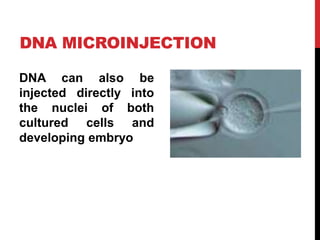
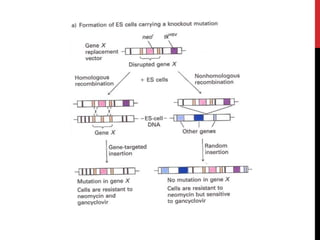
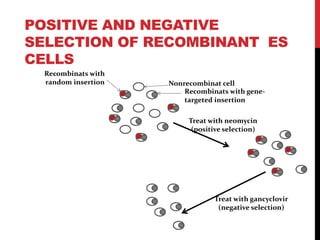
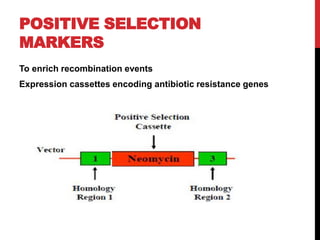
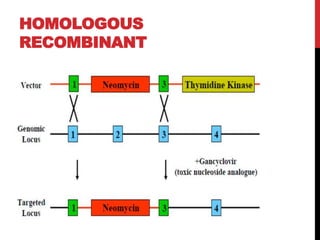
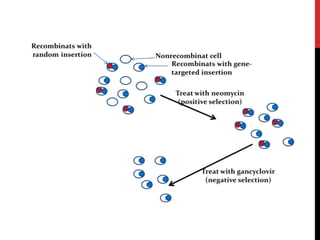
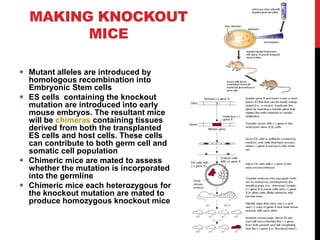
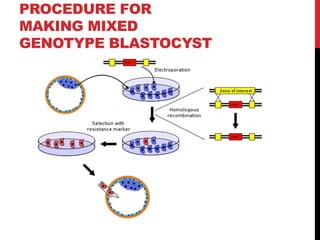
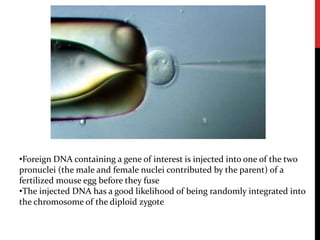
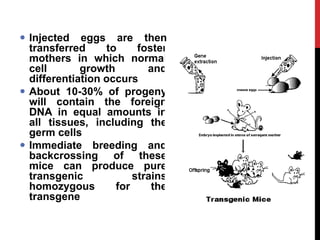
Knockout mice are genetically engineered mice where one or more genes have been inactivated through gene knockout. They are important animal models for studying the role of genes with unknown functions. By causing a specific gene to be inactive in mice and observing differences in behavior or health, researchers can infer the probable function of that gene. Transgenic mice have foreign or modified genes added, which are then integrated randomly into the mouse genome, allowing the study of these additional genes. Both knockout and transgenic mice are useful models for studying human genetic diseases.