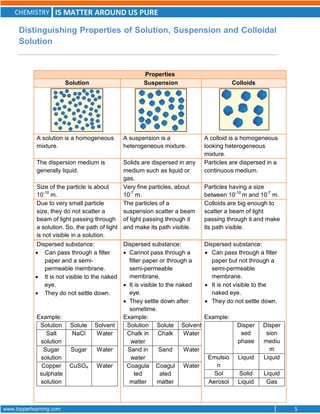
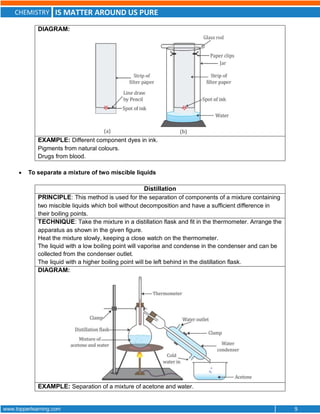
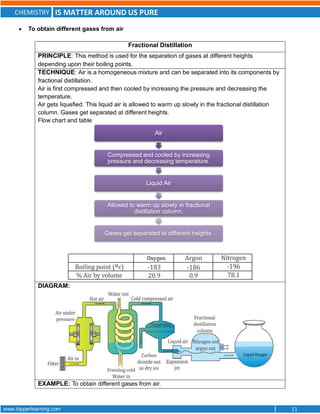
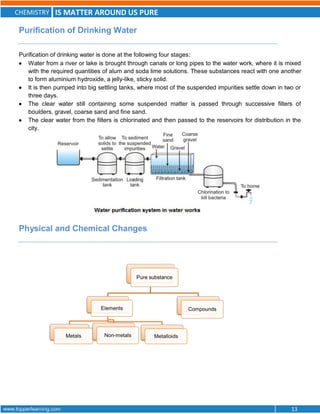
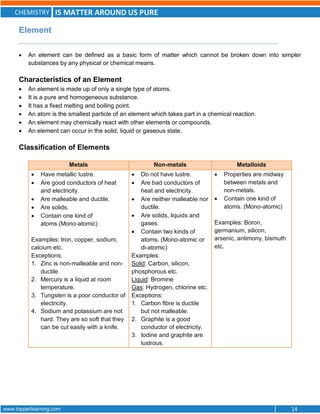
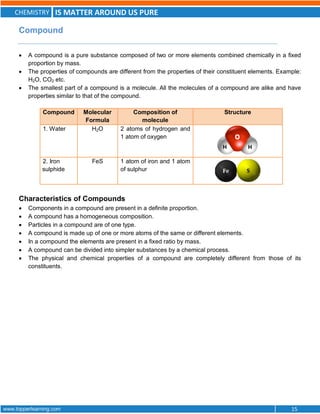
This document discusses the differences between pure substances, mixtures, and the various types of mixtures. It defines pure substances as homogeneous materials with a definite chemical composition, while mixtures contain two or more substances mixed together. Solutions are homogeneous mixtures whose composition can vary, while suspensions and colloids are heterogeneous mixtures. Several methods for separating mixtures like evaporation, centrifugation, and chromatography are also summarized.