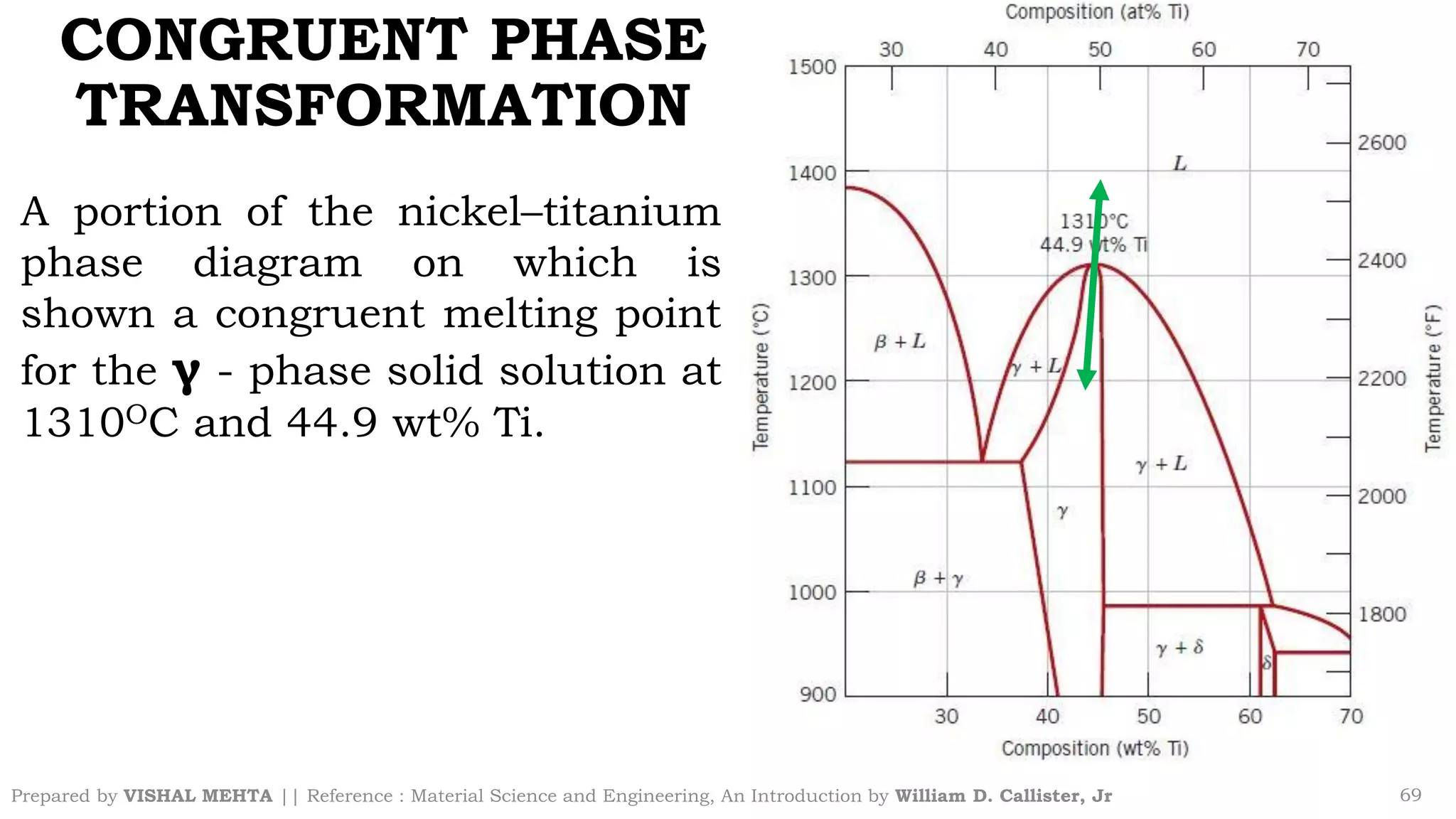
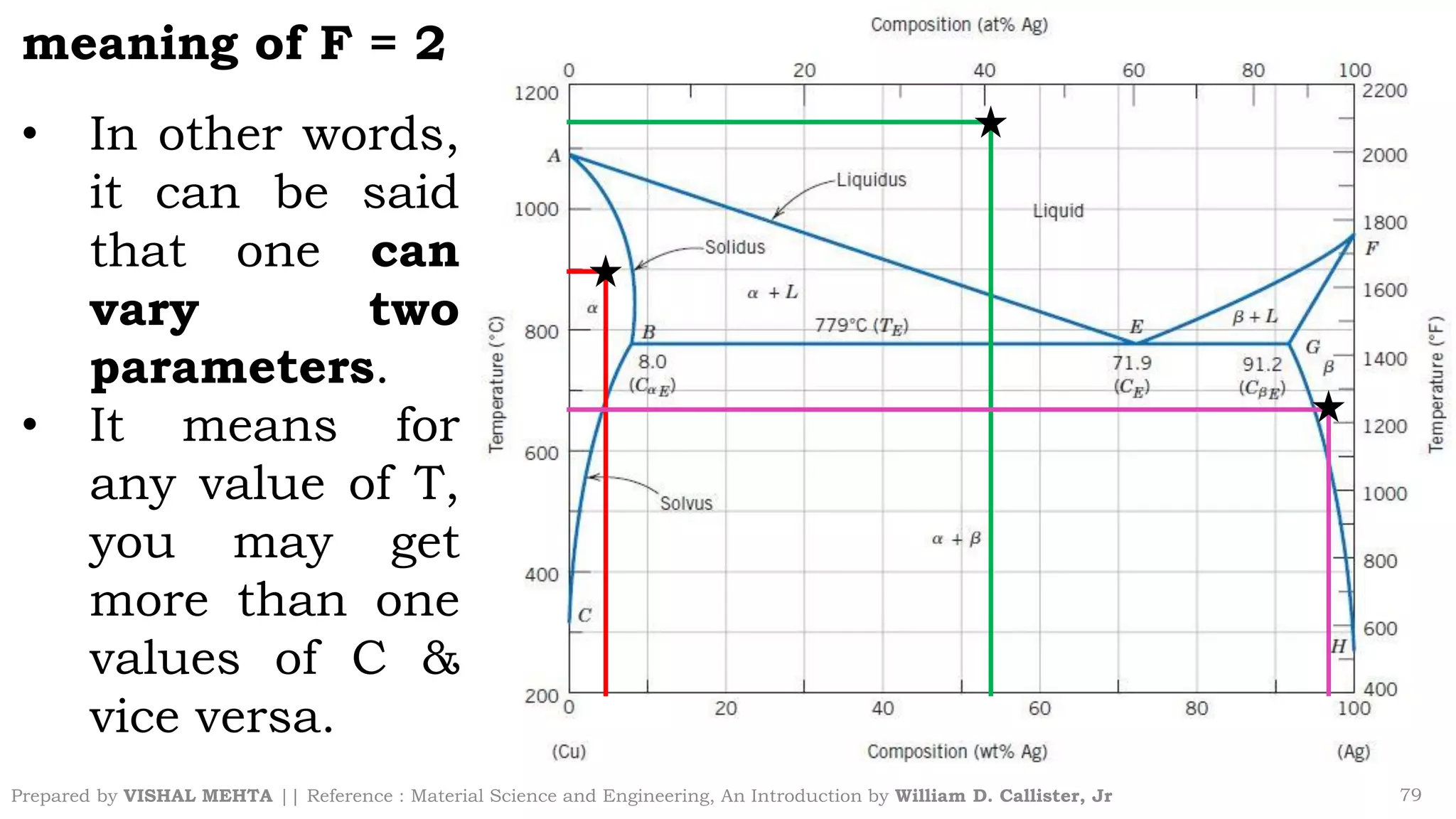
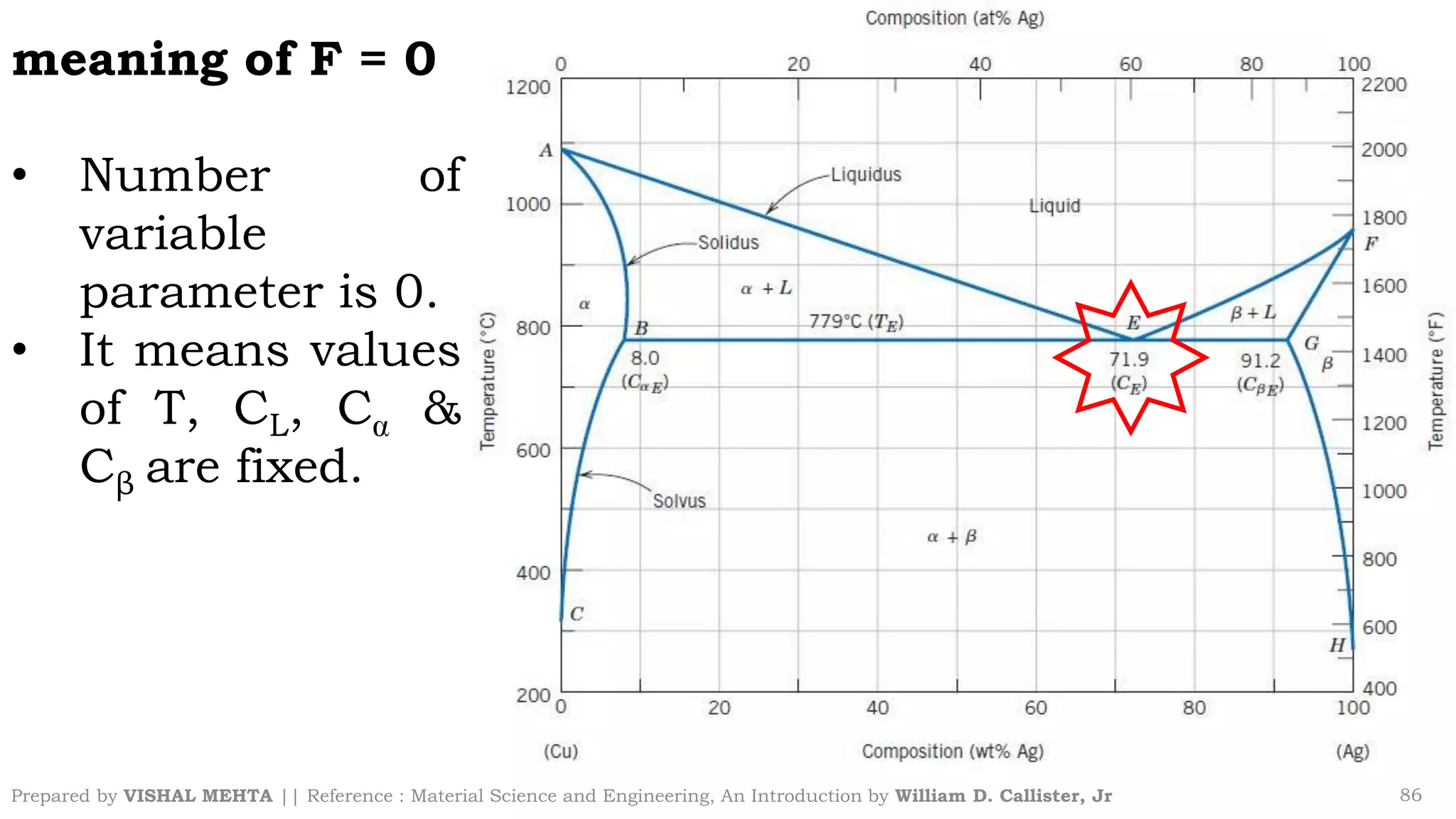
The document summarizes key concepts regarding binary eutectic systems using copper-silver as an example system. It defines terms like eutectic mixture, eutectic reaction, invariant point, liquidus line, solidus line, and solvus line. The copper-silver phase diagram is used to illustrate these concepts, including that at the eutectic point of 71.9% Cu and 779°C, the liquid transforms into solid copper and silver phases upon cooling in a eutectic reaction.