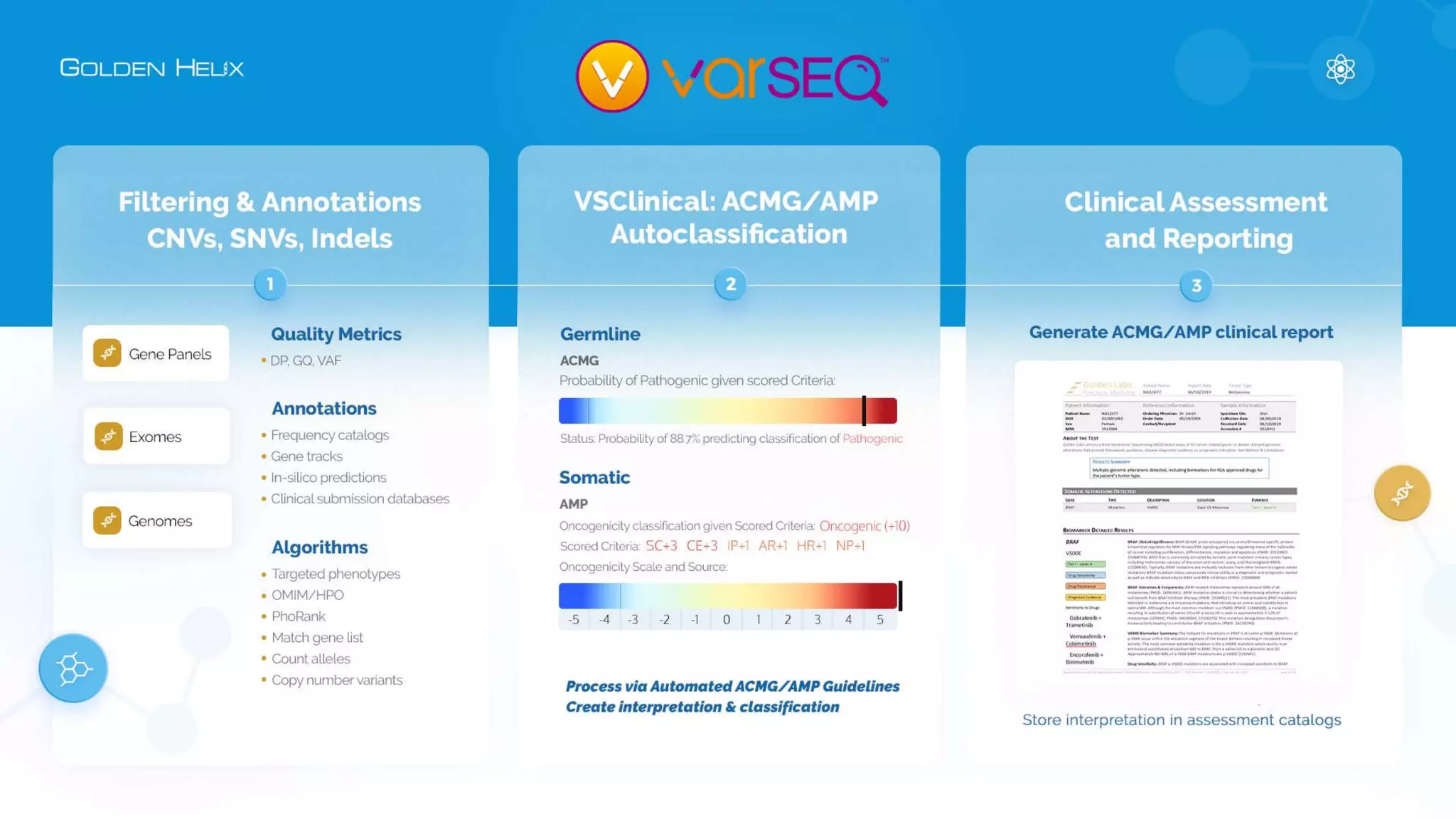
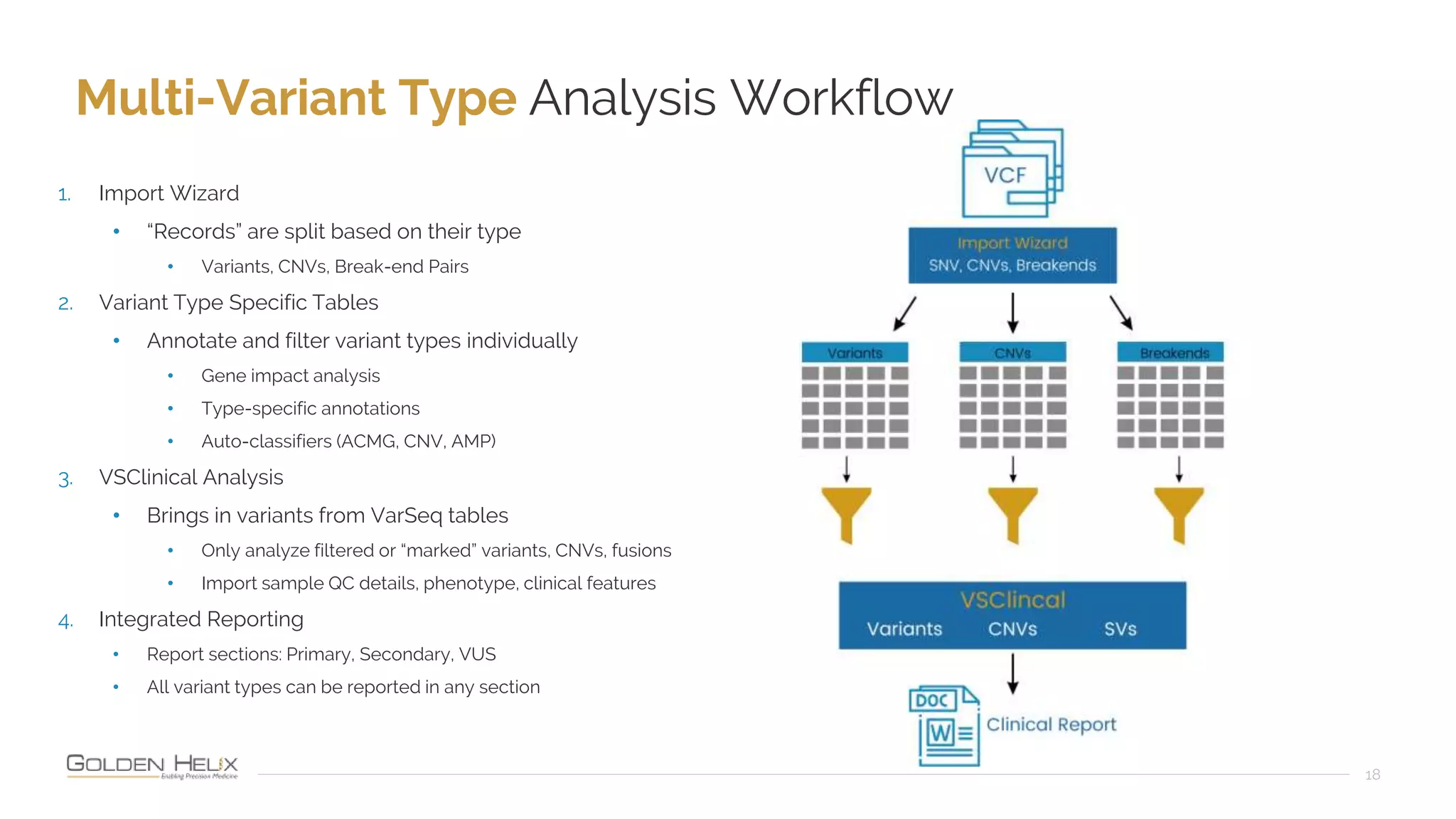
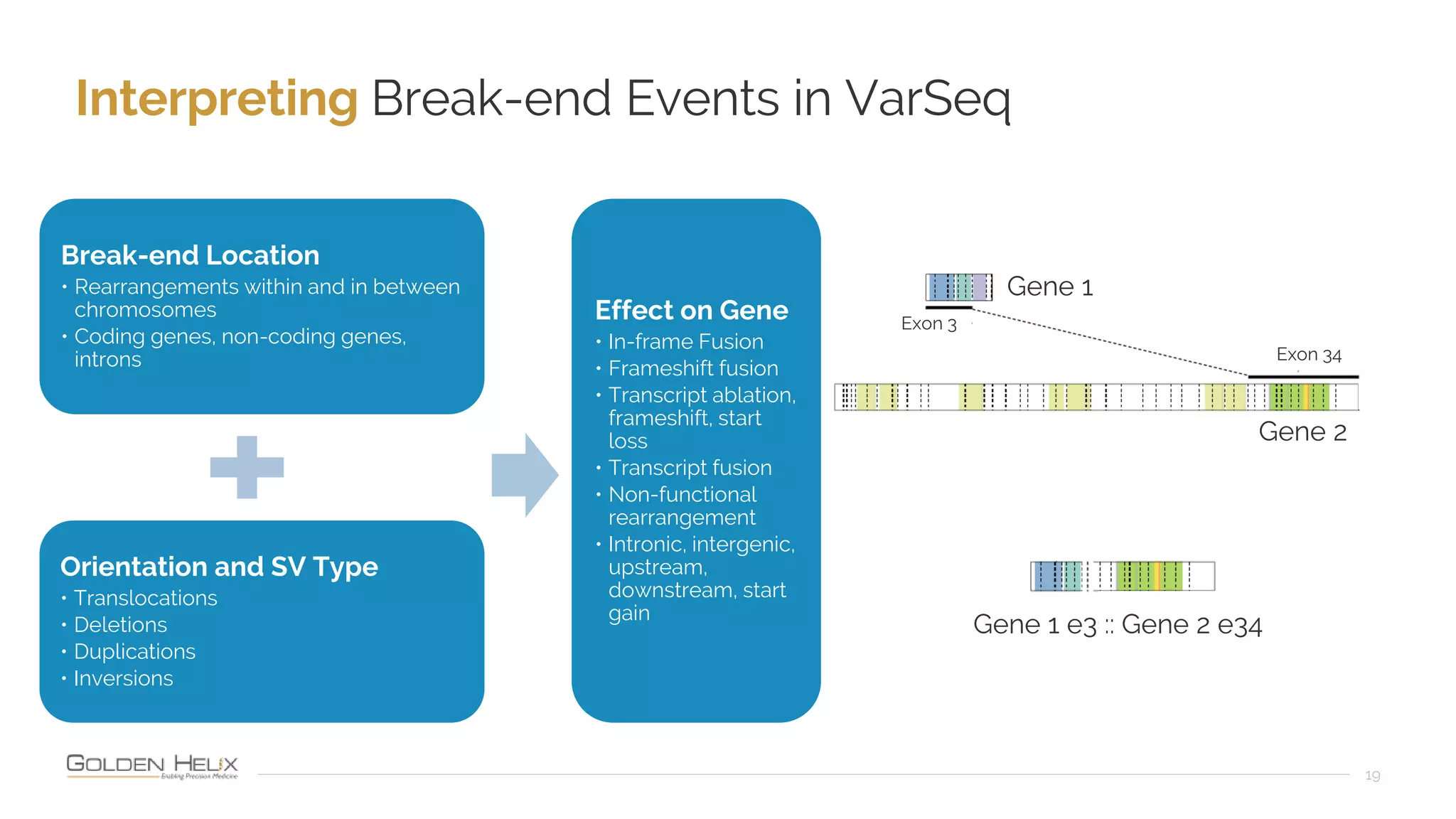
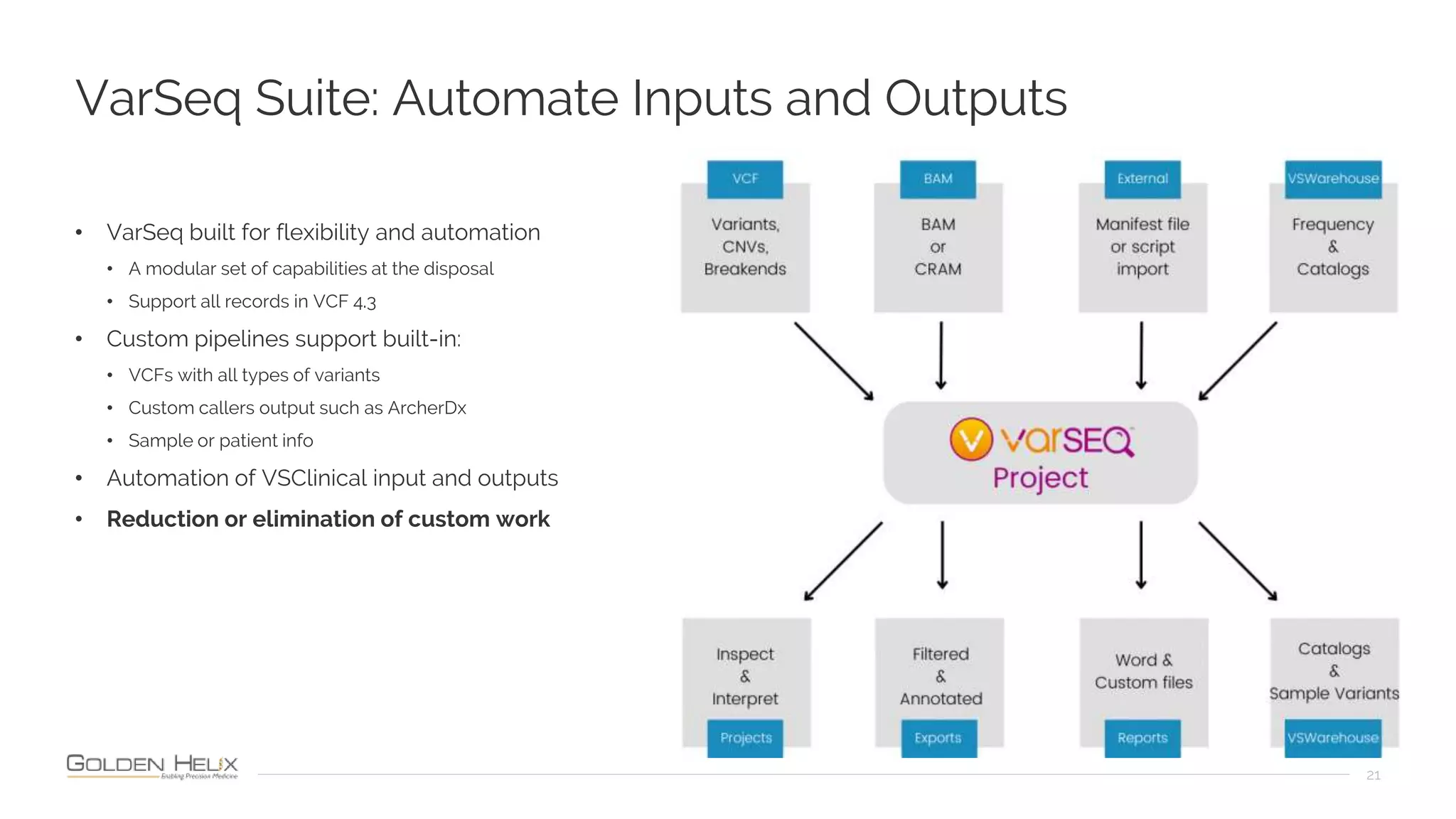
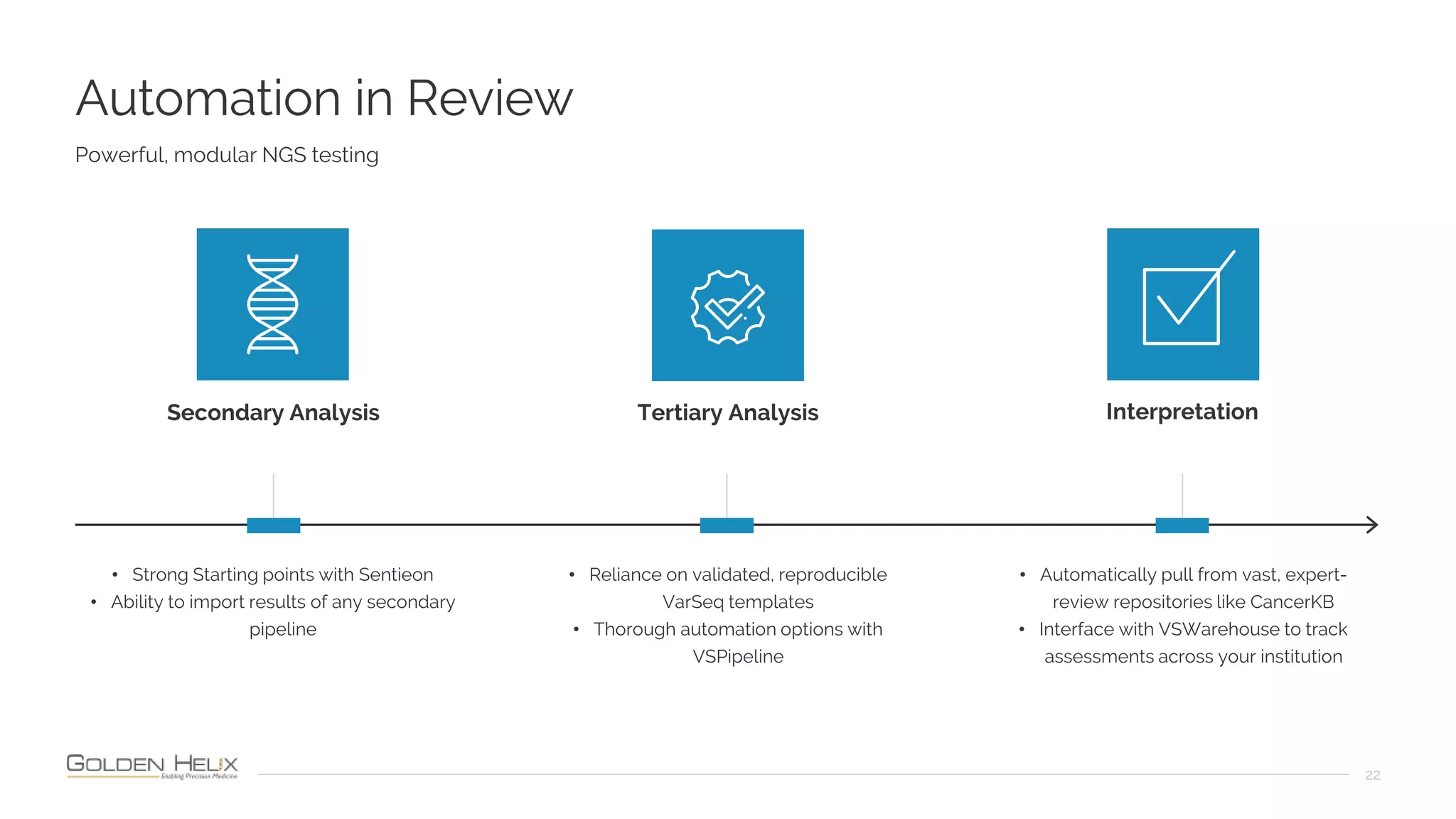
The document presents an overview of the VarSeq 2.4.0 software, focusing on its capabilities for structural variants and automation in genomic analysis, showcased in a presentation by Gabe Rudy. It highlights the support and research funding from NIH and the core features of the Golden Helix platform designed for flexible and automated clinical genomic reporting. Additionally, the document details the agenda, demonstrating the software's applications in cancer and germline testing, emphasizing the importance of automation and customization in genomic workflows.