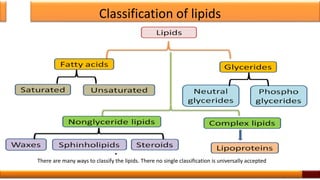
The document provides a comprehensive overview of lipids, defining them as diverse compounds related to fatty acids that are insoluble in water but soluble in organic solvents. It discusses their chemical nature, physical properties, classification, and biological functions, emphasizing their roles as energy sources, components of membranes, and chemical messengers. Additionally, it outlines various chemical properties and the classification of lipids into categories such as fatty acids, glycerolipids, and steroids.