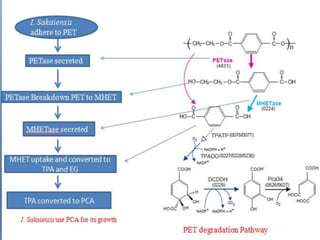
The document discusses the widespread use and environmental persistence of plastics like PET. It describes the identification of Ideonella sakaiensis bacteria that is able to use PET as a carbon source through the secretion of two enzymes, including a PET hydrolase. Testing showed the PET hydrolase efficiently breaks down PET and its intermediates, even when PET is highly crystalline. The enzyme is highly specific to PET compared to other substrates.