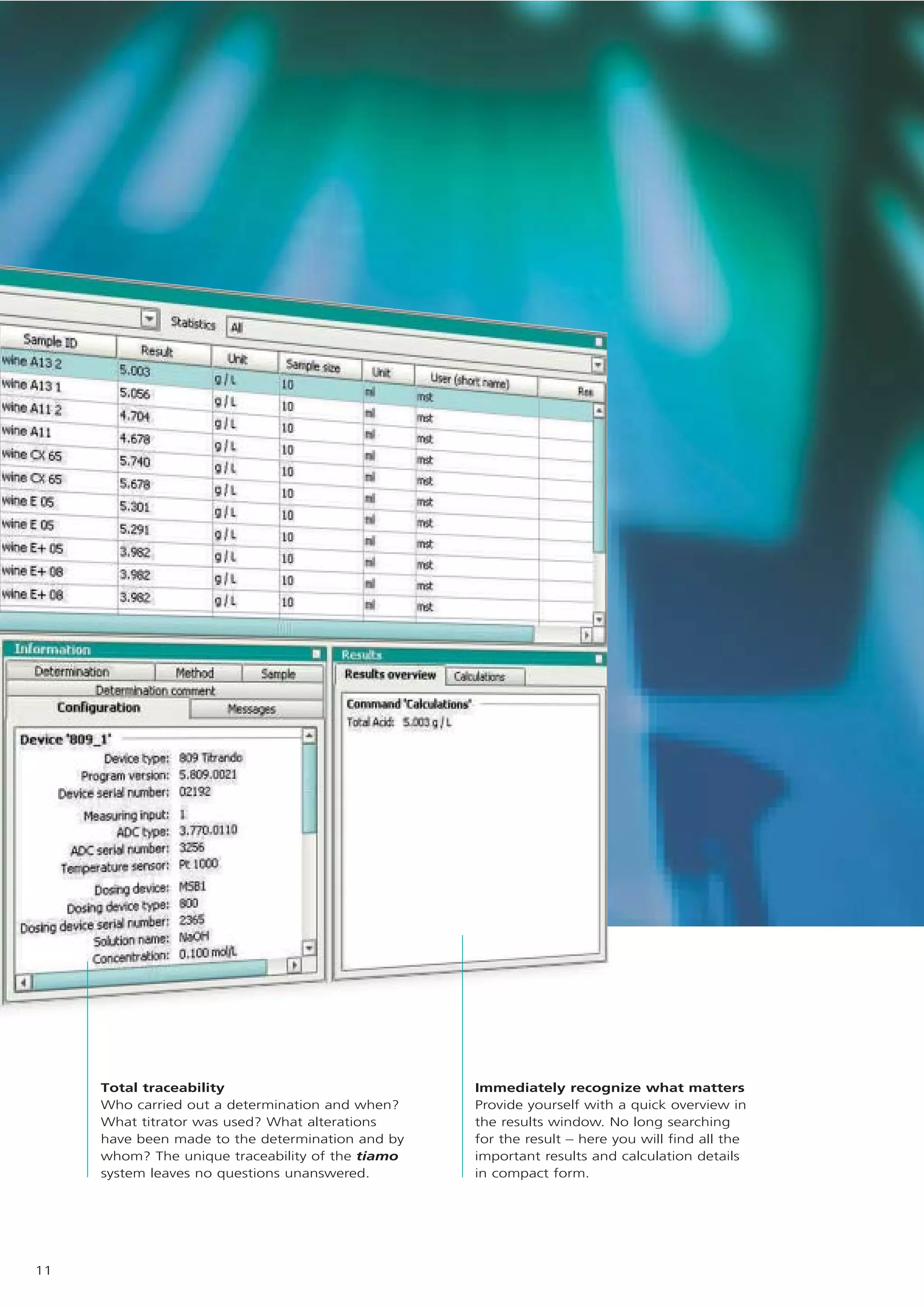
1) tiamo is Metrohm's new control and database software for titration systems and laboratory automation. It allows for titration, dosing, and complete automation of analytical workflows.
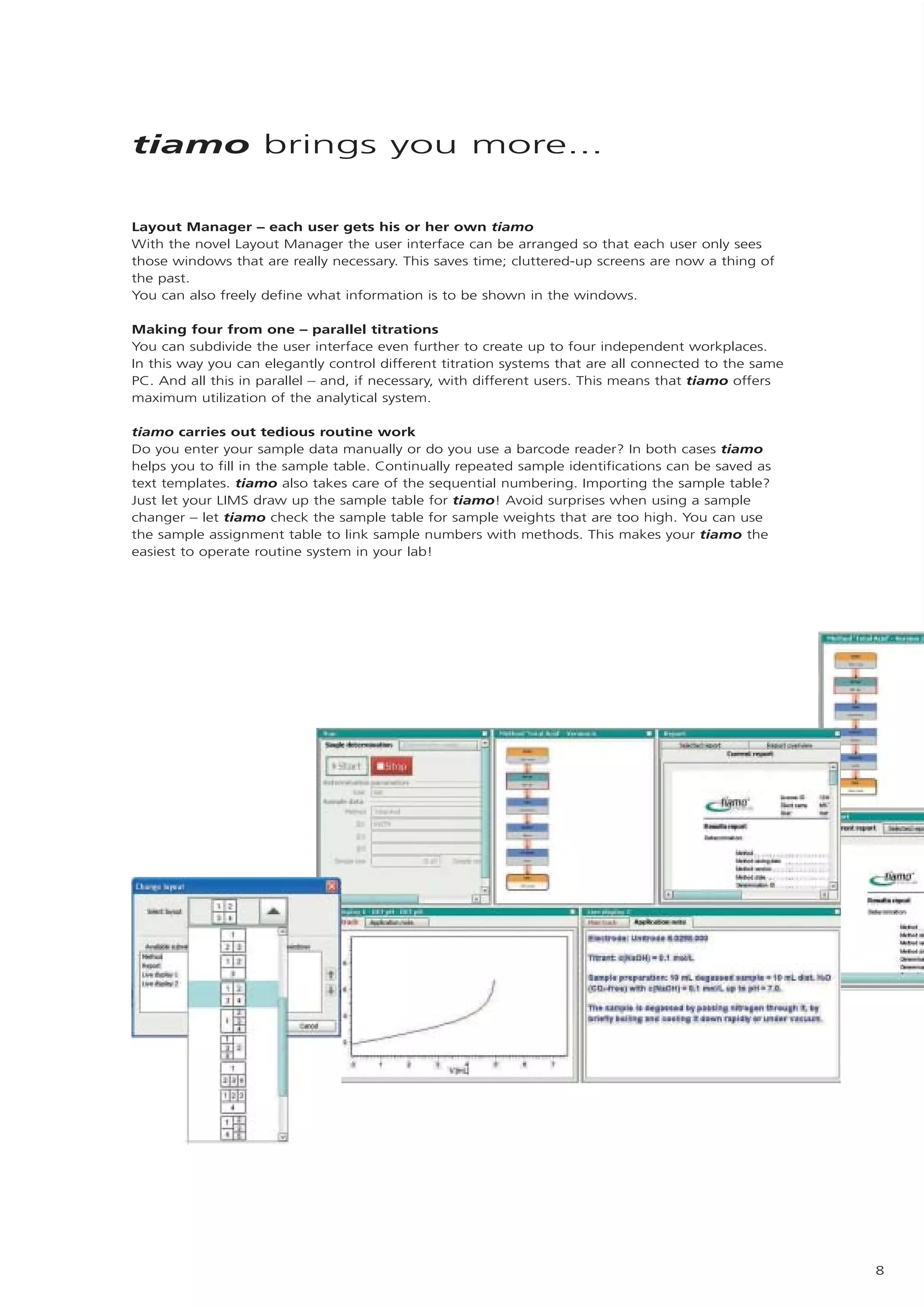
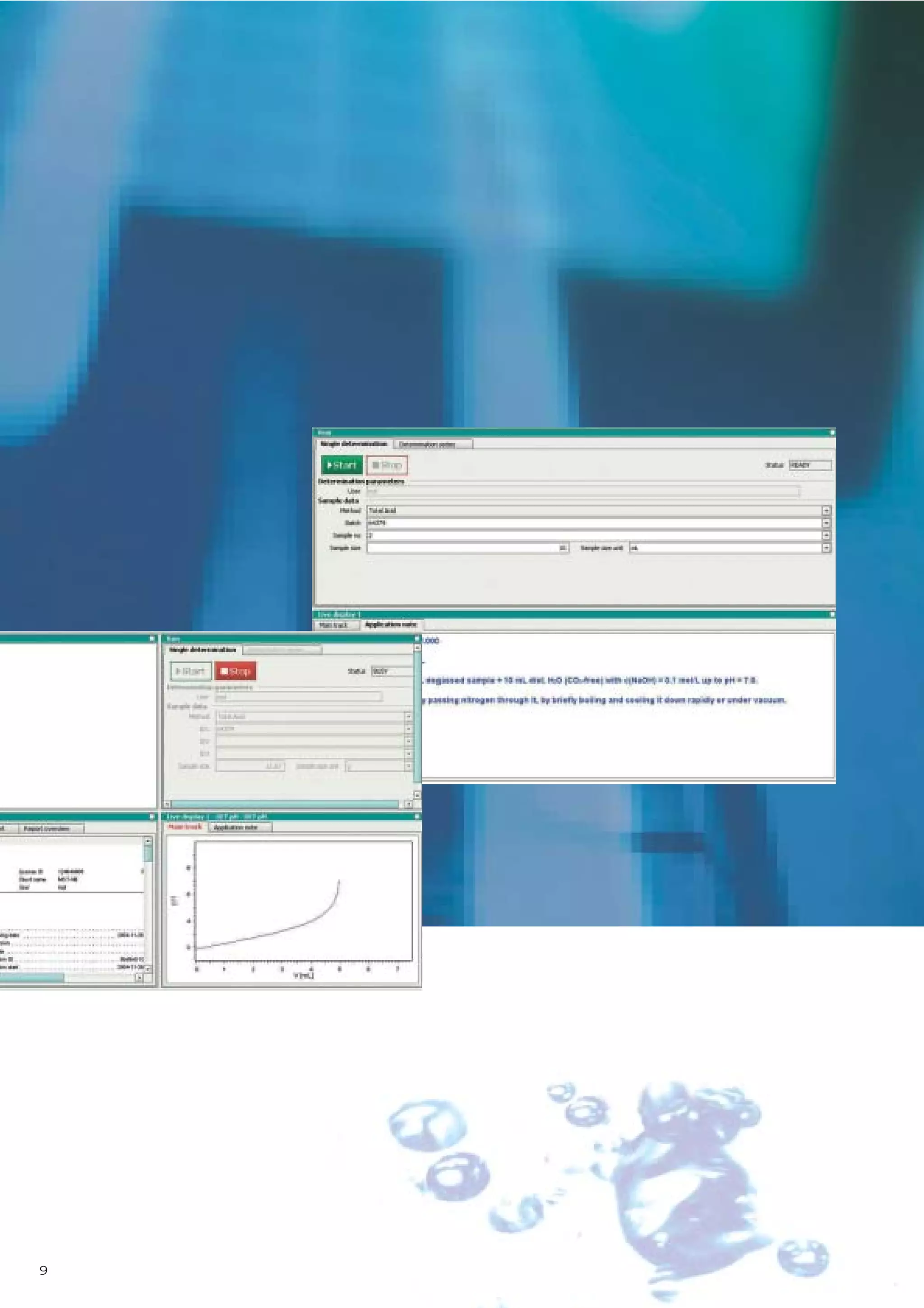
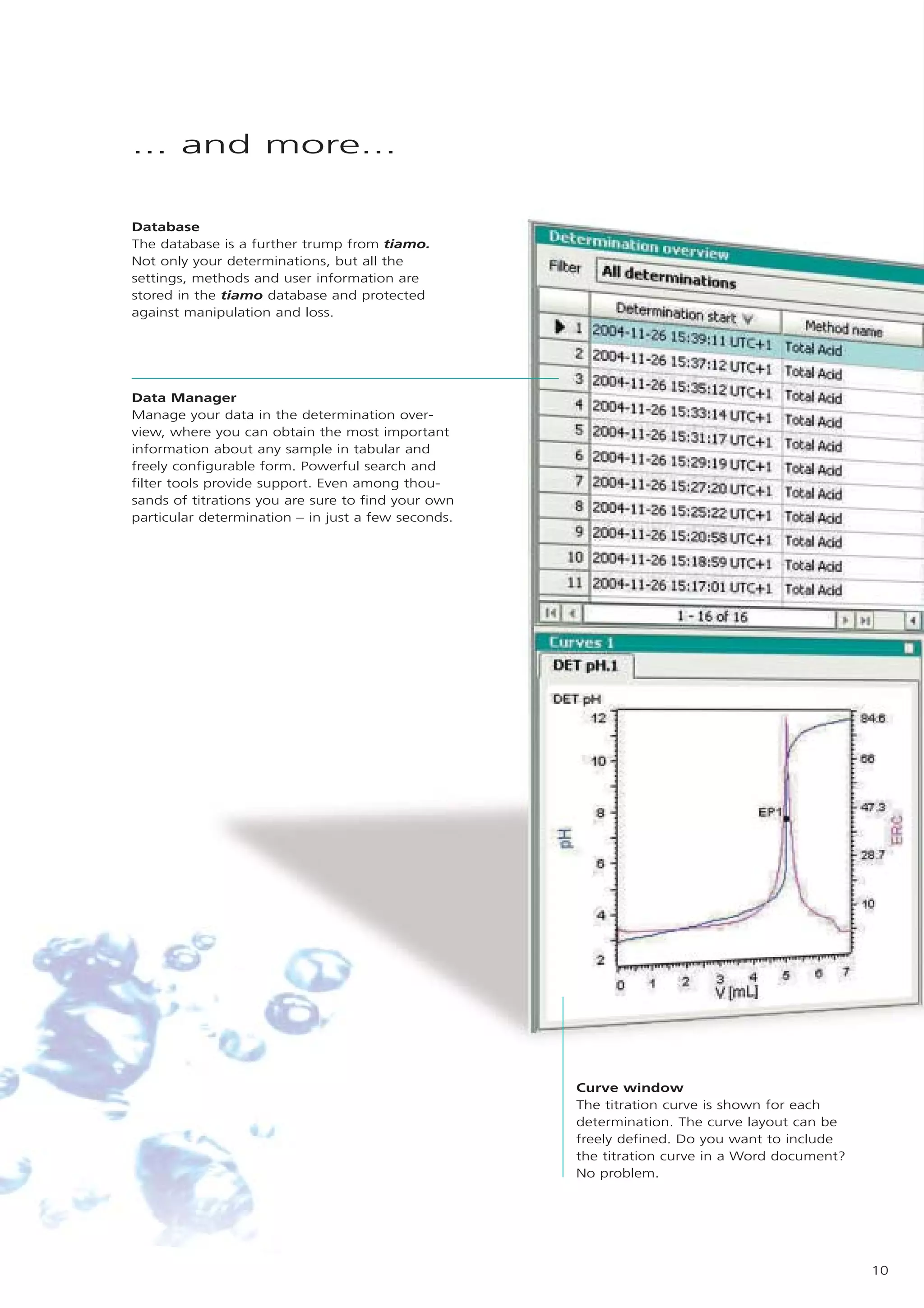
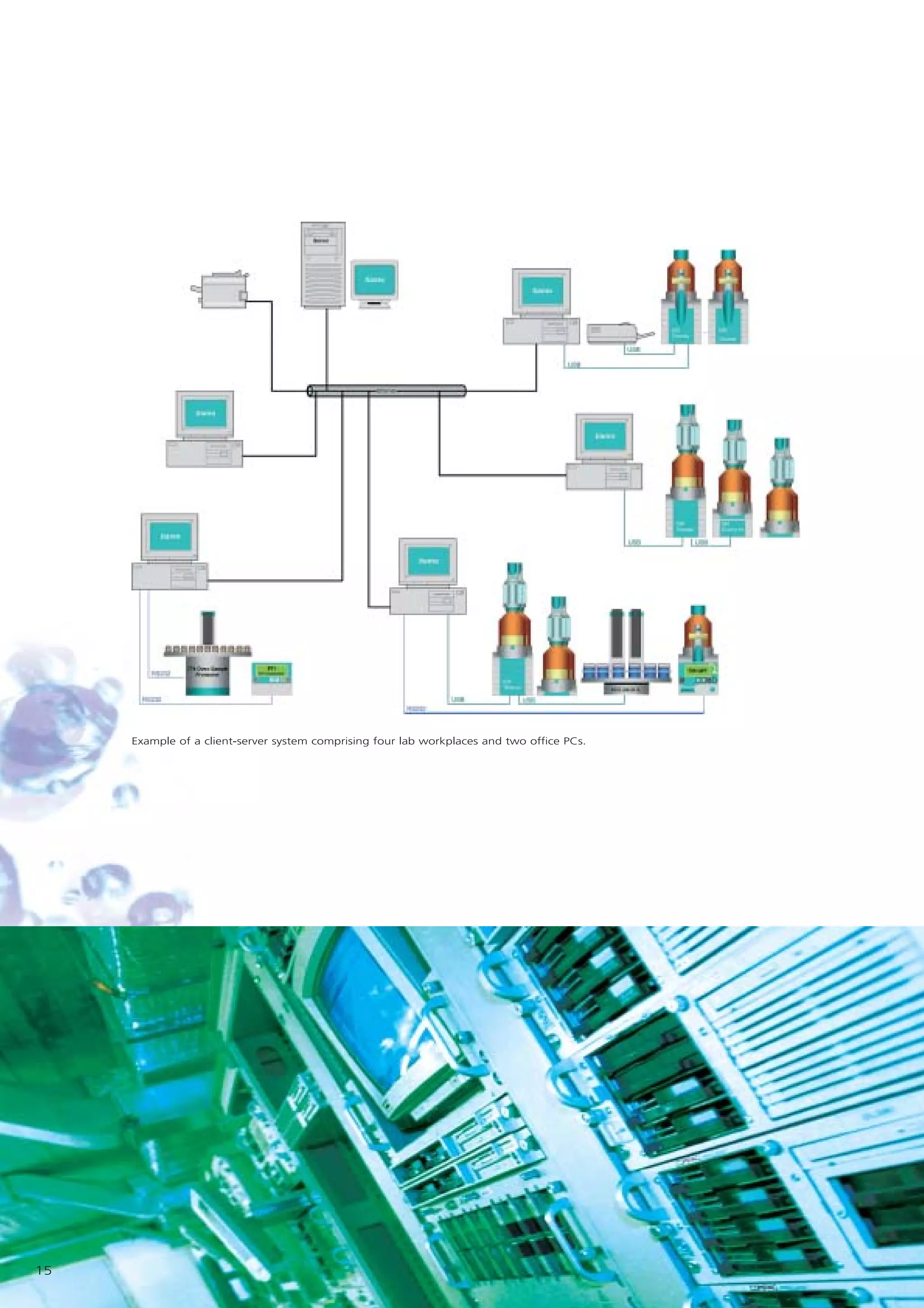
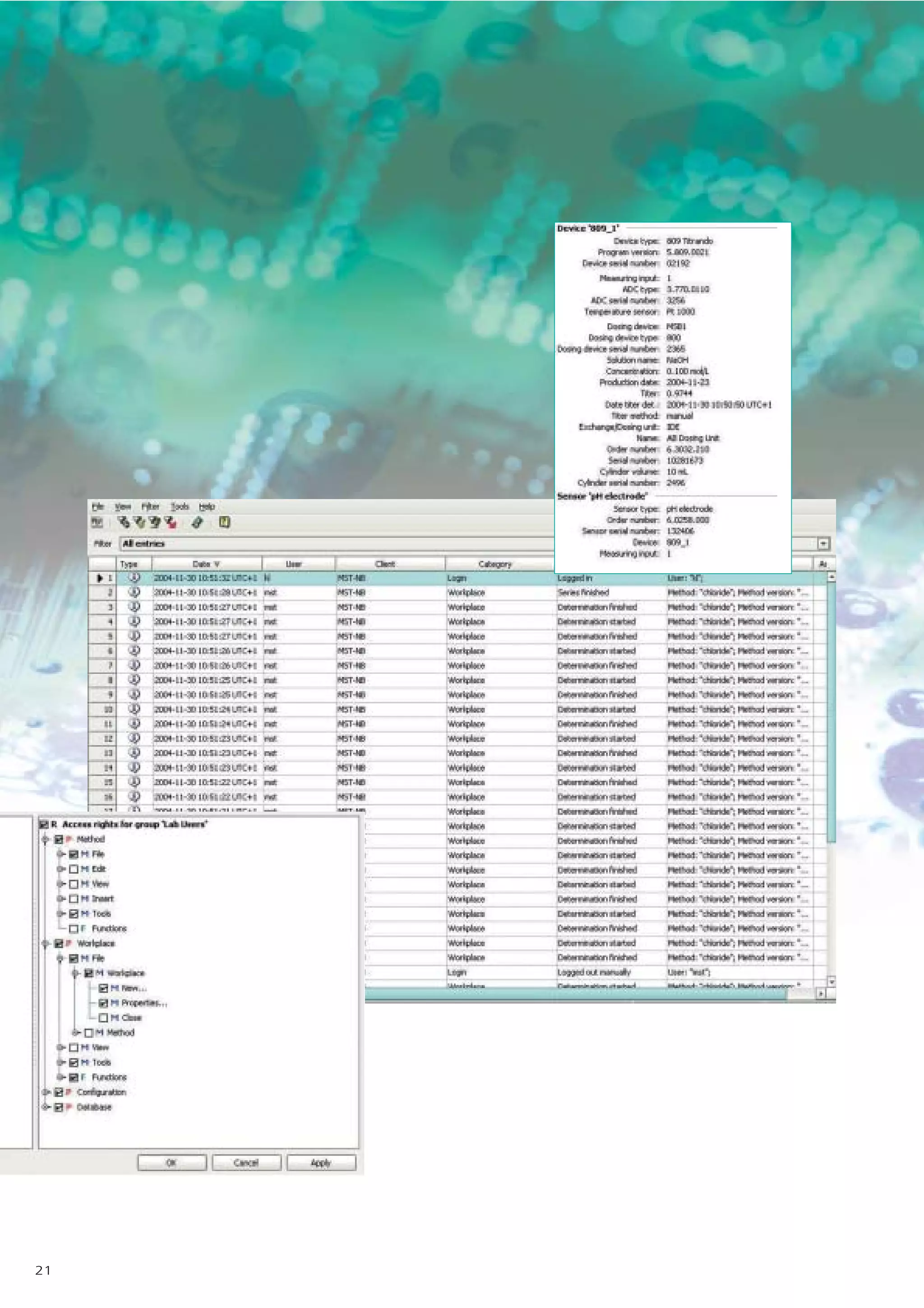
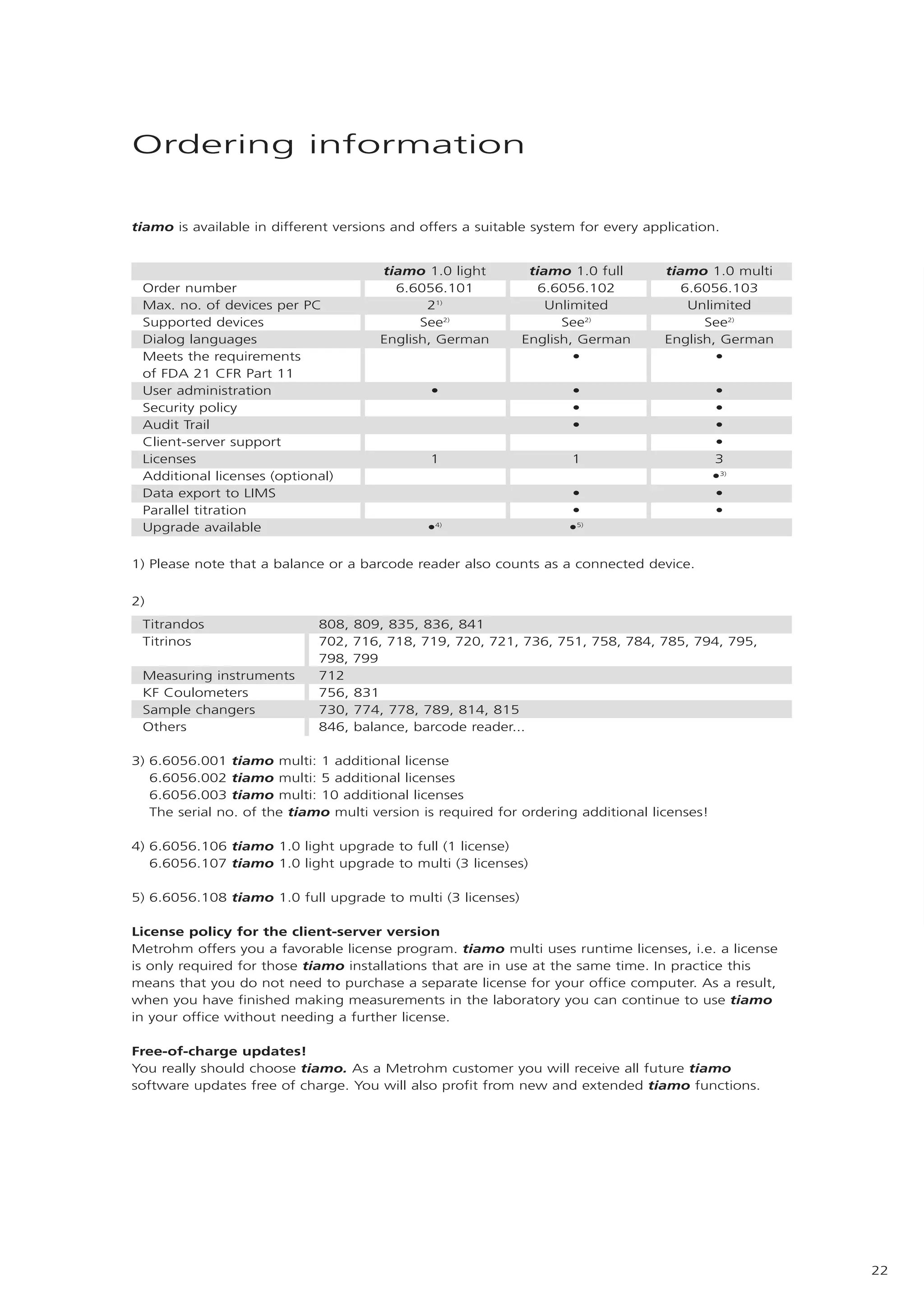
2) tiamo offers a modern user interface, powerful report generation, flexible method editor, real-time parallel titrations, client-server database, and export of data to LIMS systems.
3) As tiamo can integrate existing Titrino and new Titrando systems, it protects customers' investments while offering compatibility across different Metrohm instruments.