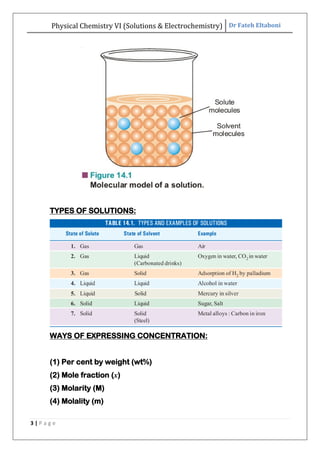
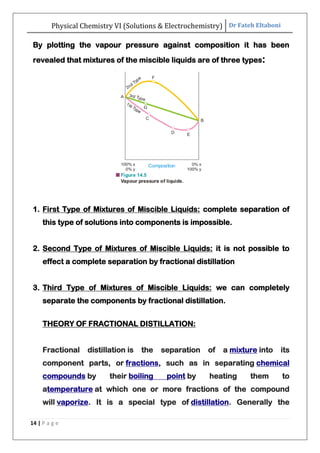
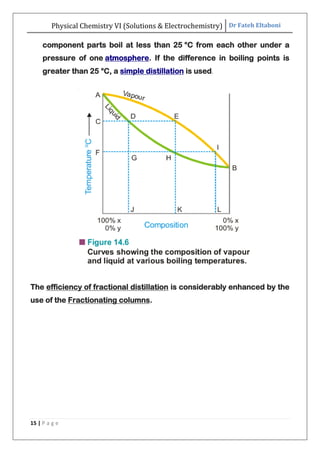
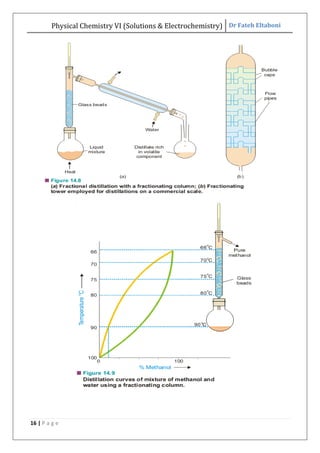
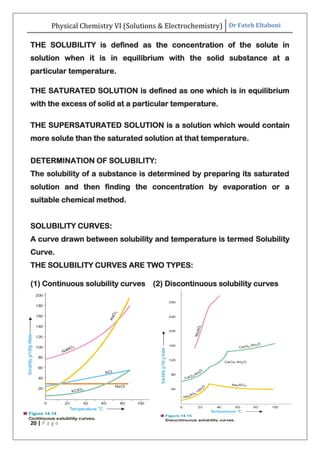
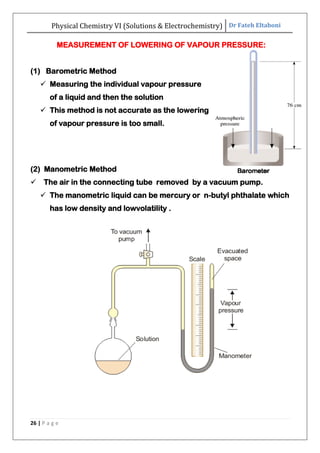
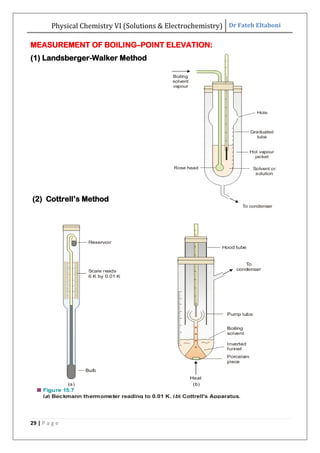
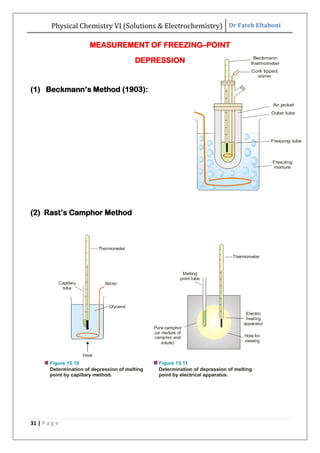
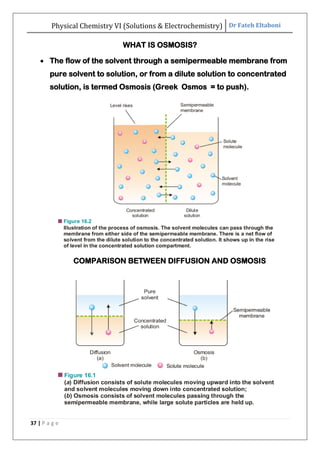
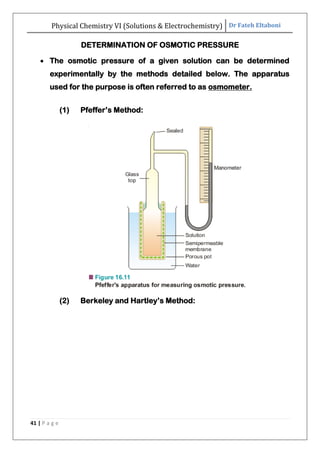
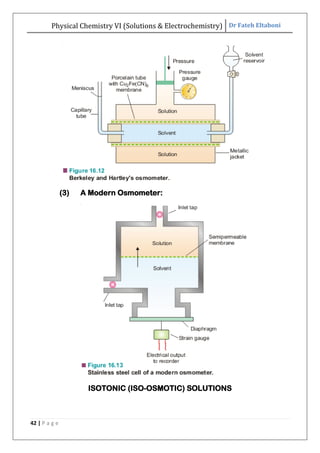
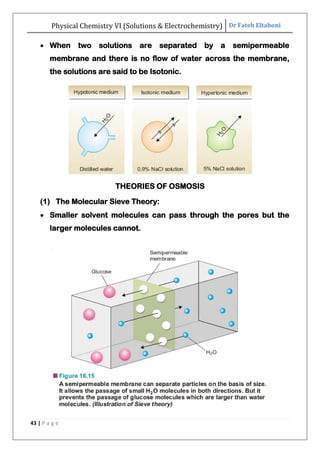
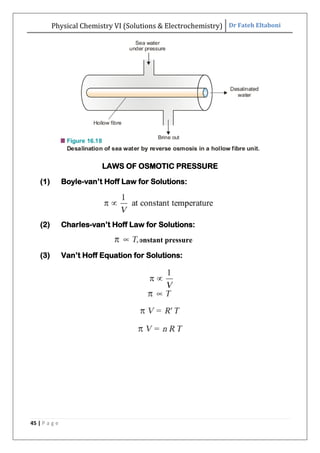
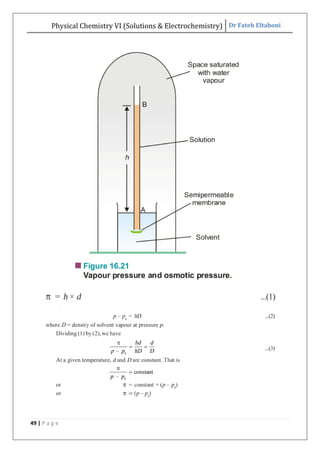
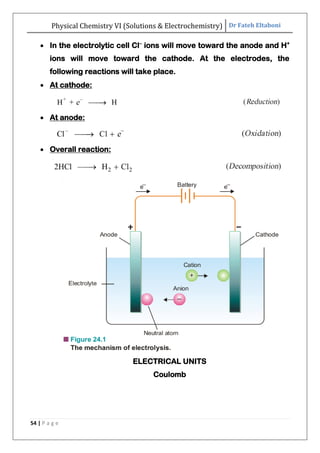
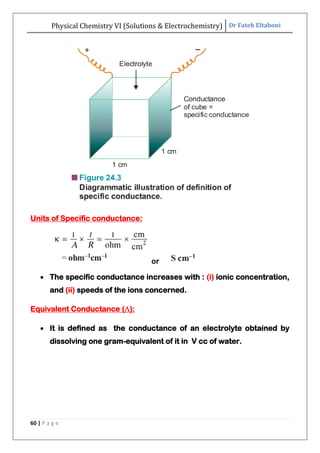
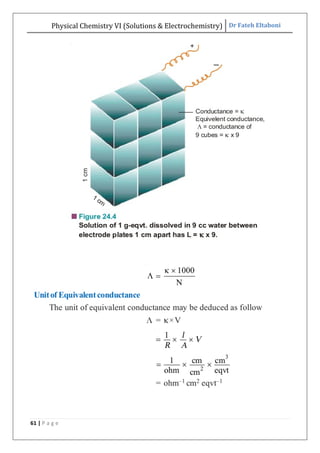
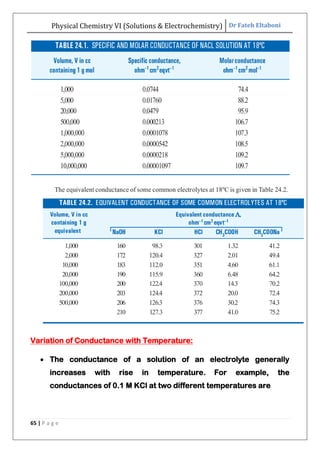
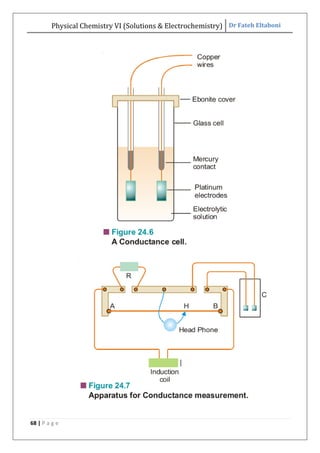
The document discusses various topics related to solutions and electrochemistry. It begins by defining solutions and different ways of expressing concentration such as molarity, molality and normality. It then discusses solutions of gases in gases based on Henry's law and solutions of liquids in liquids, including completely miscible and partially miscible liquids. Other topics covered include solubility, vapor pressure of solutions, distillation, and colligative properties of solutions such as boiling point elevation, freezing point depression, and vapor pressure lowering. Measurement techniques for various colligative properties are also summarized.