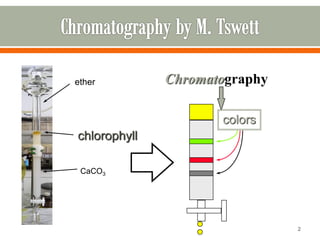
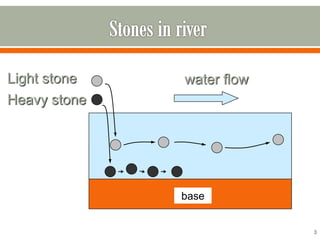
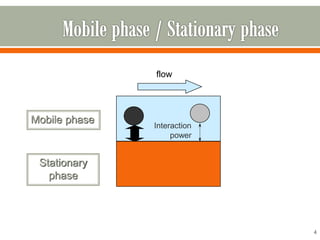
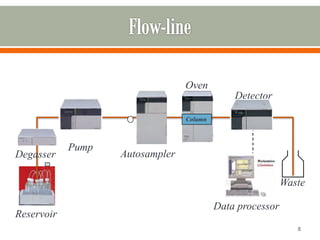
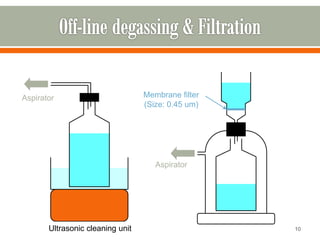
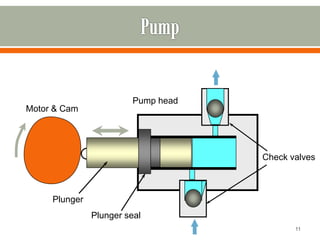
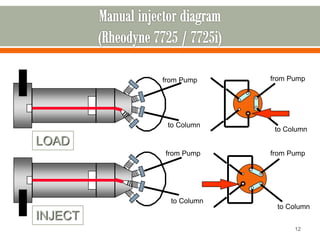
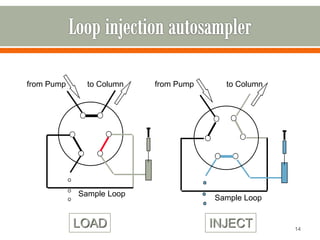
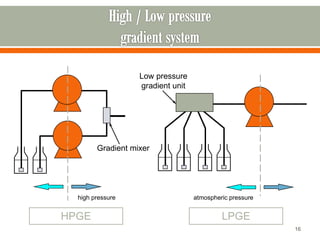
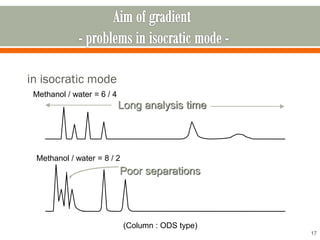
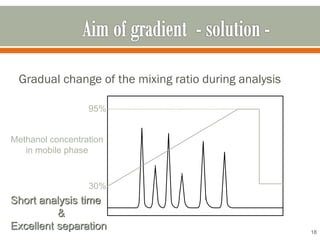
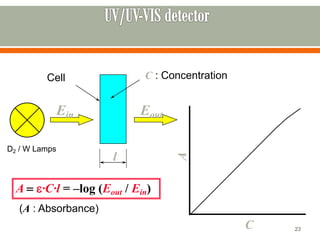
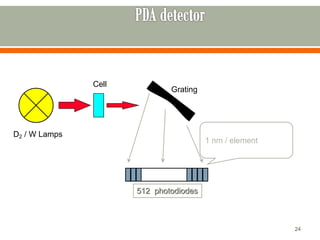
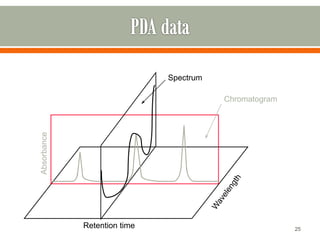
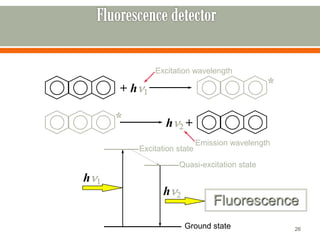
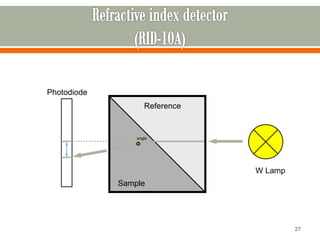
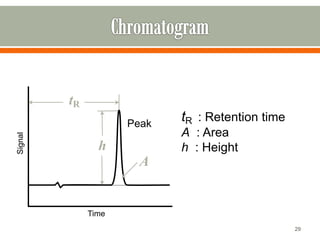
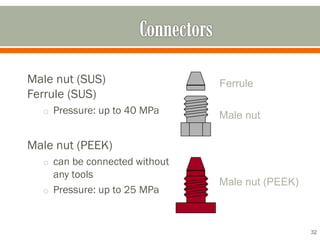
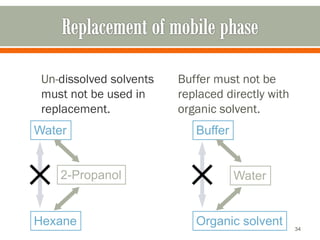
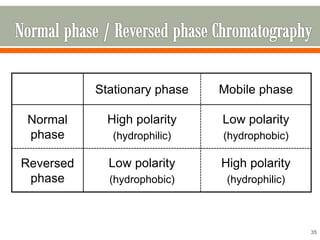
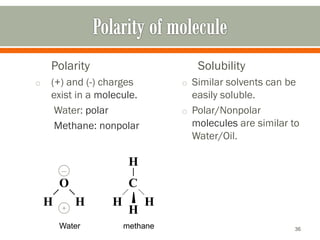
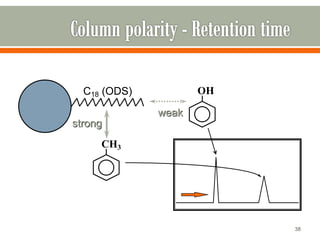
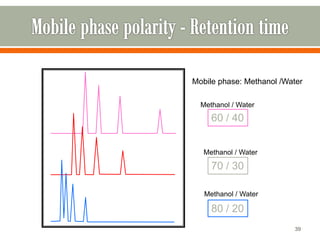
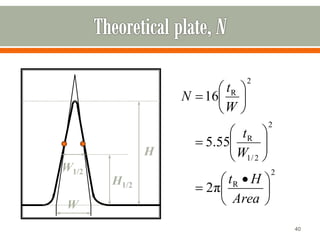
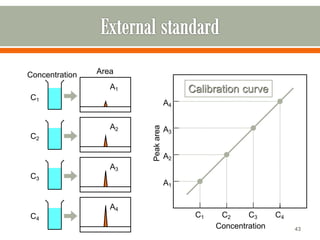
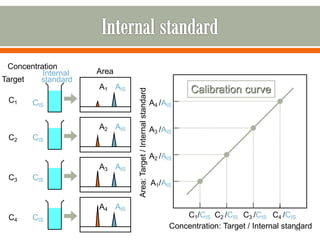
HPLC is a type of chromatography used to separate components in a mixture. It involves using a mobile phase to carry the sample through a column containing a stationary phase. Components interact differently with the phases allowing separation. Quality is important in analytical methods. Accuracy involves proper sampling, storage, analysis and minimizing errors. Quality assurance measures like method validation and quality control steps like duplicates, standards and spikes help ensure accurate results.