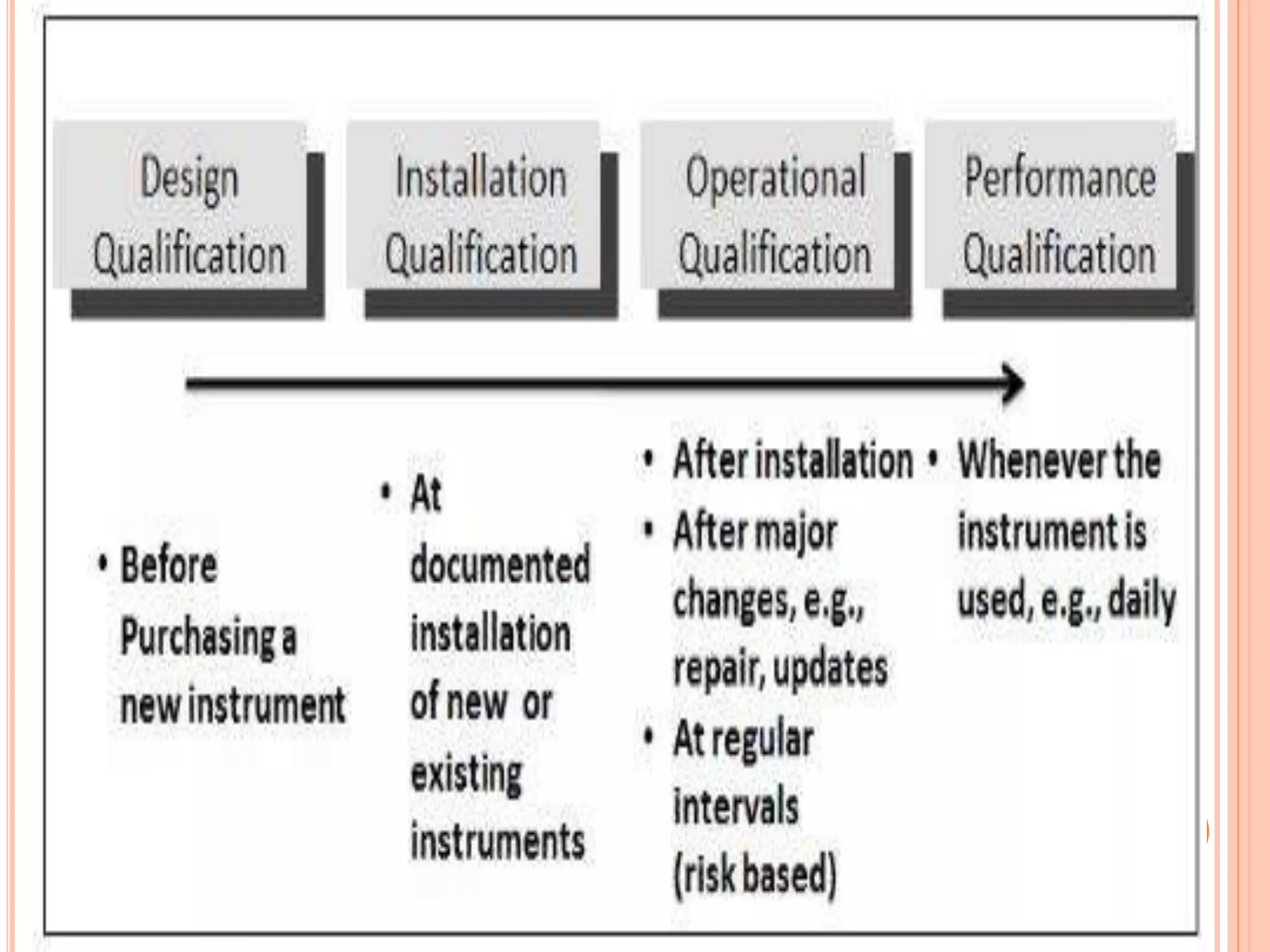
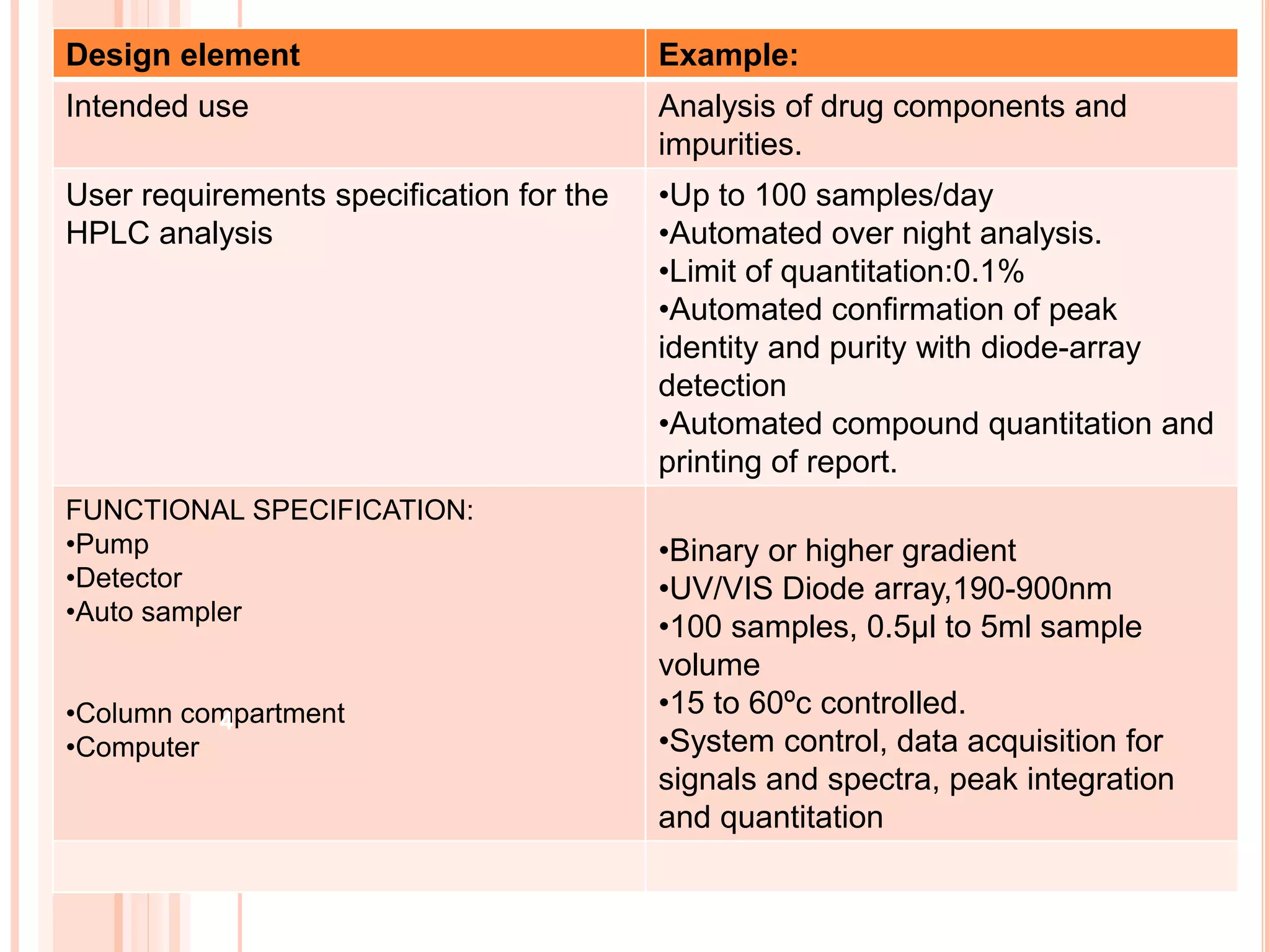
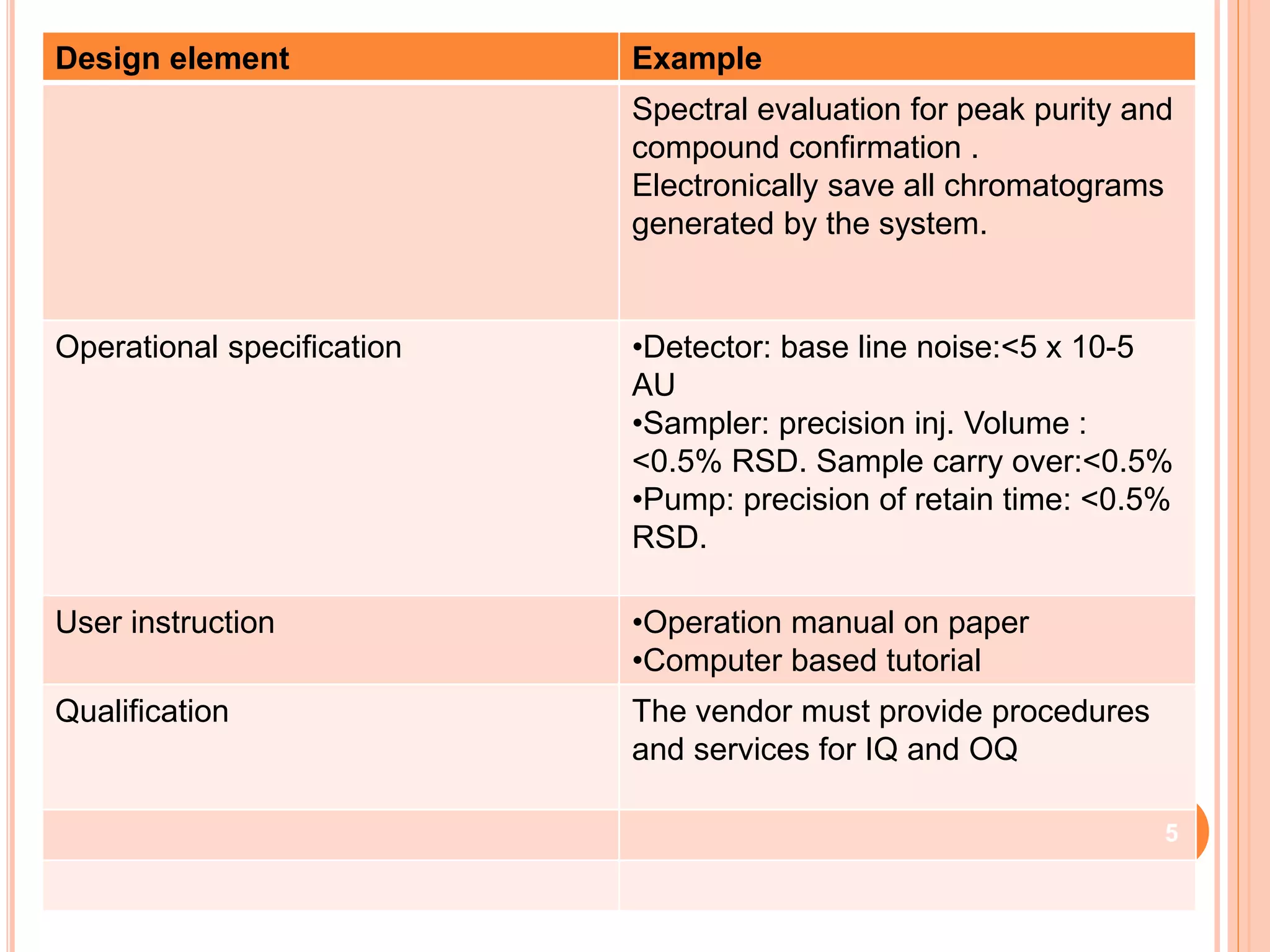
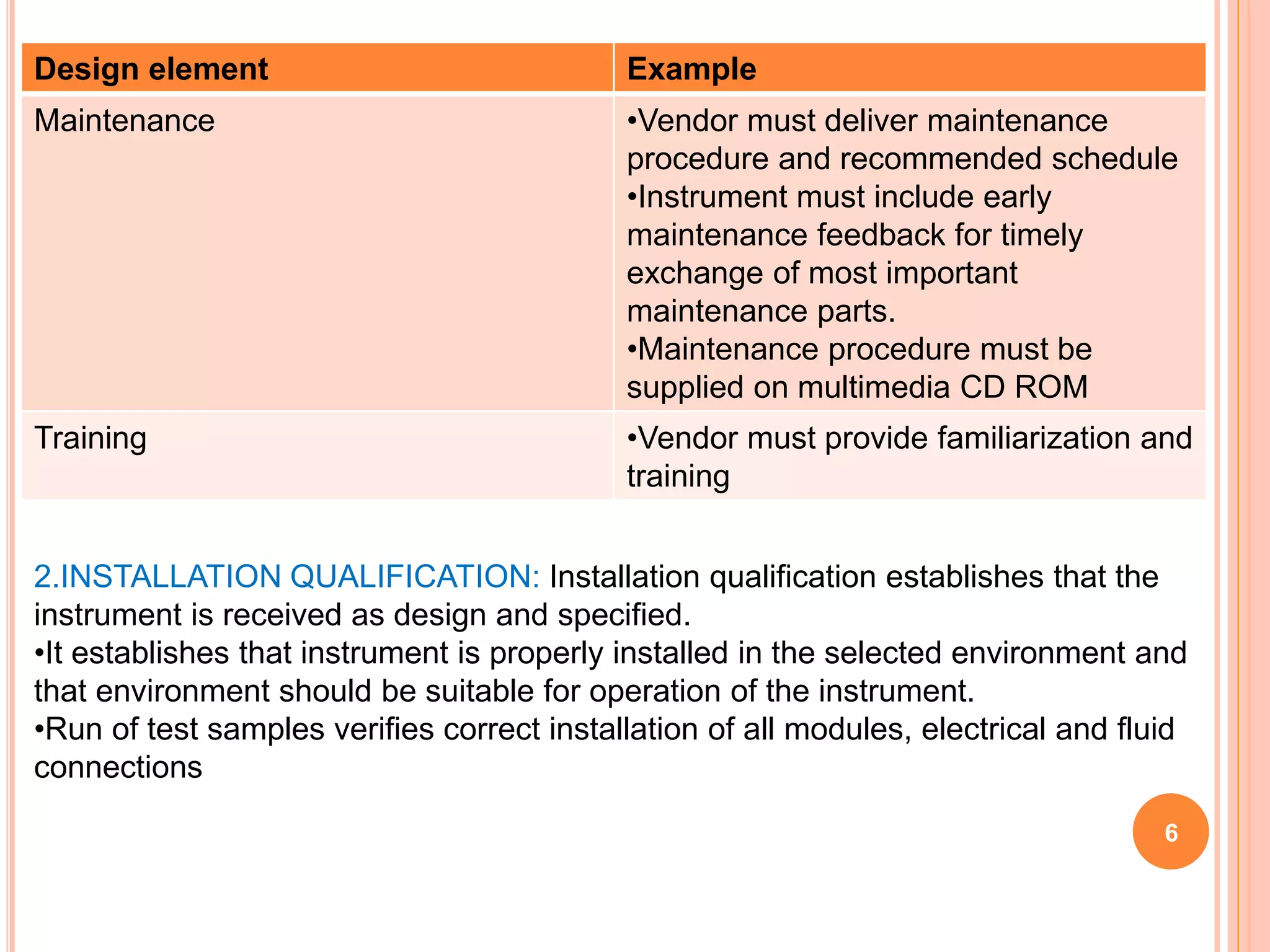
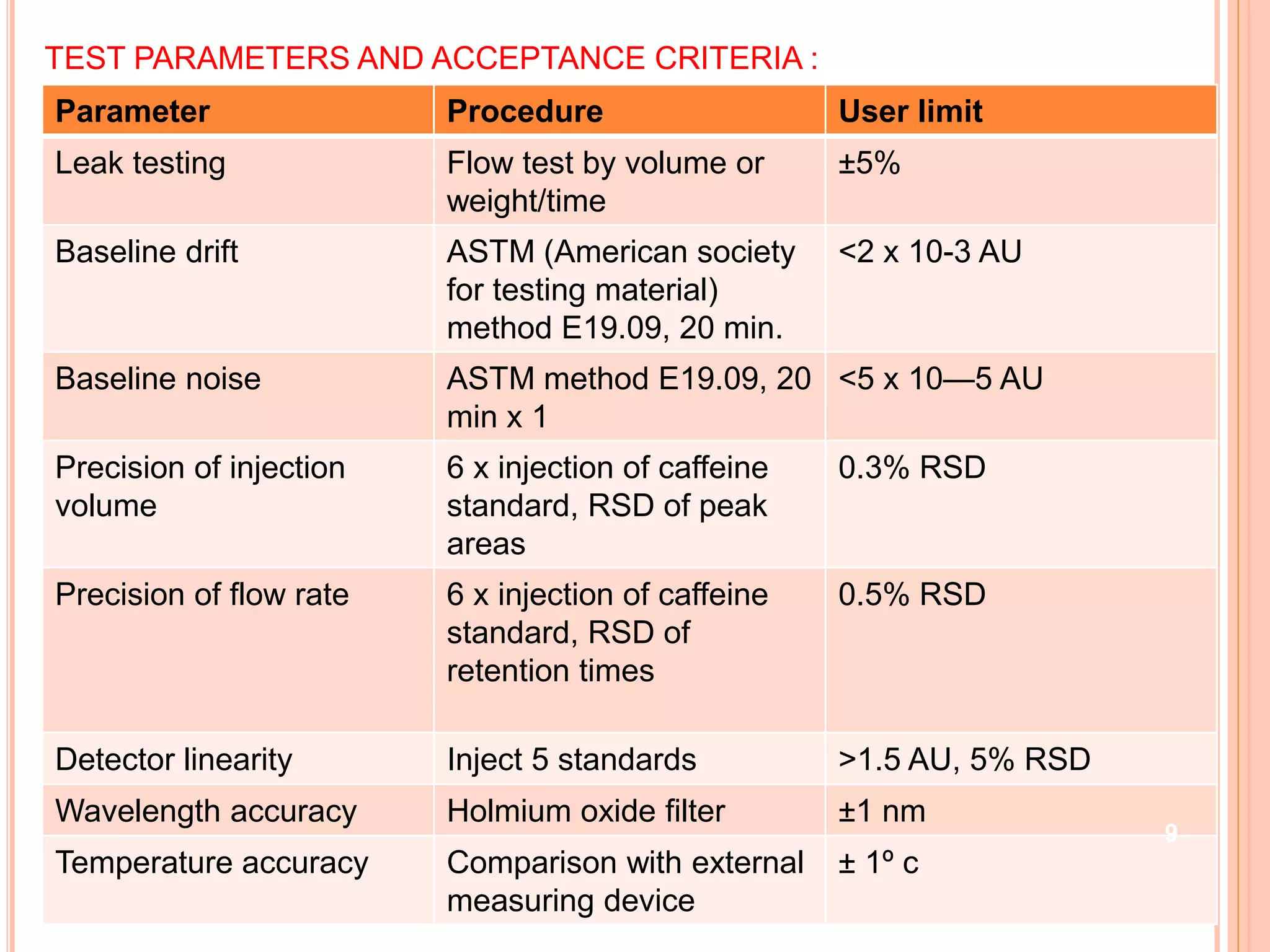
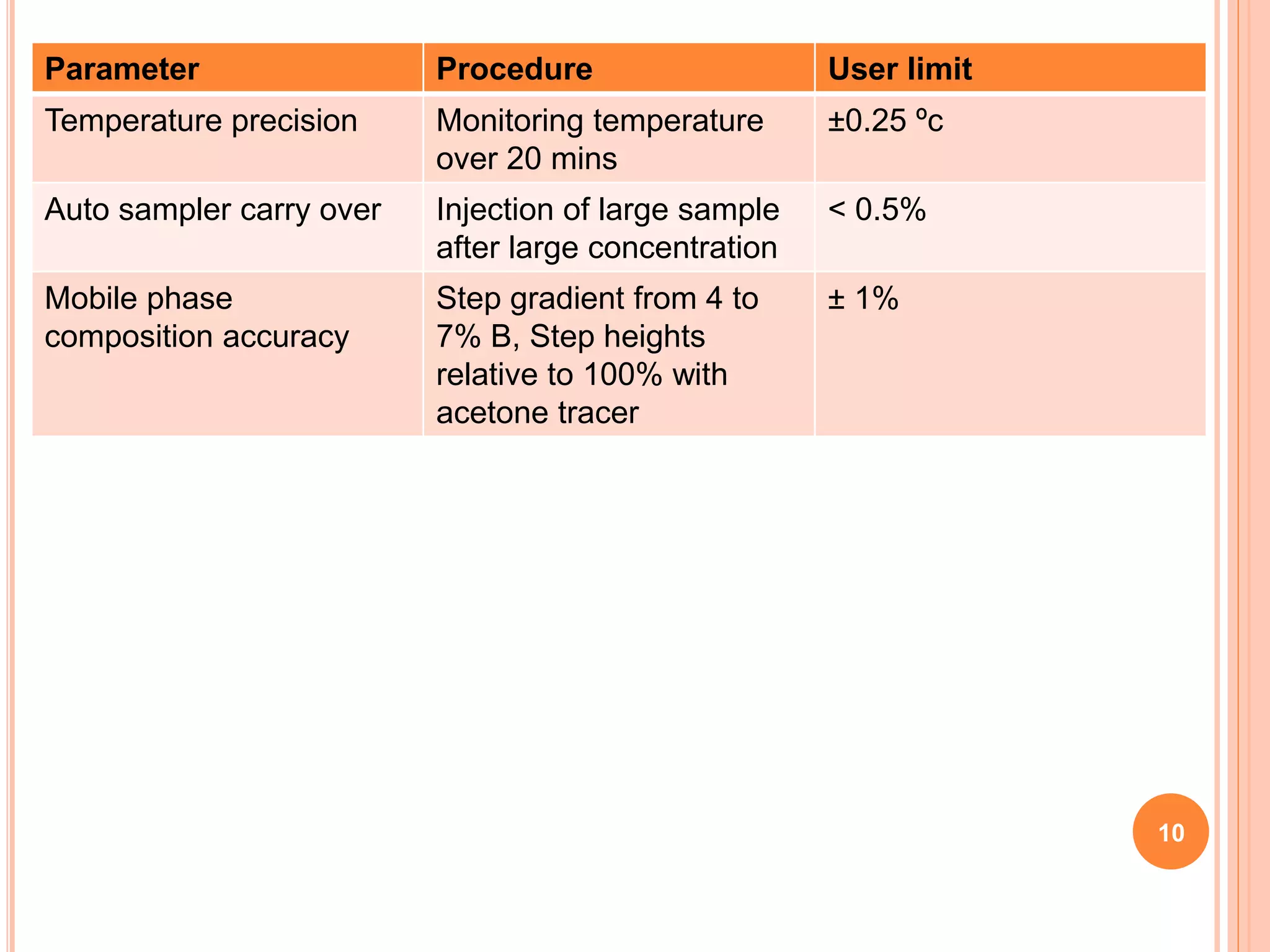
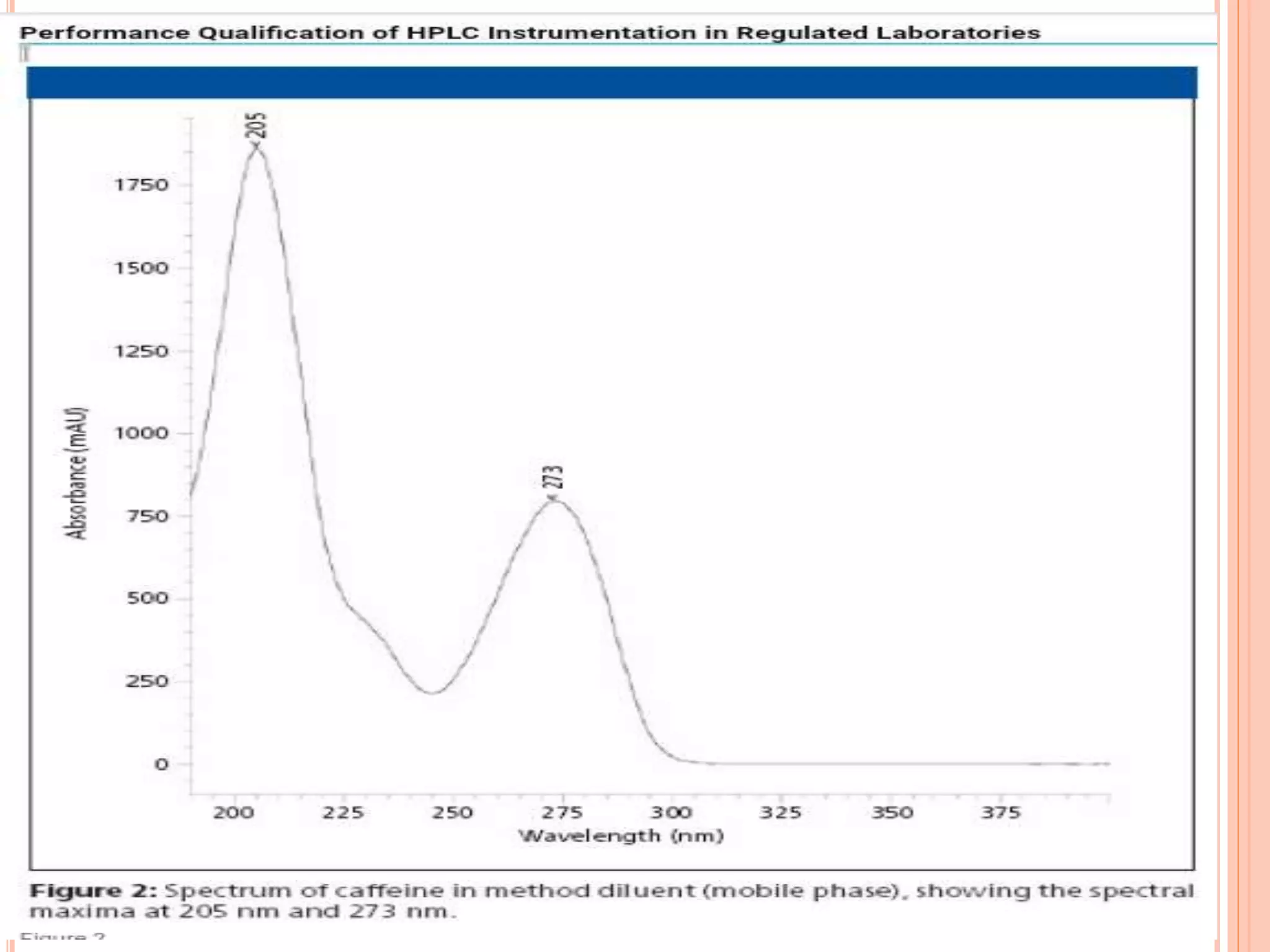
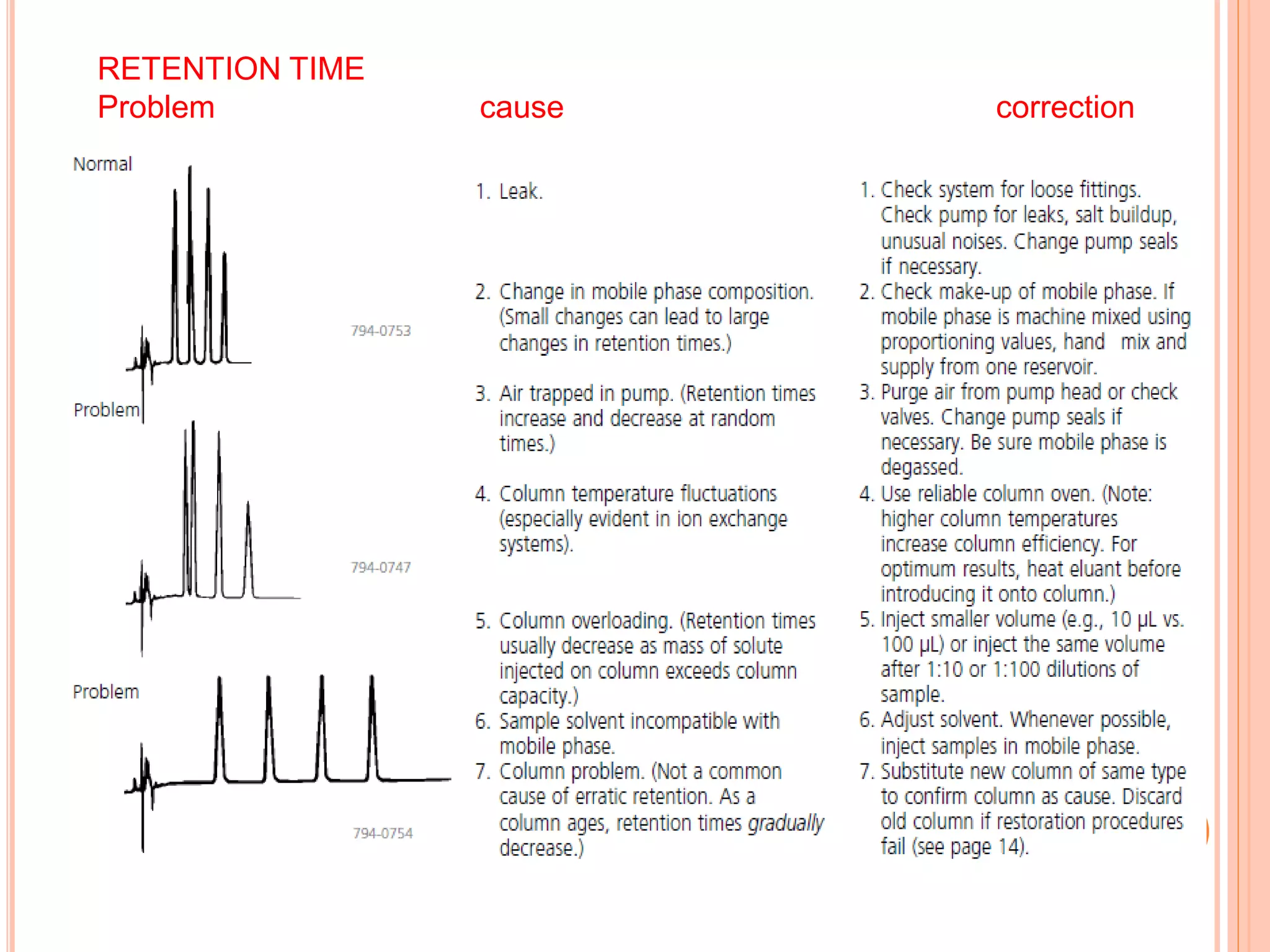
The document provides an overview of the qualification process for high performance liquid chromatography (HPLC) equipment, including design qualification (DQ), installation qualification (IQ), operational qualification (OQ), and performance qualification (PQ). It describes the objectives and procedures for each qualification step. Key aspects covered include verifying design specifications, proper installation, operational requirements such as precision, accuracy and noise levels, and ongoing performance monitoring. The goal of qualification is to ensure analytical systems are suitable for their intended use and generate reliable results.