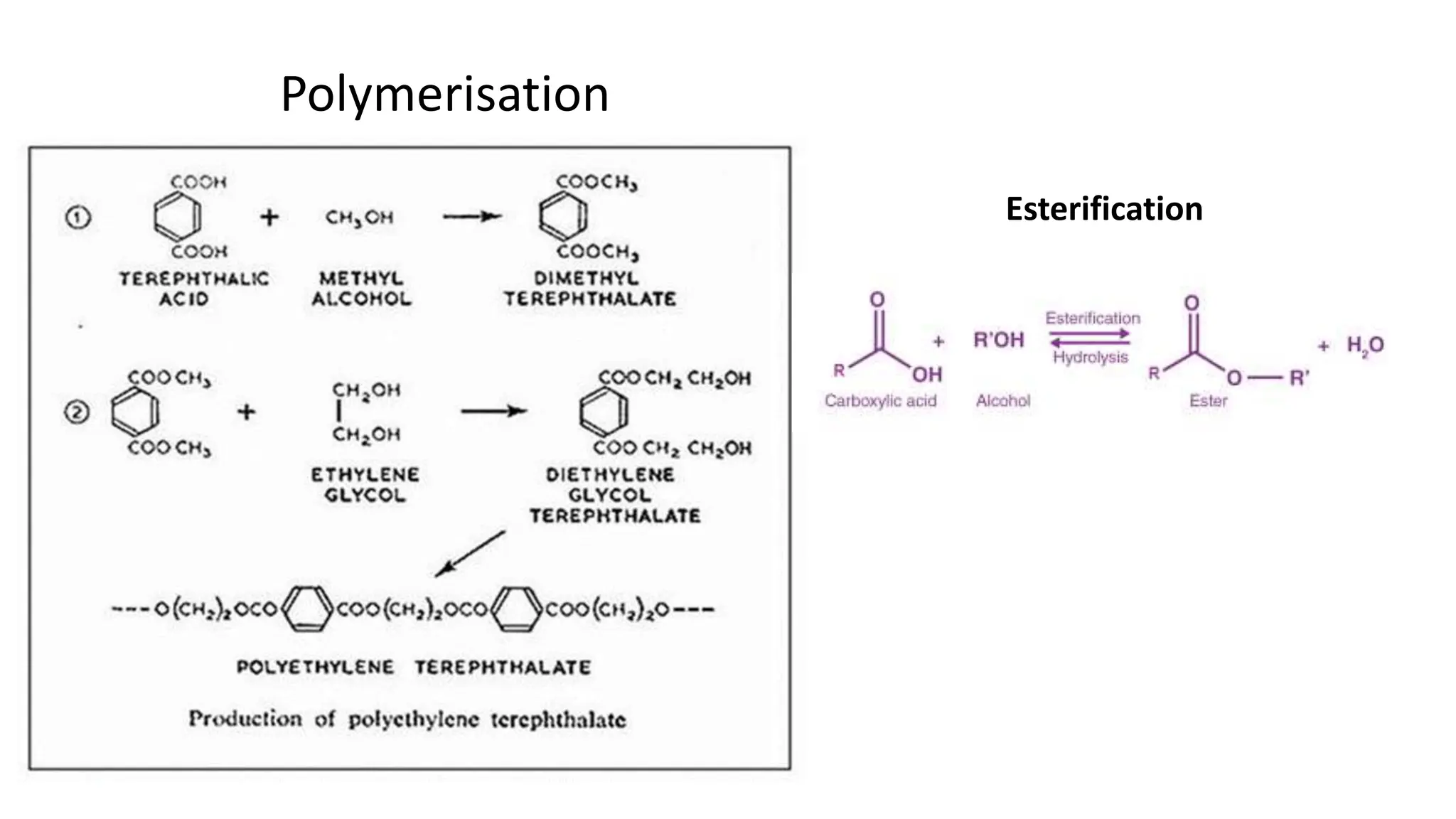
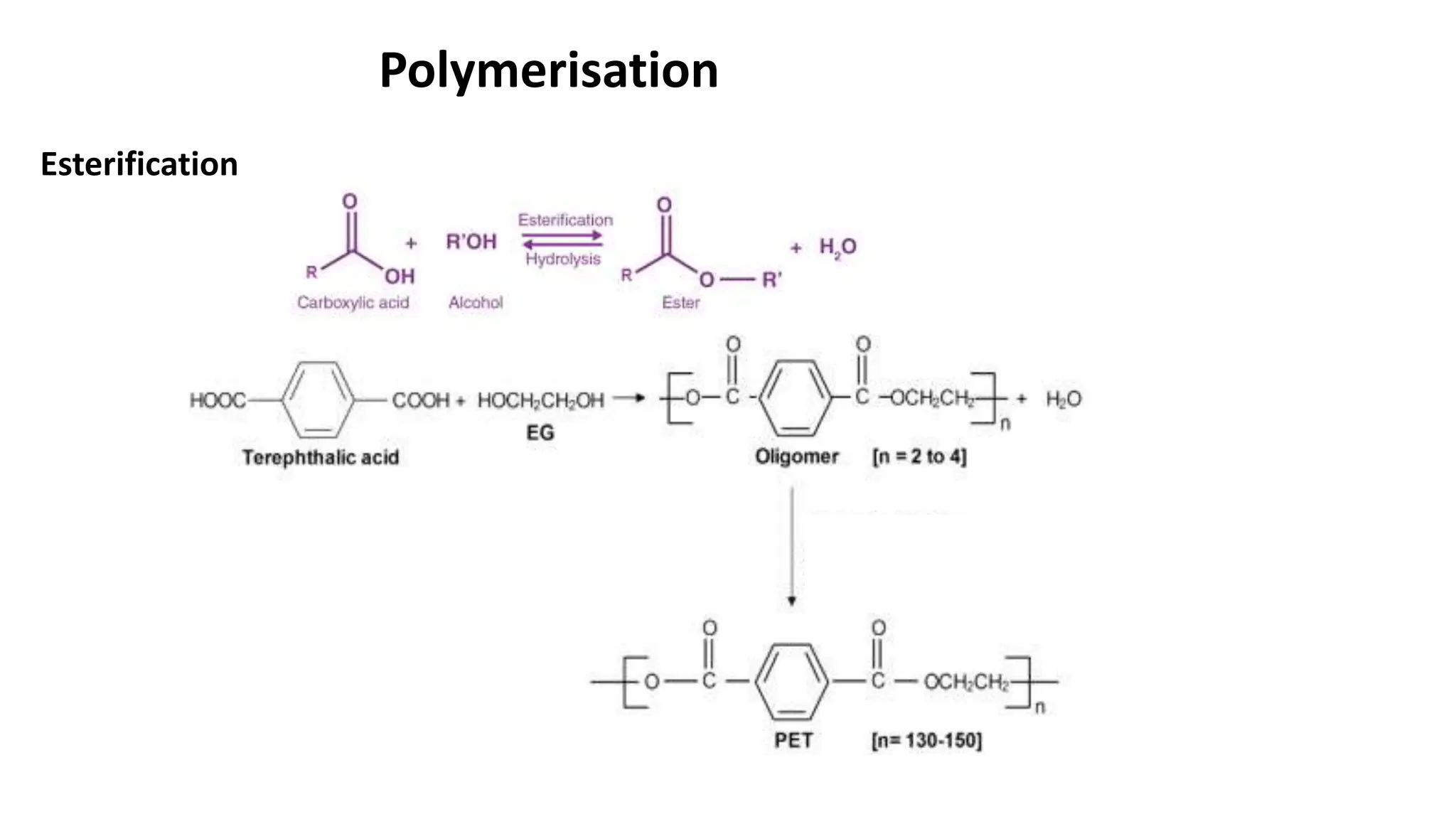
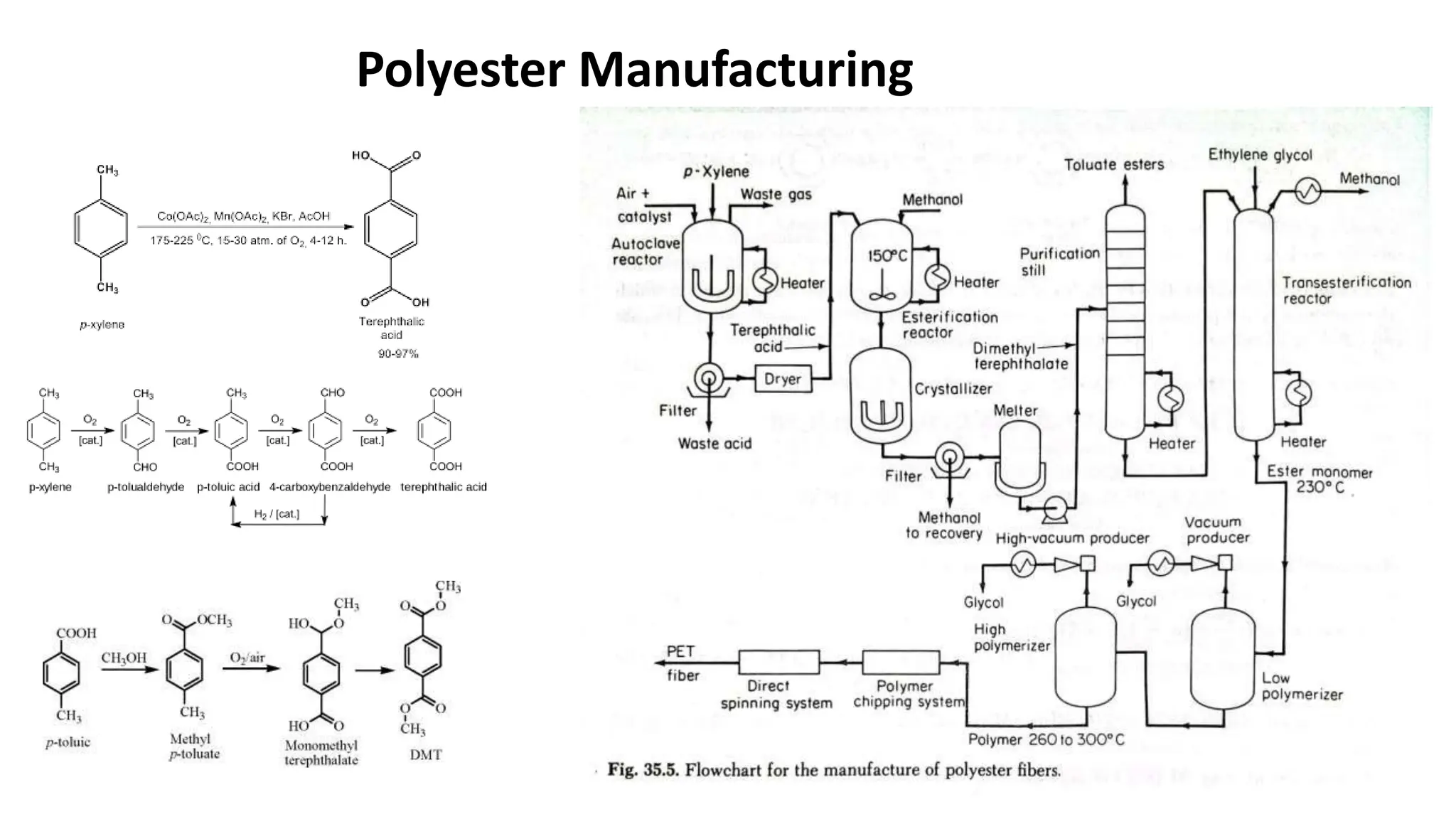
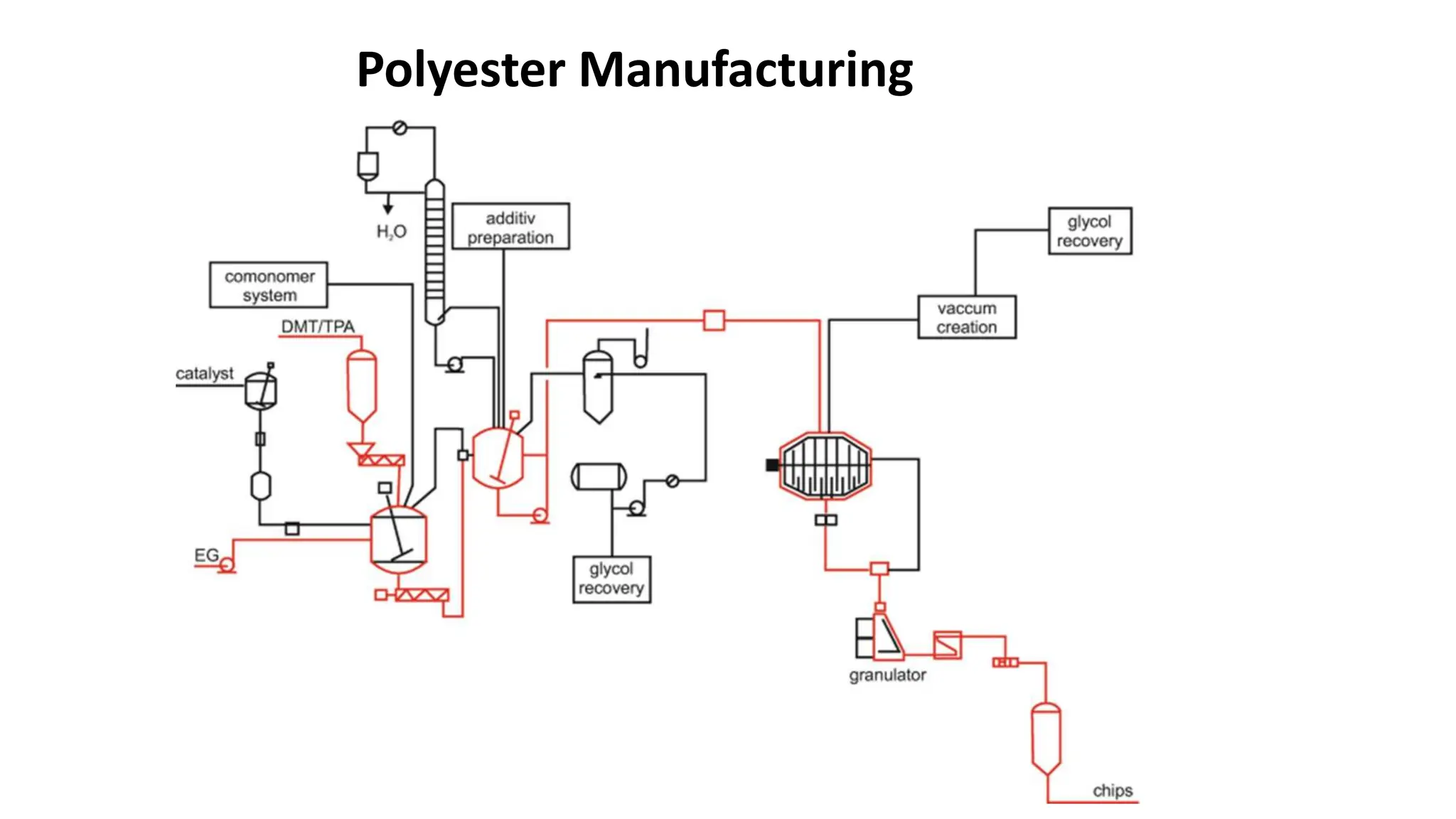
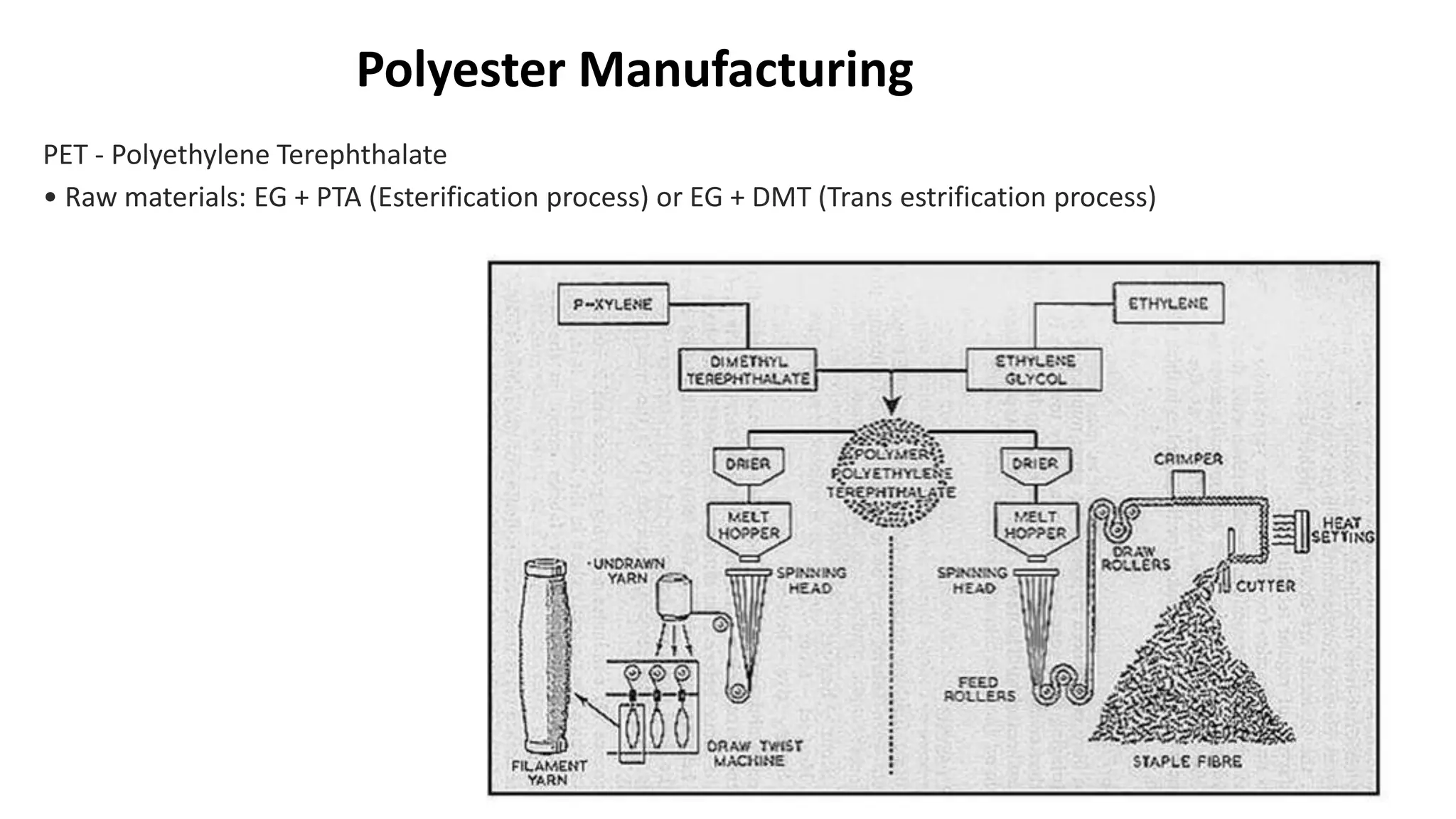
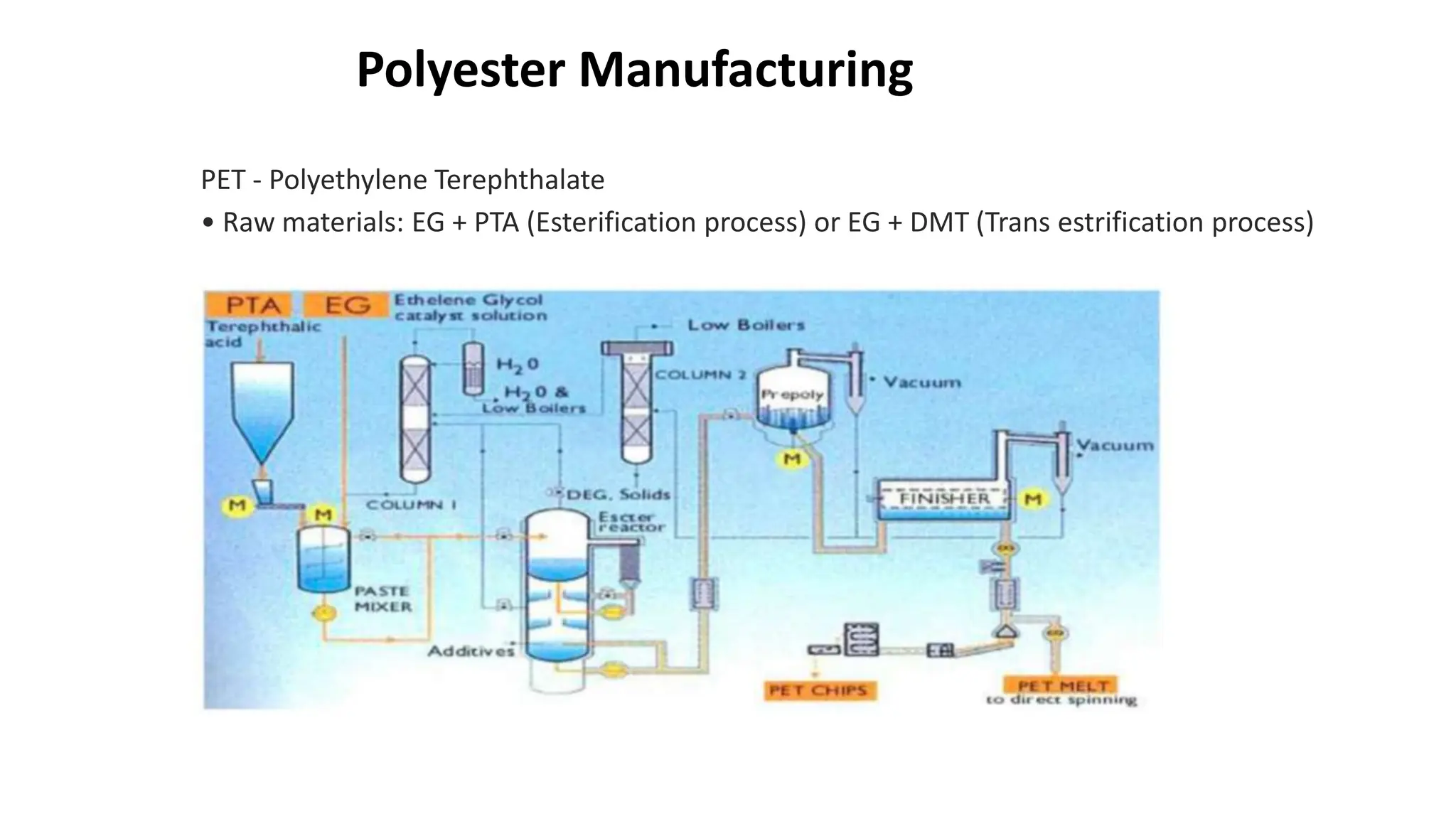
- Polyester is a polymer containing an ester functional group in each repeating unit of its main chain. Common polyesters include polyethylene terephthalate (PET) and polybutylene terephthalate (PBT).
- PET is produced from ethylene glycol and purified terephthalic acid or its ester, and is used to make polyester fibers, films, and beverage bottles. PBT has good chemical and heat resistance and is used for electrical components and automotive parts.
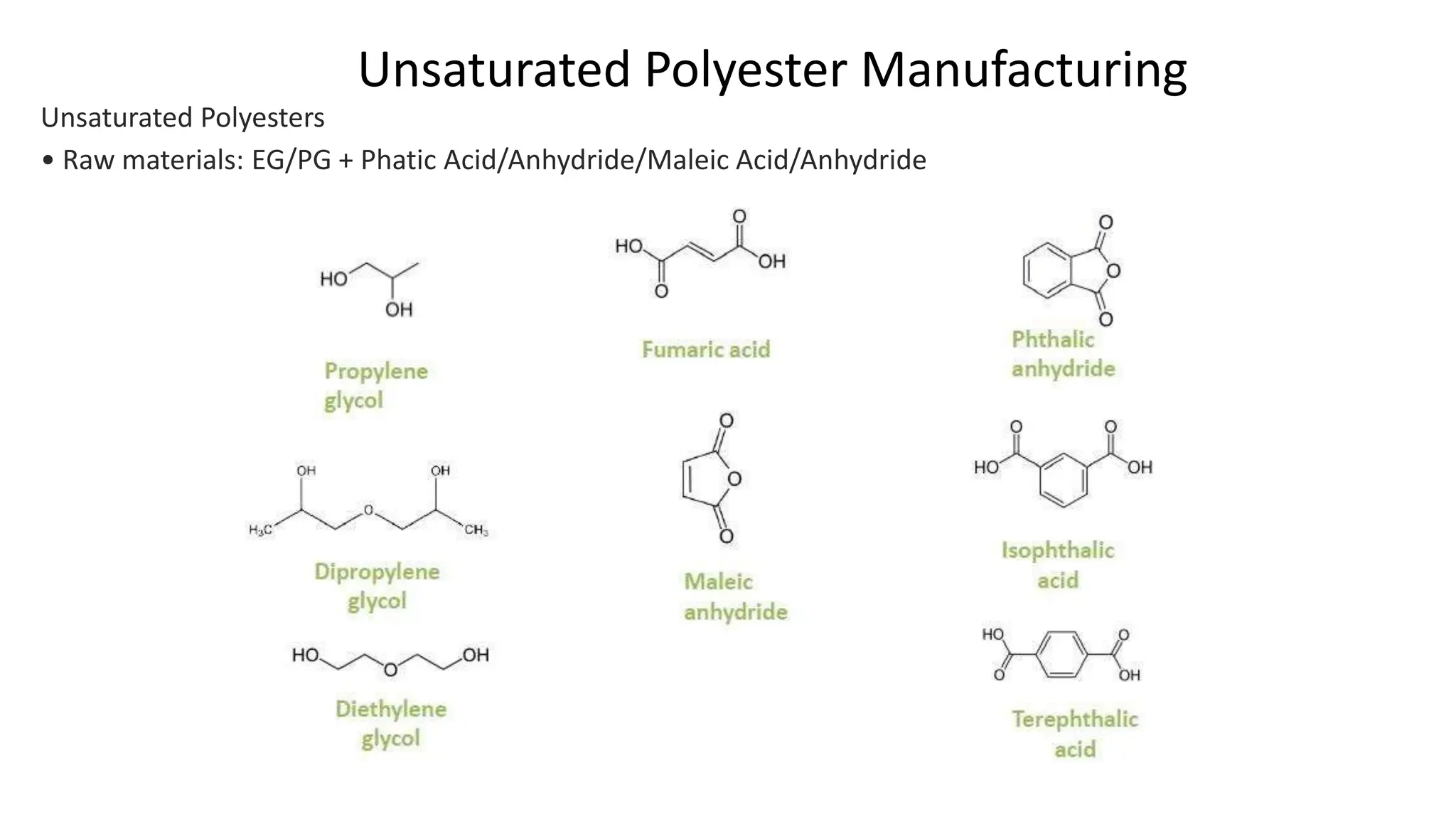
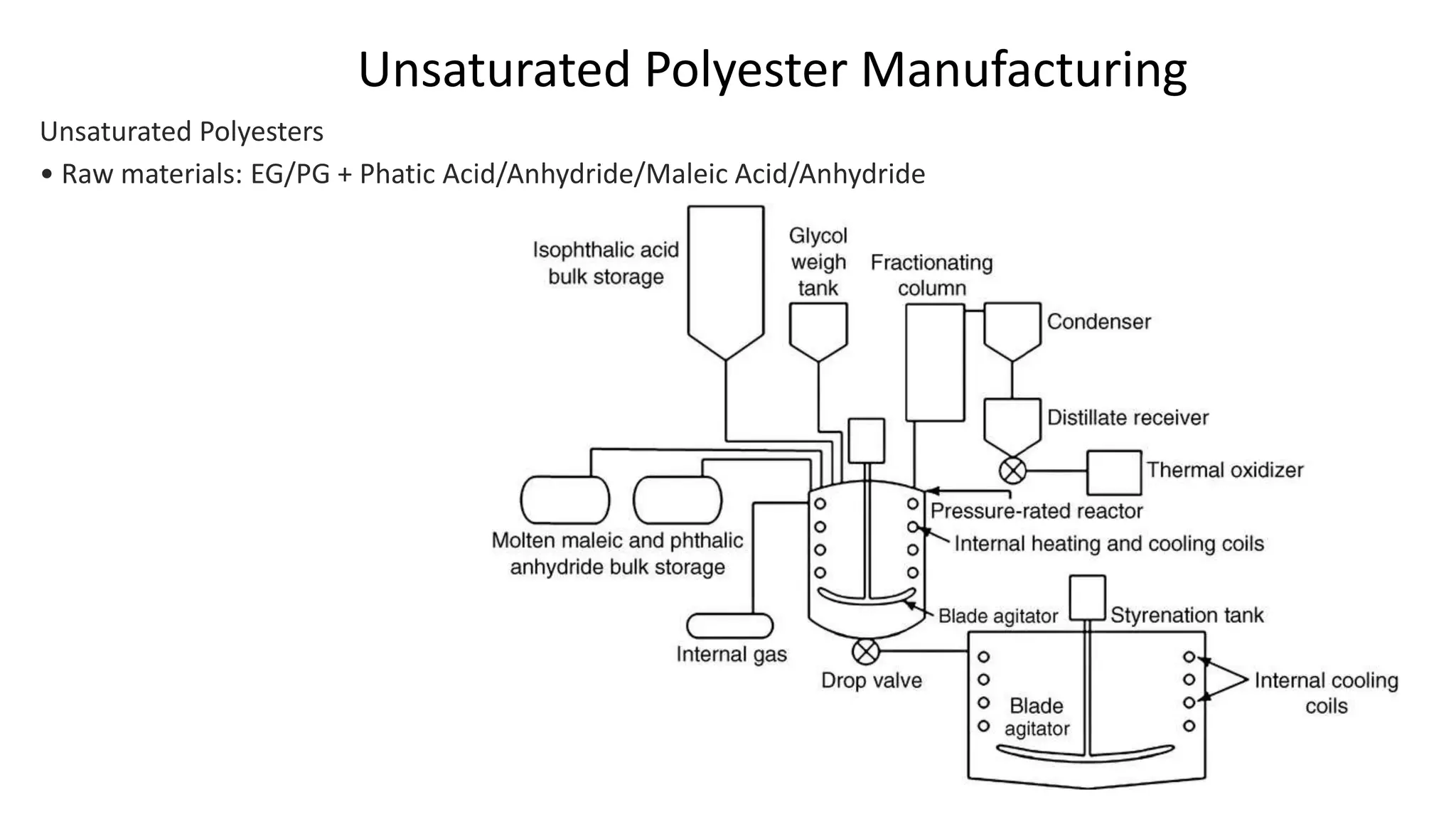
- Unsaturated polyesters are produced from glycols and acids or anhydrides and cured into thermoset polymers used in applications like fiberglass composites. Specialty polyesters include cast films