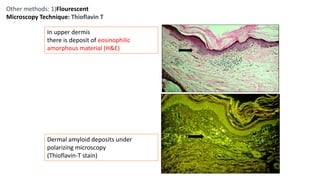
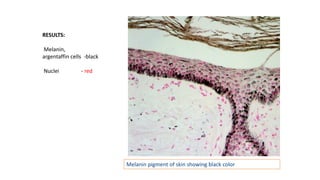
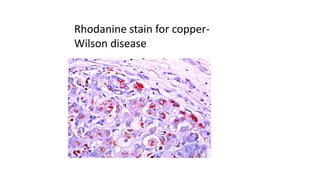
This document provides information and methods for special stains used to identify various tissues and microorganisms under the microscope. It discusses stains for amyloid, pigments, microbes and calcium. Specific stains covered include Congo red for amyloid, Perls' Prussian blue for iron, Fouchet's for bile pigments, Masson Fontana for melanin, von Kossa for calcium, rhodanine for copper, Gram stain for bacteria identification, Ziehl-Neelsen for acid-fast mycobacteria, and Warthin-Starry for spirochetes. Detailed protocols are provided for each staining method.