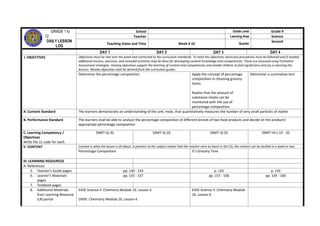
This daily lesson log from a grade 9 science teacher outlines the week's objectives and lessons on atomic structure and bonding. The objectives for the week are to understand ionic and covalent bonding through activities illustrating how ions are formed and modeling metallic bonding. Lessons cover writing electron configurations, relating valence electrons and properties, forming ions, and ionic and covalent bonding. Formative assessments evaluate the students' abilities to write configurations, describe bonding principles, and explain properties based on bonding type.