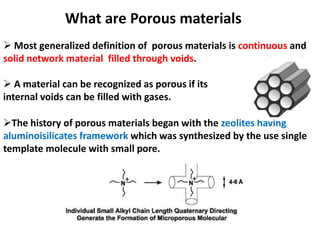
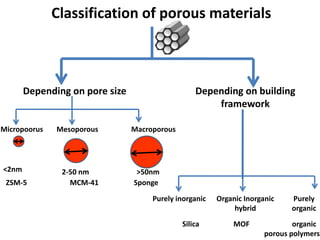
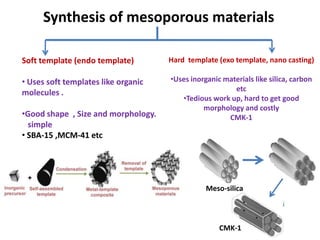
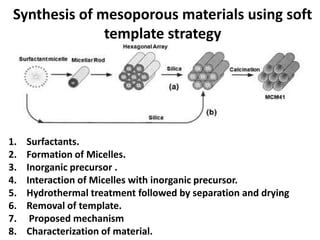
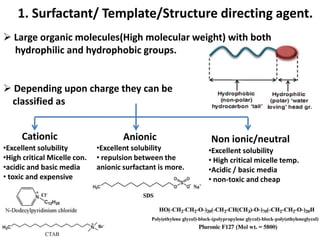
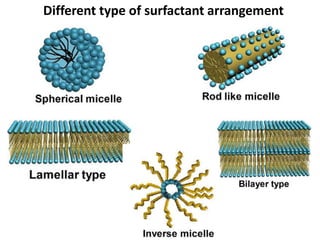
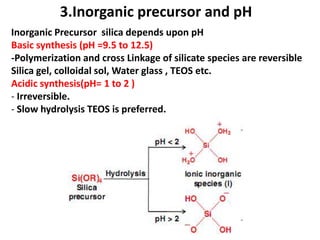
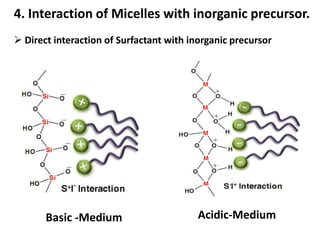
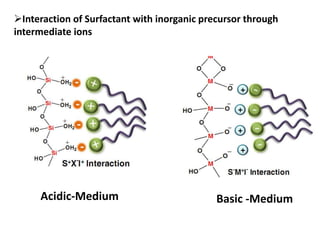
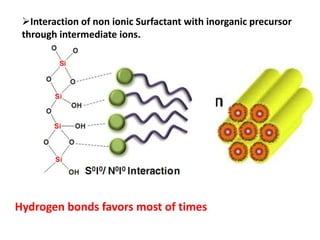
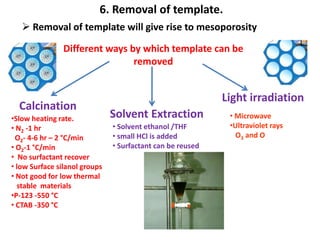
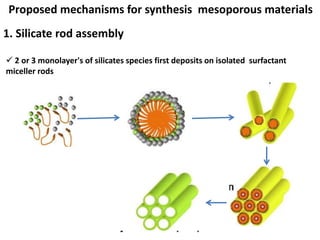
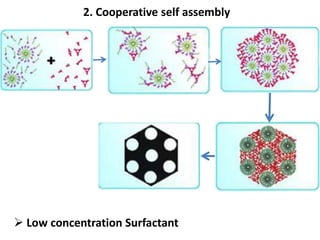
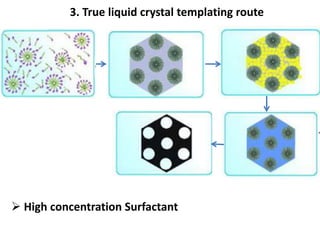
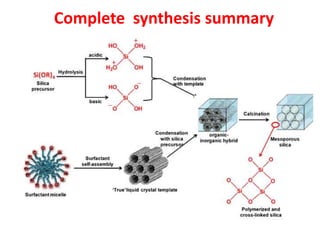
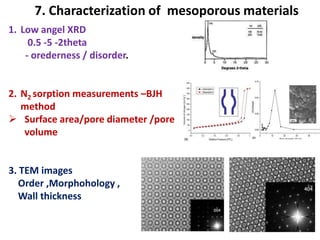
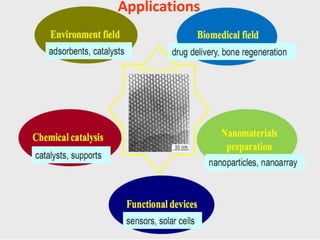
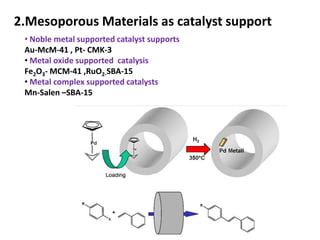
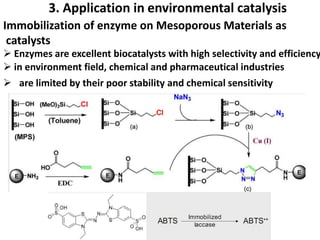
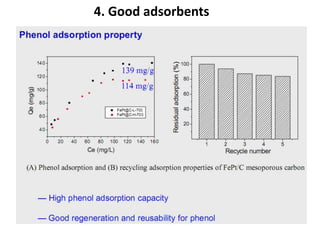
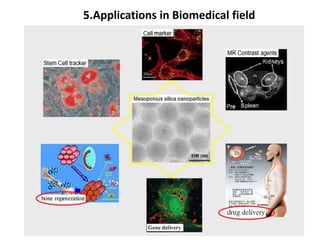
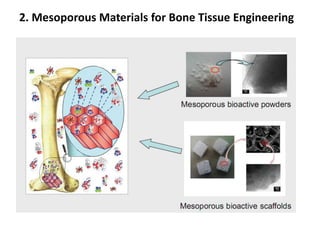
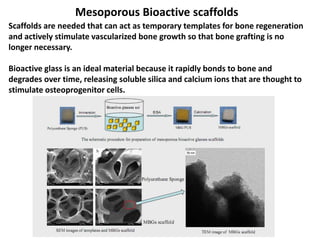
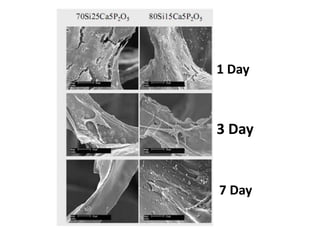
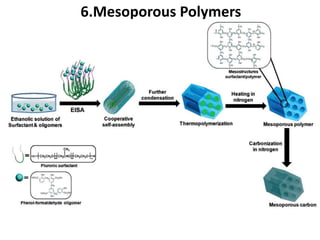
The document discusses mesoporous materials, detailing their synthesis through soft template strategies and their applications in various fields such as catalysis, environmental science, and biomedical engineering. Mesoporous materials have specific characteristics like large surface area, high stability, and are useful as drug delivery systems and in bone tissue engineering. The paper also compares zeolites and mesoporous materials, highlighting differences in properties and synthesis methods.