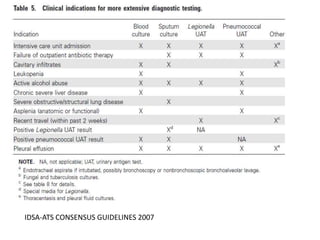
This document provides guidelines for the diagnosis and management of community-acquired pneumonia (CAP). It defines CAP and discusses its epidemiology and common causes. Streptococcus pneumoniae is often the leading cause worldwide, though causes can vary regionally in India. Chest radiography is important for diagnosis but has limitations. Computed tomography is not routinely needed. The role of microbiological testing of blood and sputum in hospitalized patients is outlined.