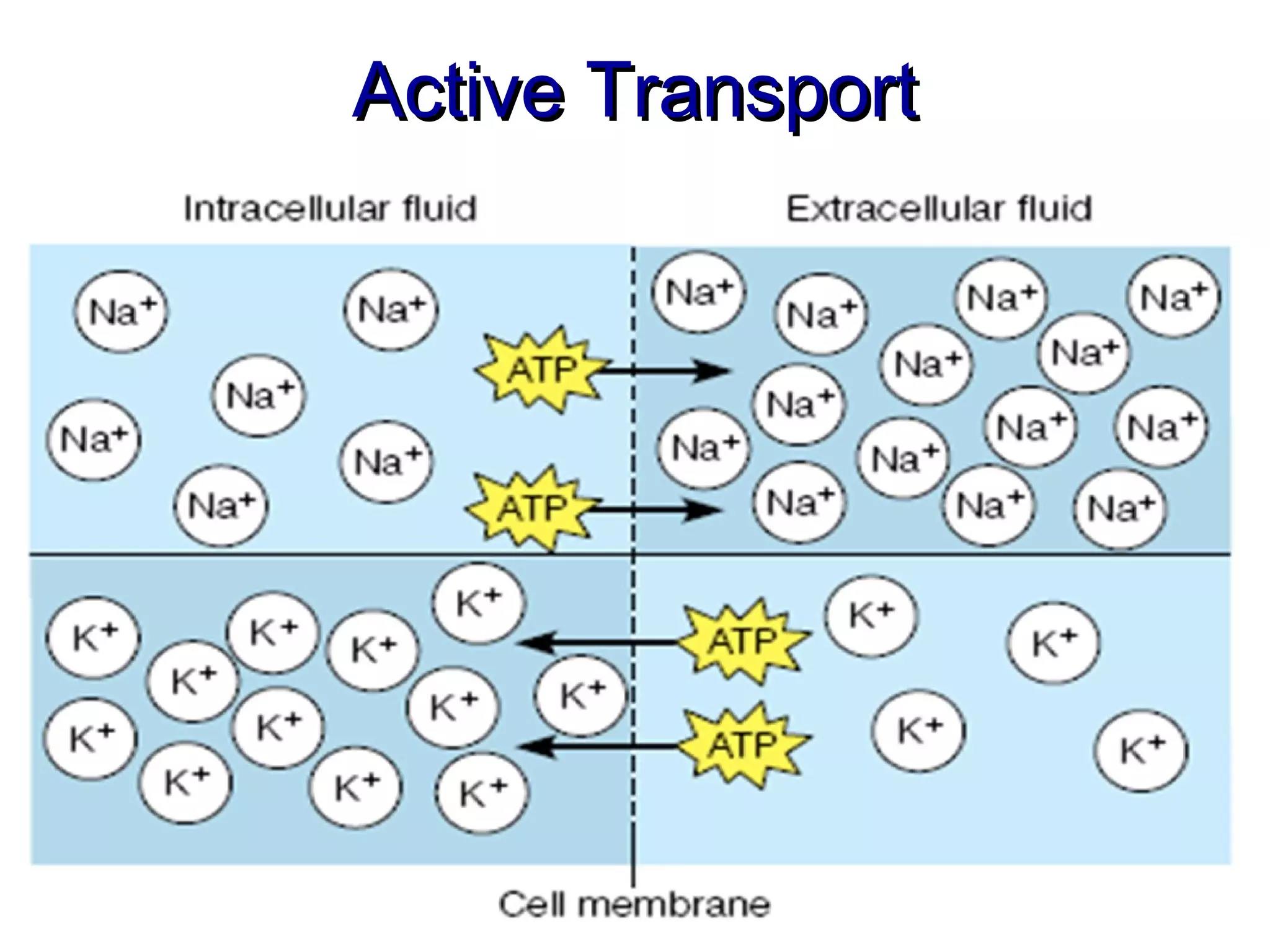
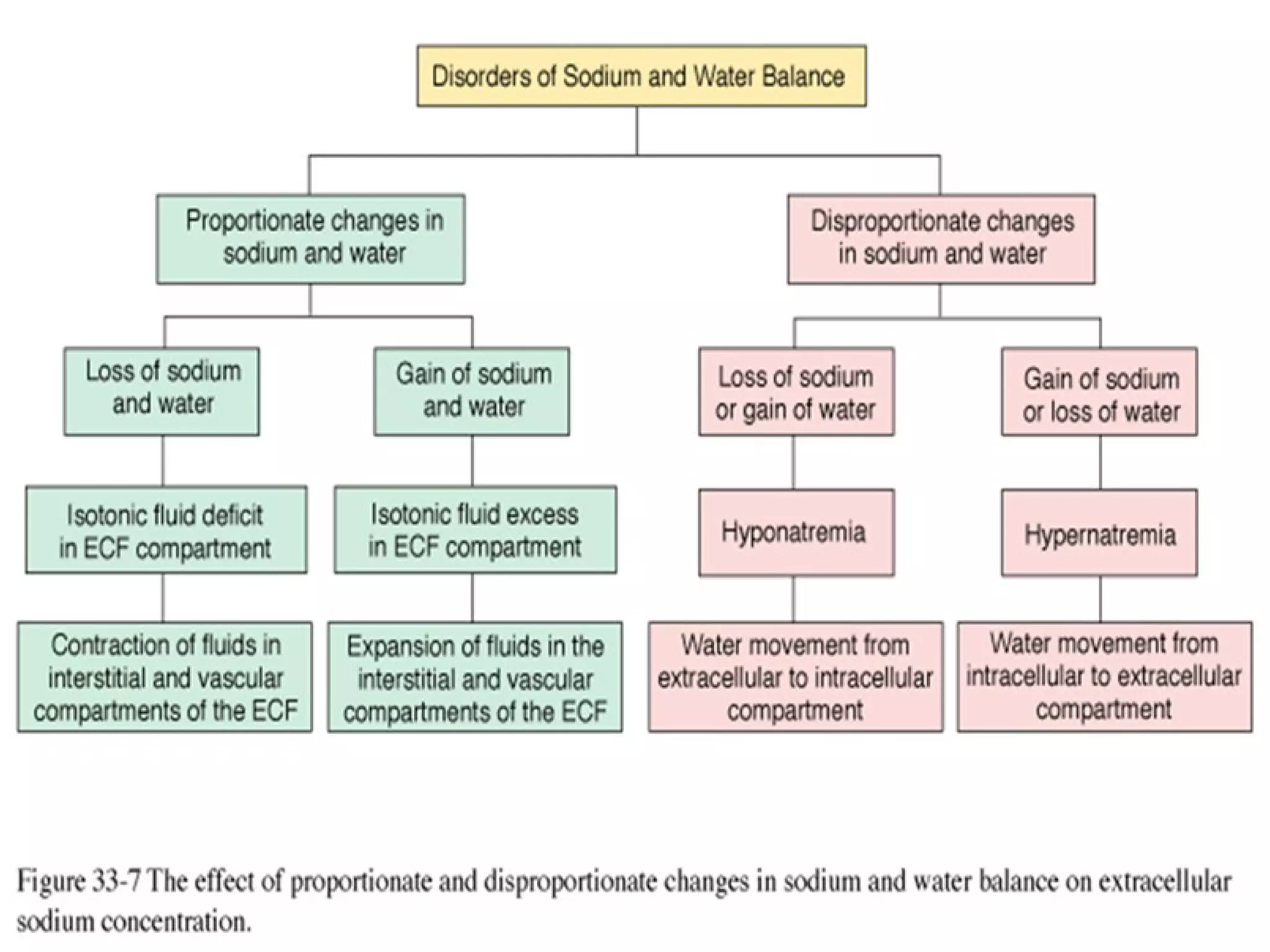
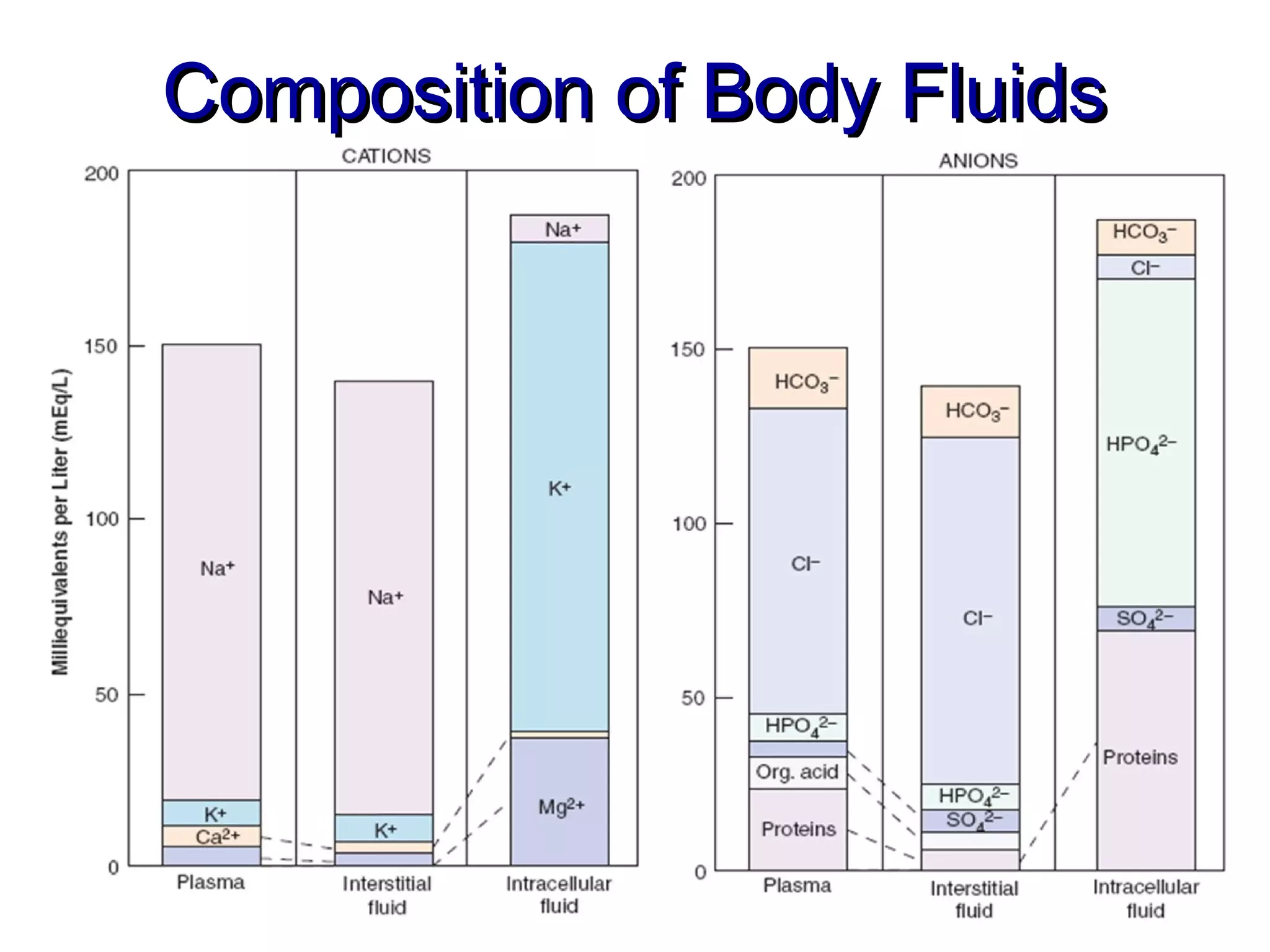
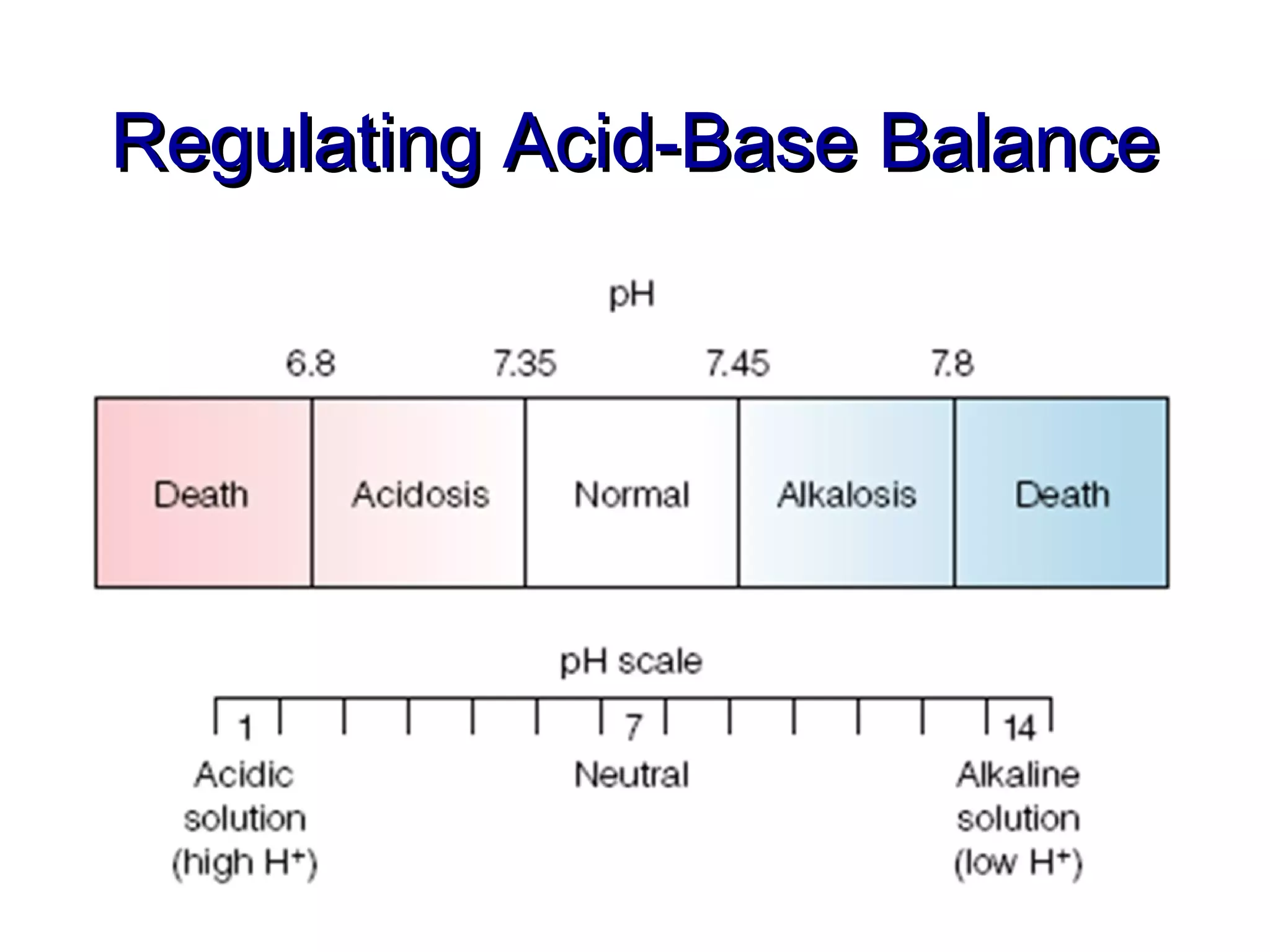
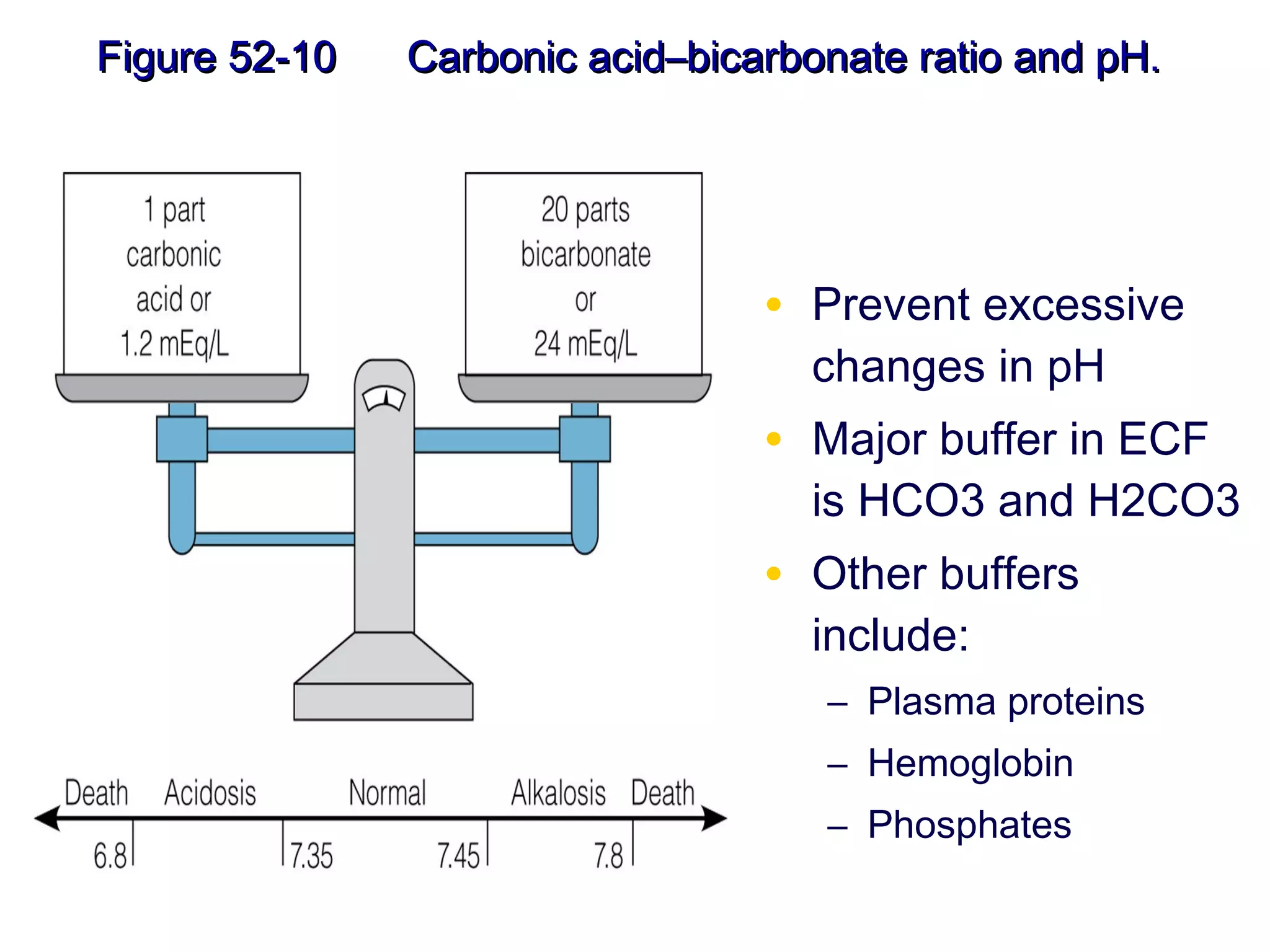
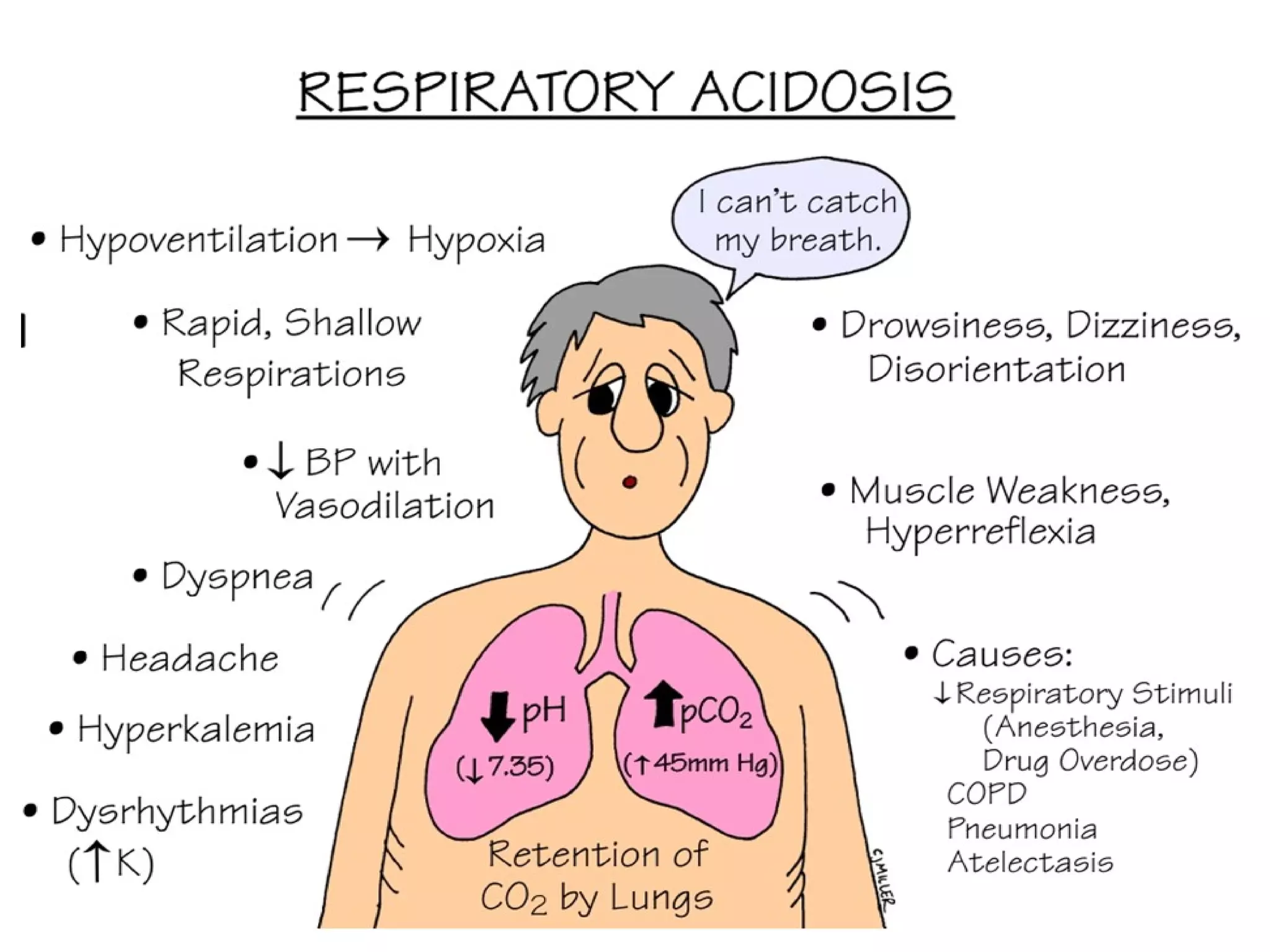
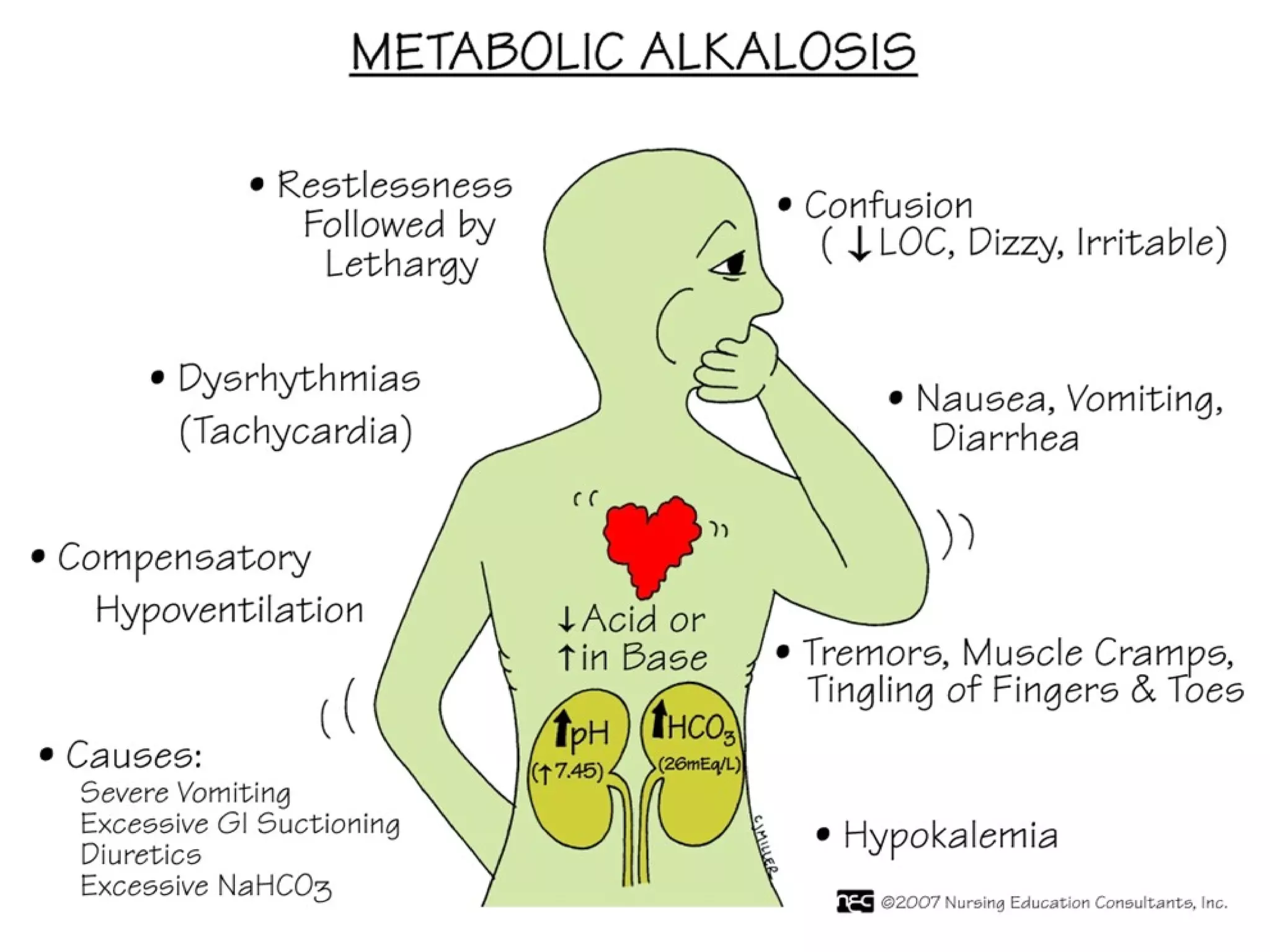
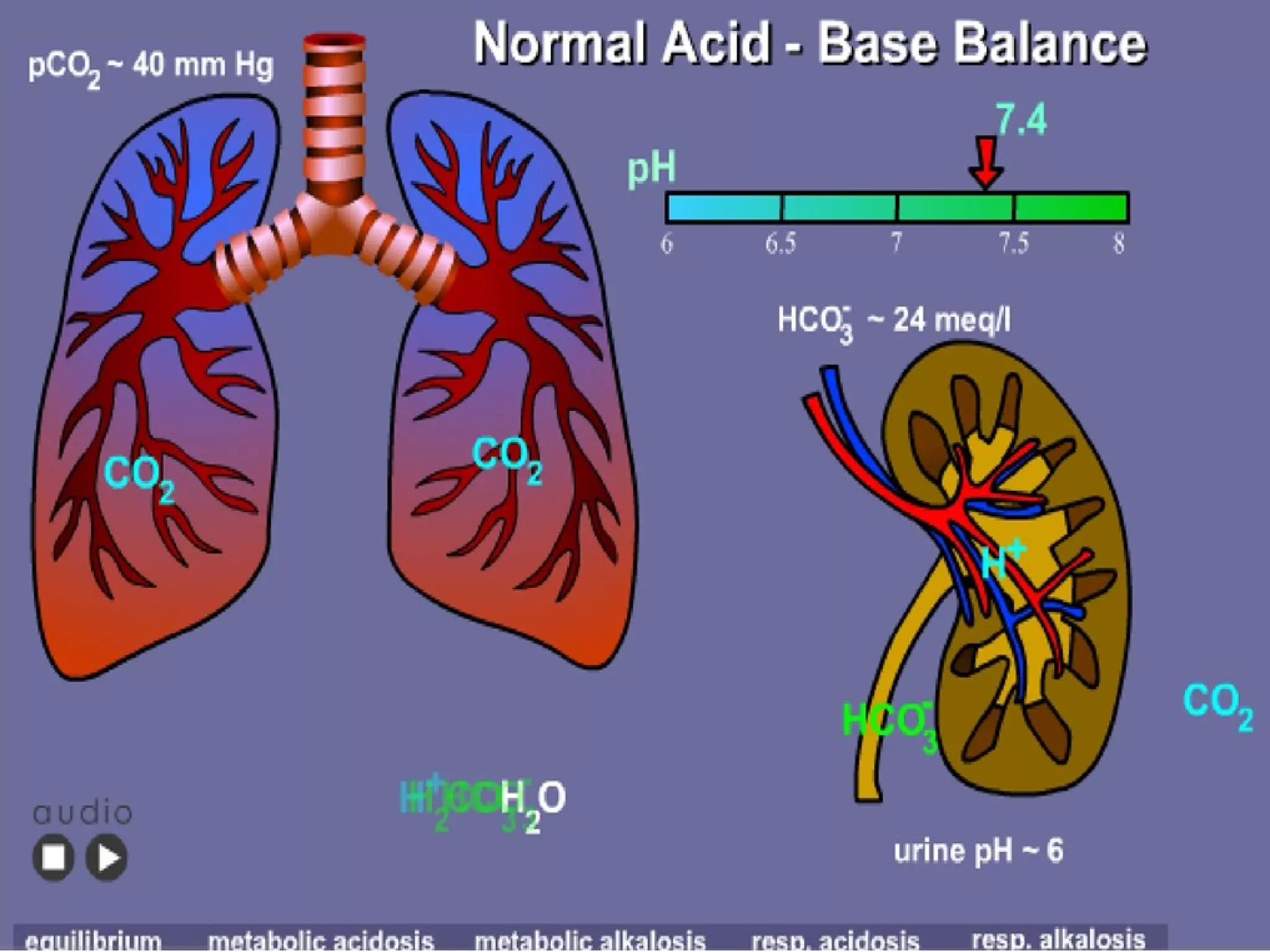
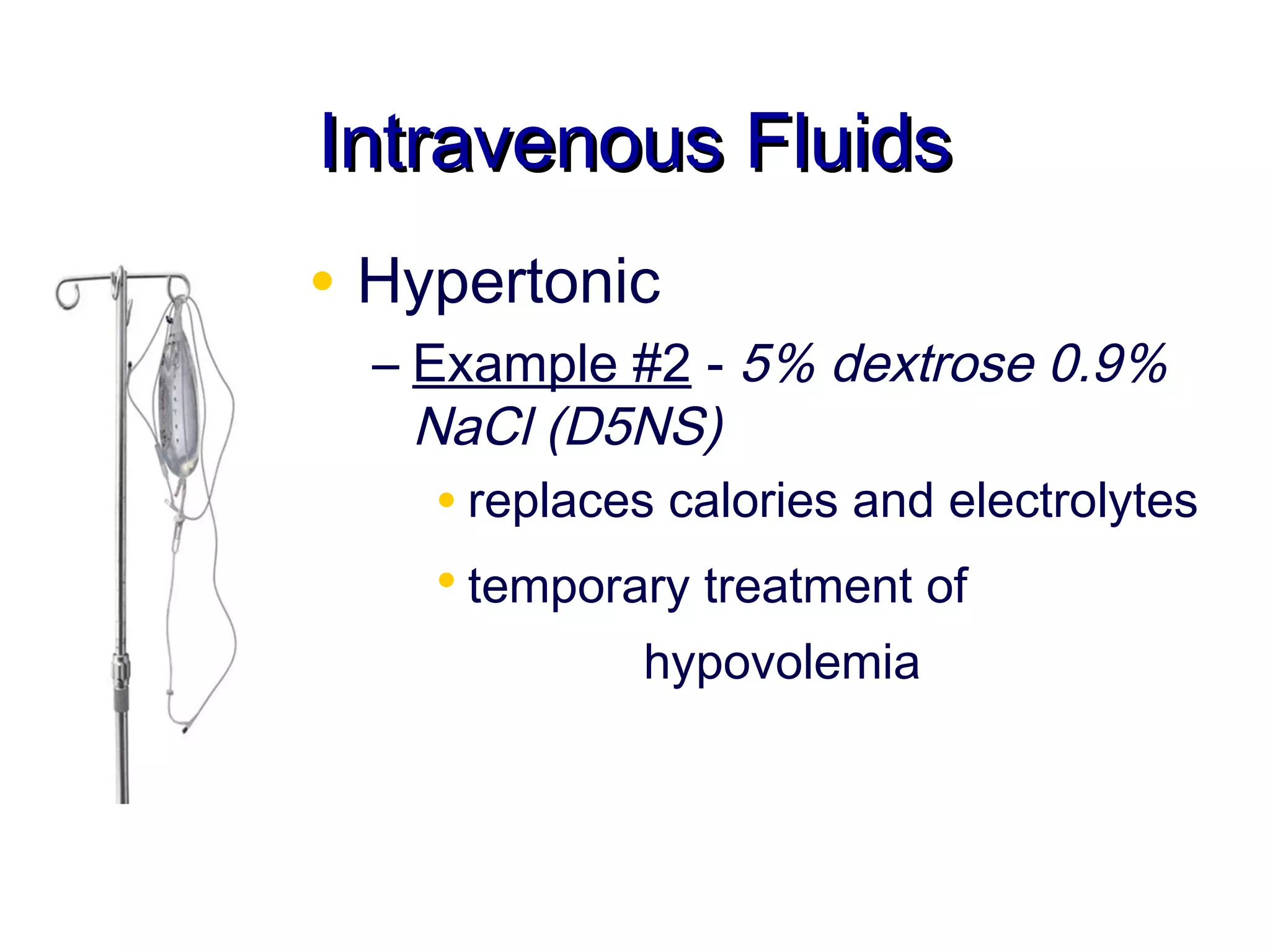
This document discusses fluid, electrolyte, and acid-base balance. It begins by listing the objectives of describing fluid and electrolyte regulation, imbalance, major electrolytes, populations at risk, and related diagnostic tests and nursing responsibilities. It then covers topics like body fluid distribution, maintaining fluid balance, fluid volume excess and deficit, electrolyte regulation and imbalances, and acid-base regulation and imbalances. It discusses related nursing interventions like monitoring intake and output, weight, lab tests, and treating imbalances through oral or IV fluid replacement depending on the situation.