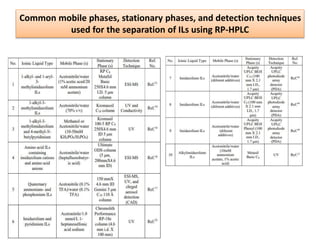
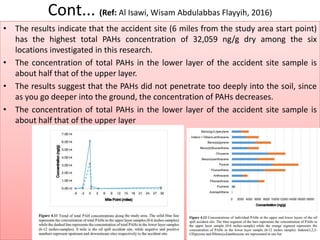
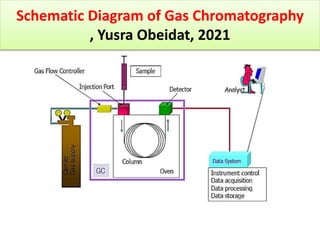
The document discusses advanced chromatographic techniques and their applications in aquatic environmental studies, detailing the history and development of chromatography since its invention by Michael S. Tswett in 1906. It highlights various types of chromatography, such as liquid chromatography (LC), gas chromatography (GC), and their combinations with mass spectrometry (MS), emphasizing their role in analyzing environmental pollutants like polycyclic aromatic hydrocarbons (PAHs). The document also presents a case study involving the analysis of PAHs from soil samples collected at an oil spill site using gas chromatography-mass spectrometry (GC-MS).