19 152-3-21-2011 colligative properties
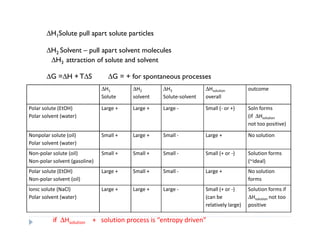
- 1. ∆H1Solute pull apart solute particles ∆H2 Solvent – pull apart solvent molecules ∆H3 attraction of solute and solvent ∆G =∆H + T∆S ∆G = + for spontaneous processes ∆H1 ∆H2 ∆H3 ∆Hsolution outcome Solute solvent Solute-solvent overall Polar solute (EtOH) Large + Large + Large - Small (- or +) Soln forms Polar solvent (water) (if ∆Hsolution not too positive) Nonpolar solute (oil) Small + Large + Small - Large + No solution Polar solvent (water) Non-polar solute (oil) Small + Small + Small - Small (+ or -) Solution forms Non-polar solvent (gasoline) (~ideal) Polar solute (EtOH) Large + Small + Small - Large + No solution Non-polar solvent (oil) forms Ionic solute (NaCl) Large + Large + Large - Small (+ or -) Solution forms if Polar solvent (water) (can be ∆Hsolution not too relatively large) positive if ∆Hsolution + solution process is “entropy driven”
- 2. Like dissolves like # 21 Which likely to be soluble in benzene CCl4 or NaCl Hexane or glycerol Acetic acid or hexanoic acid HCl or propyl chloride
- 3. Temp effect – gases Gas + liquid solution + energy Temp increase increases energy available Favors “dis” solution Thermal pollution – effect on fish
- 4. # 25 and 26 PbCl2 NaOH ∆H for dissociation to aqueous ions Using data from Ch 8 or appendix Will solubility increase with increasing temp?
- 5. Pressure effect on solubility Henry’s law Increased pressure increase gas solubility C g = k Pg Pg = partial pressure of the gas above liquid k – constant – characteristic of particular gas/liquid system Carbonated beverages The bends – Difference in solubility of gases (nitrogen or helium as carrier gas) with increasing P Decompressing after diving Hyperbaric chambers for CO poisoning, wound healing etc (direct solubility) Practice # 29
- 6. Typical Henry’s law problem He in water 3.8 X 10-4 M/atm @ 25 oC (k) ? M/mm Hg pHe =293 mm Hg @ 25 oC, what is [ ] in M C g = k Pg HW: 27, 29 Ch 10
- 7. Colligative properties Due to number of particles in solution Be familiar with Electrolytes (soluble salts, SA, SB) Completely dissociate into ions Non-electrolytes Molecular compounds (no ions) Polar molecular compounds are water soluble Weak electrolytes WA, WB – partially dissociate Note worksheet
- 8. Colligative properties Due to number of particles in solution 1 mole sugar per liter = ? mol/L particles? 1 mole NaCl per liter 1 mole aluminum sulfate per liter 1 mole acetic acid Electrolytes/nonelectrolytes
- 9. Vapor pressure Pressure of the vapor above a liquid or solution – depends on temp and identity Raoult’s law - mix with volatile solute VPsoln = XsoluteVP solute + XsolventVPsolvent evaporation
- 10. Ideal solution of 2 volatile solvents (liquids) 1. What is the VP of pure hexane ___ 2. What is the VP of pure pentane ___ 3. Which is more volatile? 4. If X = 0.5, what is VP due to pentane___ hexane ___total VP _____ Raoult’s Law VPT = XAVPA + XBVPB Related exercise: 1 mol benzene (C6H6), Po = 75 torr 2 mol toluene (C7H8) Po = 22 torr What is the mf of benzene in the mixture? What is the vp of the mixture? What is the mf of benzene in the vapor phase? Data is for a given temp since pentane is a liquid at RT, this must be for a higher T
- 11. VP lowering Non-volatile solutes decrease vp Independent of nature of solute Dependent on number of particles in solution Raoult’s law -- except solute does not exert vp (non- volatile) VPsolution = Xsolvent VPsolvent VPlowering = Xsolute Vpsolvent VPsolution + Vplowering = VPsolvent
- 12. VPsolution = Xsolvent VPsolvent VPlowering = Xsolute VPsolvent VPsolution + VPlowering = VPsolvent Normally used with non-electrolytes # 35 Note: mole fraction concentration unit used
- 13. Boiling point elevation If solutes depress vp, to boil (vp = 760) temp must be elevated Depends on concentration of particles in solution and on solvent Use molality mol solute particles/kg solvent BP elevation = k i m k= bp constant for solvent i= activity of solute (# particles/mol) m = molality of solute (mol solute/kg solvent) 31,32 (bp’s) Lab Wed benzoic acid in lauric acid
- 14. Freezing point depression Solutes interfere with crystal formation Salt added to ice – interferes with crystal structure, ice structures breaks down – endothermic process!!!! Ocean water – ice bergs are fresh water --salt is not included 1.86oC/mole of particles in 1 kg water Melting ice 31, 32 General Chemistry Online: FAQ: Solutions: Why does salt melt ice?
- 15. Review spec lab end of class Assignments due Make sugar solns Spec lab: Calc k Calc energies Calc unk conc Emitted and abs colors Resubmit HW Ch 10 28-30 (Henry’s) 39 40 42-44 (fp, bp)