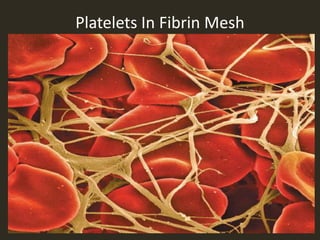
This document discusses hemostasis and the use of tranexamic acid (TXA) in treating hemorrhagic shock. It describes the three mechanisms of hemostasis - vascular spasm, platelet plug formation, and coagulation. It explains the extrinsic and intrinsic coagulation pathways and the factors involved. TXA inhibits fibrinolysis and reduces bleeding. Large clinical trials like CRASH-2 found TXA significantly reduces mortality in trauma patients when given within 3 hours of injury. The document proposes a prospective study to evaluate pre-hospital administration of TXA to reduce mortality, blood product usage, and blood loss in trauma patients with hemorrhagic shock.