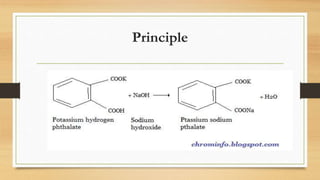
The document details the procedure for preparing and standardizing a 0.1 M sodium hydroxide solution using potassium hydrogen phthalate as the primary standard. It outlines necessary materials, the theory behind the process, and a step-by-step guide to perform the titration, including the proper preparation of reagents and equipment. The goal is to accurately determine the concentration of the sodium hydroxide solution through titration and record the results.