The document provides an in-depth analysis of electrochemistry, focusing on Debye-Hückel theory and Onsager equation to explain the conductance of strong electrolytes and their behavior under dilution. It discusses various effects impacting ionic mobility, including relaxation and electrophoretic effects, while detailing how conductivity varies with concentration for both strong and weak electrolytes. Additionally, it includes mathematical derivations and applications of conductivity and dissociation calculations for different types of electrolytes.








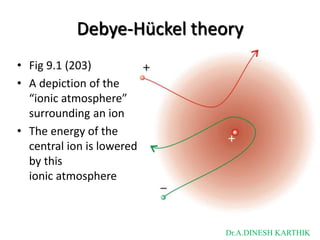
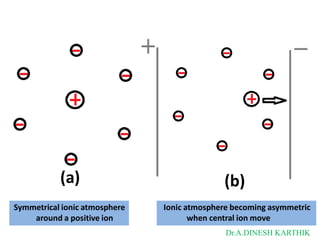
![Debye-Hückel theory
• Fig 9.2 (203)
• (a) the limiting law for
a 1,1-electrolyte
(B & C = 1)
• (b) the extended law
for B = 0.5
• (c) the extended law
extended further by
the addition of the
C I term
[in the graph, C=0.2]
Dr.A.DINESH KARTHIK](https://image.slidesharecdn.com/bscvsembelectrochemistry-221120153650-89641ab8/85/BSc-V-SEM-B-Electrochemistry-1-pptx-9-320.jpg)
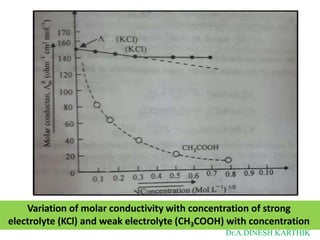




![λc = λ o – [
𝟖𝟐.𝟒
+
𝟖.𝟐𝟎+𝟏𝟎𝟓
ƞ(𝑫𝑻)𝟏/𝟐 (𝑫𝑻)𝟑/𝟐 𝐨
λ ] 𝑪
Where
λ c= equivalent conductance at concentration C
λ o = equivalent conductance at infinity dilution
D = Dielectric constant of the medium
ƞ= Viscosity of the medium
T = Temperature of the solution in degrees absolute
C = concentration of the solution in moles/litre
As D and ƞ are constant for a particular solvent,
therefore, at constant temperature, the above
equation can be written in the form
Dr.A.DINESH KARTHIK](https://image.slidesharecdn.com/bscvsembelectrochemistry-221120153650-89641ab8/85/BSc-V-SEM-B-Electrochemistry-1-pptx-18-320.jpg)





![3-Application of Ostwald dilution Law
Calculation of dissociation constant of weak electrolytes
AB ↔ +
A+ B –
Initial concentration C 0 0
Concentration at equilibrium C (1-α) Cα Cα
Kd=
[𝑨]+[𝑩]− 𝑪𝑎 . 𝑪𝑎
𝑨𝑩
= 𝑪(𝟏−𝑎)
𝑪𝑎𝟐
Kd = 𝟏−𝑎
If the degree of dissociation α is very small, as in the case of weak electrolytes at ordinary
concentration, α can be neglected in comparison to 1 in the denominator of expression.
d
K = Cα2 or α2 = 𝐊𝒅
𝑪
α =
𝑲𝒅
𝐂
α = degree of dissociation, C = Concentration (mole/L),
Kd = dissociation constant
Dr.A.DINESH KARTHIK](https://image.slidesharecdn.com/bscvsembelectrochemistry-221120153650-89641ab8/85/BSc-V-SEM-B-Electrochemistry-1-pptx-24-320.jpg)



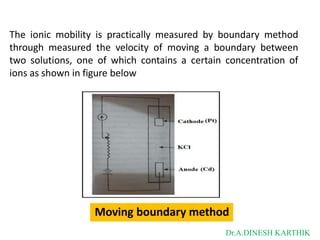
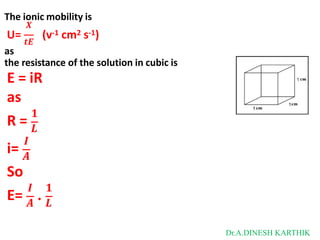
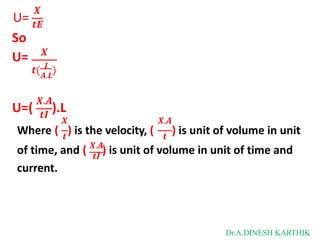

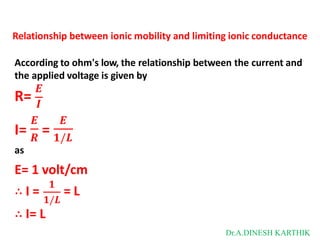

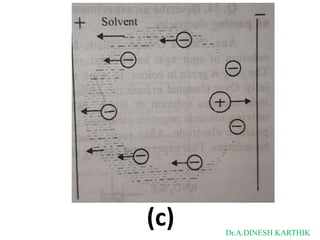
![Then
The total current is
I = L = F [C+ U+ + C- U- ]
L = CF [U+ + U-]
as
𝑳 + -
λ = 𝑪
= CF [U + U ]
as
λ = λ+ + λ-
λ+ = FU+
λ- = FU-
where
λ+ and λ- are limiting ionic conductance for cation and anion respectively.
Dr.A.DINESH KARTHIK](https://image.slidesharecdn.com/bscvsembelectrochemistry-221120153650-89641ab8/85/BSc-V-SEM-B-Electrochemistry-1-pptx-34-320.jpg)