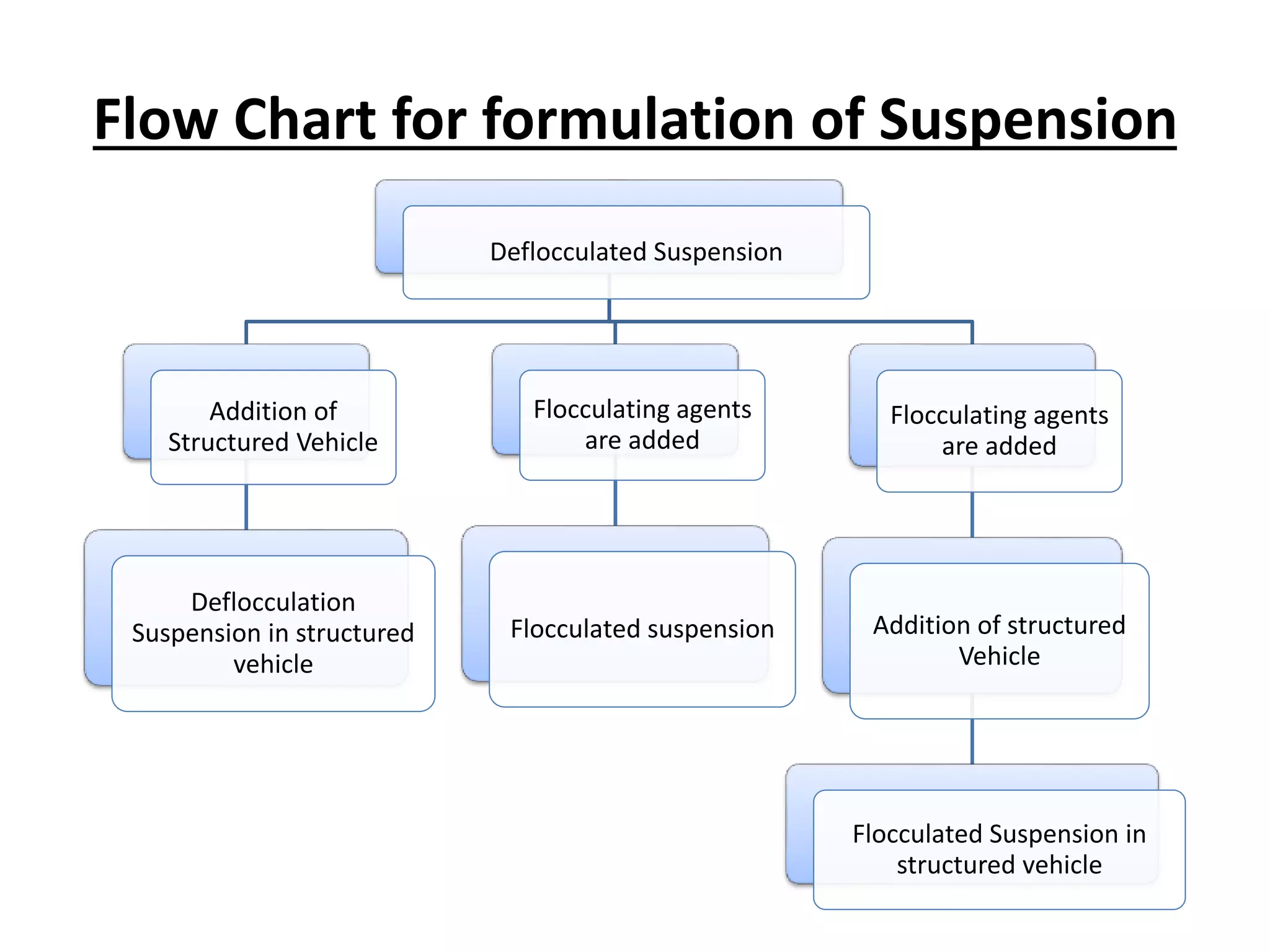
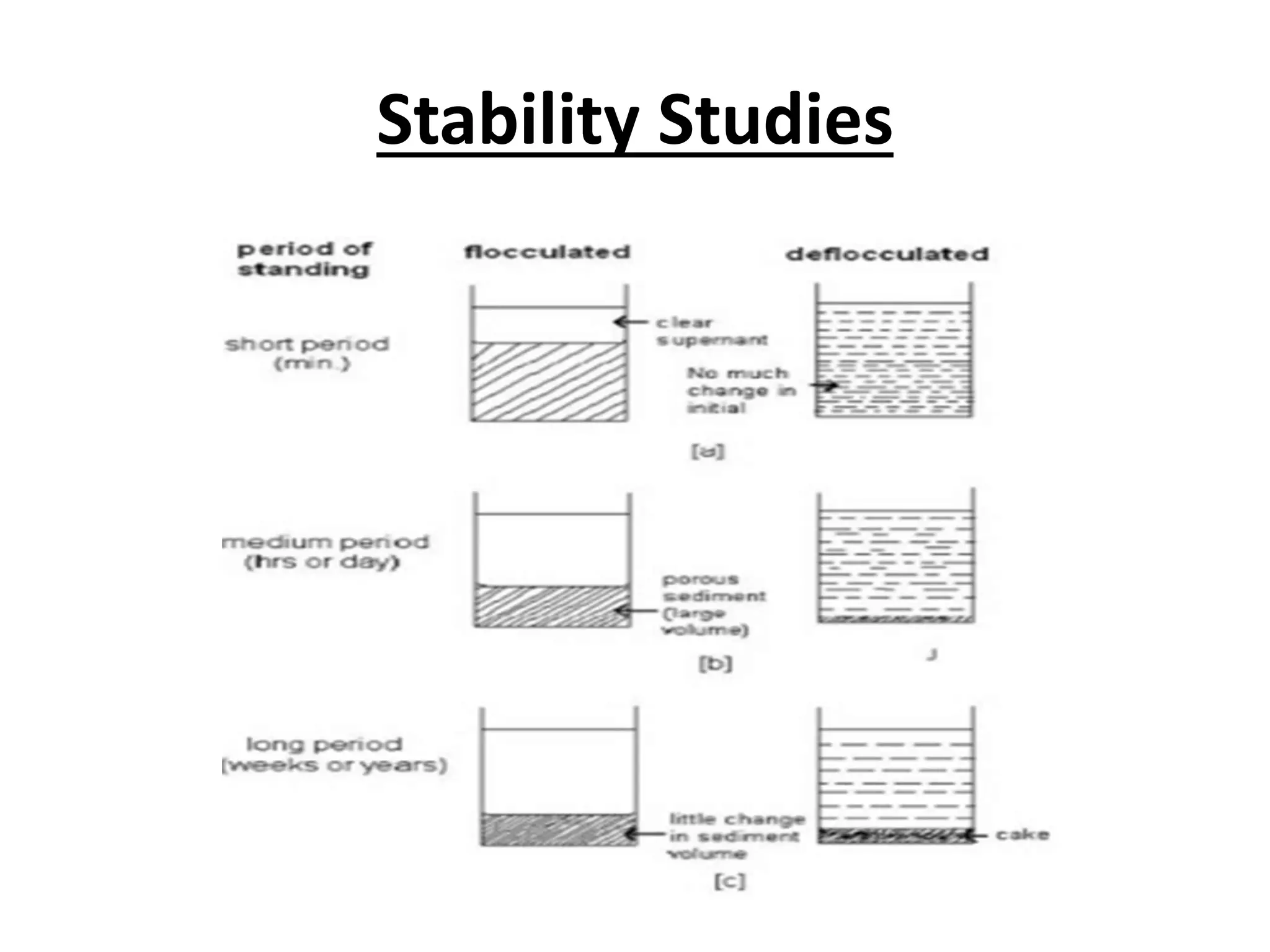
The document discusses suspensions, including definitions, classifications, properties, advantages, disadvantages, formulation methods, components, and equipment used. A suspension is a heterogeneous system with small insoluble particles dispersed uniformly throughout a liquid medium. Suspensions can be classified based on physical state, proportion of solids, particle behavior, size, and application (oral, topical, parenteral). Key aspects in formulation include reducing particle size, selecting suspending agents, wetting agents, and structured vehicles. Common equipment used are mortar and pestle, mechanical stirrers, colloid mills, homogenizers, and ultrasonic devices.