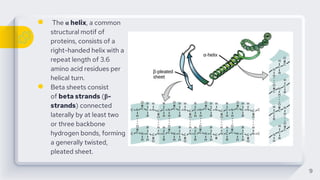
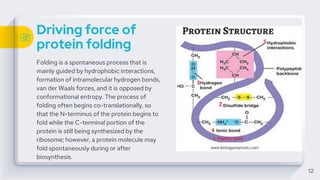
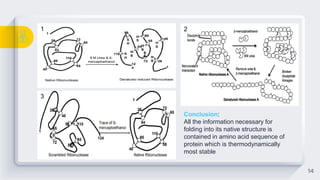
The document discusses protein folding, which is the process by which proteins achieve their functional three-dimensional structure from their linear amino acid sequence. It describes the different levels of protein structure, including primary, secondary, tertiary, and quaternary structure. The folding process depends on factors like temperature, pH, and molecular chaperones, which assist in protein folding. Proper folding is required for proteins to carry out their functions in the cell.