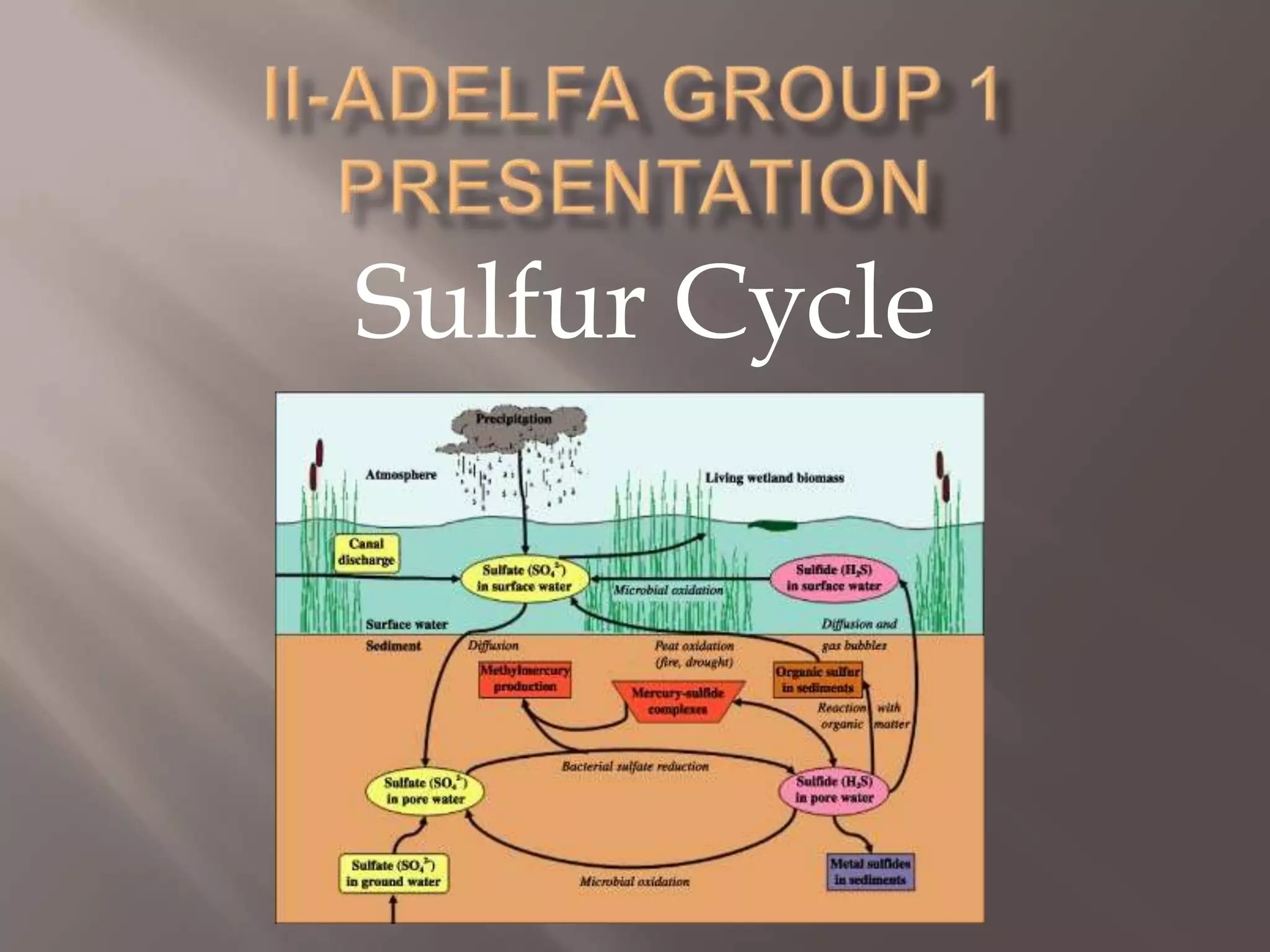
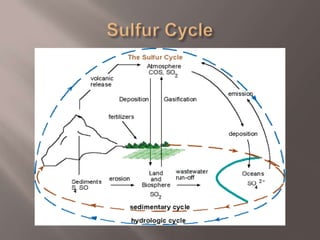
Sulfur is an abundant yellow solid element that cycles through the environment in rocks, soil, oceans, atmosphere and living things. The sulfur cycle involves weathering of rocks, uptake by plants and organisms, release through decomposition, emission from natural and human sources, and settling back into the earth or oceans. The sulfur cycle regulates sulfur levels and prevents acid rain, but human emissions have increased atmospheric sulfur and contributed to issues like acid rain and smog.