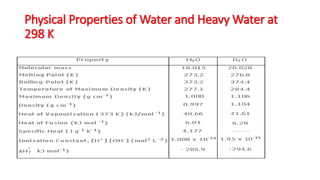
Water has a simple molecular structure composed of one oxygen atom covalently bonded to two hydrogen atoms. This gives water a polar molecular structure with partial positive charges near the hydrogen atoms and a partial negative charge near the oxygen atom. This polarity allows water to dissolve ionic compounds through electrostatic interactions between the ions and polar water molecules, but not non-polar covalent compounds which do not interact strongly with water. Heavy water, composed of deuterium instead of hydrogen, is used as a neutron moderator in nuclear reactions and to produce deuterium through electrolysis or reaction with metals.