DNA Vaccine.pptx
- 1. Dr. Rachana Choudhary Department of Microbiology Shri Shankaracharya Mahavidyalya,Junwani Bhilai DNA VACCINE
- 2. CONTENTS •Introduction •History •Active & Passive Immunization •Types of Vaccines •DNA Vaccine •Advantages & Disadvantages of DNA Vaccines •Conclusion •References
- 3. INTRODUCTION Vaccination is the phenomenon of preventive immunization. Term Vaccination ( Latin:vacca – cow) is so named because the first vaccine was derived from a virus affecting cows relatively begins cowpox virus which provides a degree of immunity to smallpox, a contagious and deadly disease. Traditional vaccines have relied on either live- replicating or nonliving preparations of microorganisms. The injected material functions as a vaccine by generating immunity against the inoculum, and the resulting immune responses function to prevent disease.
- 4. HISTORY 1. The history of vaccine use dates back to ancient times. In the 7th century, Indian Buddhist drank snack venom to protect themselves from snake bite. 2. In 9th century China, the correct treatment of smallpox was written by Buddhist nun. The manuscript recommended that a mixture of ground dried smallpox scabs and herbs be blown into the nostrils of children. 3. It was on May 14, 1796, that Jenner extracted the contents of a pustule from the arm of a cowpox-infected milkmaid, Sarah Nelmes, and injected it into the arm of eight-year- old James Phipps. 4. A vaccine meeting held at the Cold Spring Harbor Laboratory in September 1992 described the use of DNA immunization to generate humoral and cellular immune responses against a human pathogen as well as protection from both tumors and viral challenges in animal system.
- 5. Active and Passive Immunization Immunity to infectious microorganisms can be achieved by active or passive immunization. In each case, immunity can be acquired either by natural processes or by artificial means such as injection of antibodies or vaccines. Passive immunization, in which preformed antibodies are transferred to a recipient, occurs naturally by transfer of maternal antibodies across the placenta to the developing fetus. Active immunization can be achieved by natural infection with a microorganism, or it can be acquired artificially by administration of a vaccine
- 6. Acquisition of passive and active immunity Type Acquired Through Passive Immunity Natural maternal antibody Immunoglobulin Humanized monoclonal antibody Antitoxin Active Immunity Natural infection Vaccines Attenuated organisms Inactivated organisms Purified microbial macromolecules Cloned microbial antigens Expressed as recombinant protein As cloned DNA alone or in virus vectors Multivalent complexes Toxoid
- 7. Designing Vaccines for Active Immunization The adaptive immune response provides a more flexible resistance to pathogens. Vaccination educates the adaptive immune system, preparing it to deal effectively and swiftly with pathogen not readily eliminated by innate immunity. Several factors must be kept in mind in developing a successful vaccine. First and foremost, vaccine must determine differences between activation of the humoral and the cell mediated branches. A second factor is the development of immunologic memory.
- 8. 1. Live attenuated Vaccine 2. Inactivated “killed” Vaccine 3. Subunit Vaccine 4. Toxoid Vaccine 5. DNA Vaccine 6. Recombinant Vector Vaccine
- 9. Classification of common vaccines for humans Disease or Pathogen Type of Vaccine Whole Organism Bacterial cell Anthrax Plague Tuberculosis Typhoid Inactivated Inactivated Live attenuated BCG Live attenuated Viral Particle Hepatitis A Influenza Measles Mumps Inactivated Inactivated Live attenuated Live attenuated Purified Macromolecules Toxoids Diphtheria Tetanus Capsular polysaccharides Haemophilus influenzae type b Streptococcus pneumoniae Surface antigen Hepatitis B Inactivated exotoxin Inactivated exotoxin Polysaccharide protein carrier 23 distinct capsular polysaccharides Recombinant surface antigen (HBsAg)
- 10. Characteristic Attenuated vaccine Inactivated vaccine DNA vaccine Production Selection for avirulent organisms: virulent pathogen is grown under adverse culture conditions or prolonged passage of a virulent human pathogen through different hosts Virulent pathogen is inactivated by chemicals or irradiation with γ- rays Easily manufactured and purified Booster requirement Generally requires only a single booster Requires multiple boosters Single injection may suffice Relative stability Less stable More stable Highly stable Type of immunity induced Humoral and cell-mediated Mainly humoral Humoral and cell- mediated Reversion tendency May revert to virulent form Cannot revert to virulent form Cannot revert
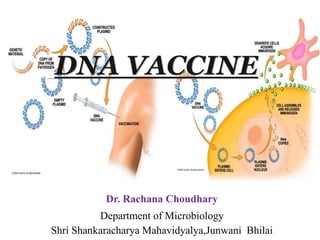
- 11. • DNA vaccines consist of plasmids that contains genes for certain types of antigens. • Once administered, the plasmid is taken up by the target cell and the genes are expressed. • The cell then either excretes the antigen or displays it on an MHC-I molecule. • DNA Vaccine constructs are produces as small circular vehicles or plasmids . These plasmids are constructed with a promoter site, which starts the transcription process DNA Vaccine construct consisting of a mammalian expression vector. The plasmids are constructed with a promoter, an antigenic DNA sequence, and a mRNA stop site containing the poly A tract.
- 12. Genetic immunization by using DNA vaccines is a novel approach that came into being in 1990. A DNA vaccine consists of a gene encoding an antigen protein, inserted onto a plasmid, and then incorporated into the cells in a target animal. The plasmid carrying DNA vaccine normally contains a promoter site, cloning site for the DNA vaccine gene, origin of replication, a selectable marker sequence (e.g. a gene for ampicillin resistance and a terminator sequence a poly A tail )
- 13. Mechanism of DNA Vaccines In body’s immune system, cells need to process and present antigenic peptides to lymphocytes in order to stimulate antigen- specific immune response. Thus, antigen must be processed and presented to T lymphocyte by antigen – presenting cells (APC’s). Four primary components are critical in the professional APC’s ability to present the antigen to T- cells and activate them for appropriate immune response. These components are as following- a) MHC – antigen complexes. b) Costimulatory molecules (primarily CD80 & CD86). c) Intracellular adhesion molecules. d) Soluble cytokines.
- 14. DNA Vaccine for HIV – 1 • The HIV-1 is a retrovirus, which preferentially infects and kills CD4+ T-cells and macrophages, ultimately resulting in immune system failure and multipathogen infections. • Within the HIV genome, there are several potential immunological targets for DNA vaccination. The HIV-1 genome is organized into 3 major structural and enzymatic genes, 2 regulatory genes and 4 accessory genes. • The first major gene target is env, which codes for the outer viral envelope proteins. HIV enters the CD4 receptor complex following entry of HIV viral core, synthesis of a ds DNA version of the HIV genome begins by the viral DNA polymerase reverse transcriptase.
- 15. DNA Vaccines for Cancer Tumor-associated antigens (TAA) are proteins produced by tumor cells that can be presented on the cell surface in the context of MHC complexes. Immunological effects of 2 such TAAs: carcinoembryonic antigen(CEA) and prostatespecific antigen(PSA). 1.DNA Vaccine Strategies using CEA- The immune response to nucleic acid vaccination using a CEA insert was cloned into vector containing the cytomegalovirus(CMV) early promoter /enhancer and injected intramuscularly. These responses were detected in the immunized mice. 2.DNA Vaccine Strategies using PSA- The immune responses induced by a DNA vaccine encoding for human PSA has been investigated in a murine model. The vaccine construct was constructed by cloning a gene for PSA into expression vectors under control of a CMV promoter.
- 16. Safety and Efficacy Studies in Humans • The ultimate goal of vaccine development is to demonstrate safety and efficacy in humans. The first vaccine studies to enter the clinic were DNA vaccines encoding for HIV-1 envelope. • 15 healthy HIV-1 seropositive volunteers with >500 CD4+ lymphocytes/ml were enrolled in this study. Patients in the trial received 3 injections, each separated by 10 weeks, with escalating dosage (3 dosage groups of 5 subjects) of envelope vaccine. • Preliminary results reveal no significant clinical or laboratory adverse effects measured in any of the dosage groups (30,100,300µg). • Low doses of a single immunogen DNA vaccine is capable of augmenting both existing humoral and cellular immune responses in humans.
- 17. Advantages of DNA Vaccine • Subunit vaccination with no risk for infection. • Antigen presentation by both MHC class I and class II molecules. • Able to polarize T-cell help toward type 1 or type 2. • Immune response focused only on antigen of interest. • Ease of development and production. • Stability of vaccine for storage and shipping • Cost-effectiveness • Obviates need for peptide synthesis, expression and purification of recombinant proteins and the use of toxic adjuvants. • In vivo expression ensures protein more closely resembles normal eukaryotic structure, with accompanying post-translational modifications.
- 18. Disadvantages of DNA Vaccine • Limited to protein immunogens (not useful for non-protein based antigens such as bacterial polysaccharides). • Risk of affecting genes controlling cell growth. • Possibility of inducing antibody production against DNA. • Possibility of tolerance to the antigen (protein) produced. • Potential for atypical processing of bacterial and parasite proteins.
- 19. Conclusions 1. DNA immunization holds great promise for providing safe inexpensive vaccines for many infectious pathogens, including HIV-1. 2. DNA vaccine constructs for cancer-targeting tumor-specific antigens have also been studied in a variety of animal models. 3. DNA vaccines can be combined with other vaccines including recombinant protein, poxvirus, adenovirus as well as others to further enhance initial immune responses. 4. An important problem for developing DNA vaccine is the huge quantity of DNA needed for inducing immune responses in large animals and human. The production and purification of large quantity of plasmid DNA not only would be a financial burden but would increase the risks of side effects of DNA immunization, especially causing autoimmune responses or plasmid DNA integration into host cells.
- 20. REFERENCE Immunology Seventh Edition -(David Male , Jonathan Brostoff, David B. Roth, Ivan Roitt). Immunology & Immunotechnology – Ashim K. Chakravarty. Immunology by Kuby Cellular And Molecular Immunology- A.K Abbas Prescott, Herley, Klein’s Microbiology
- 21. THANK YOU