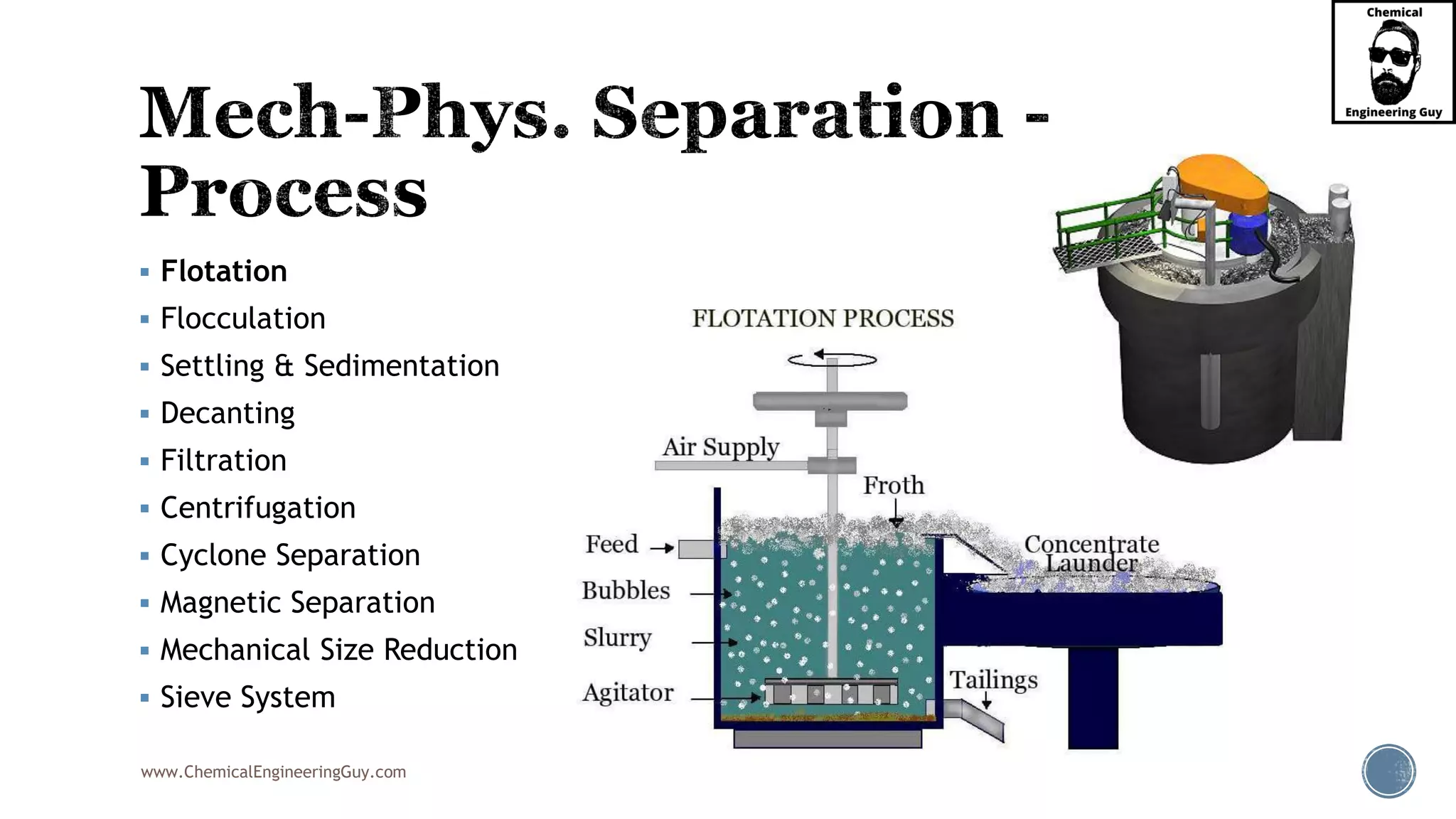
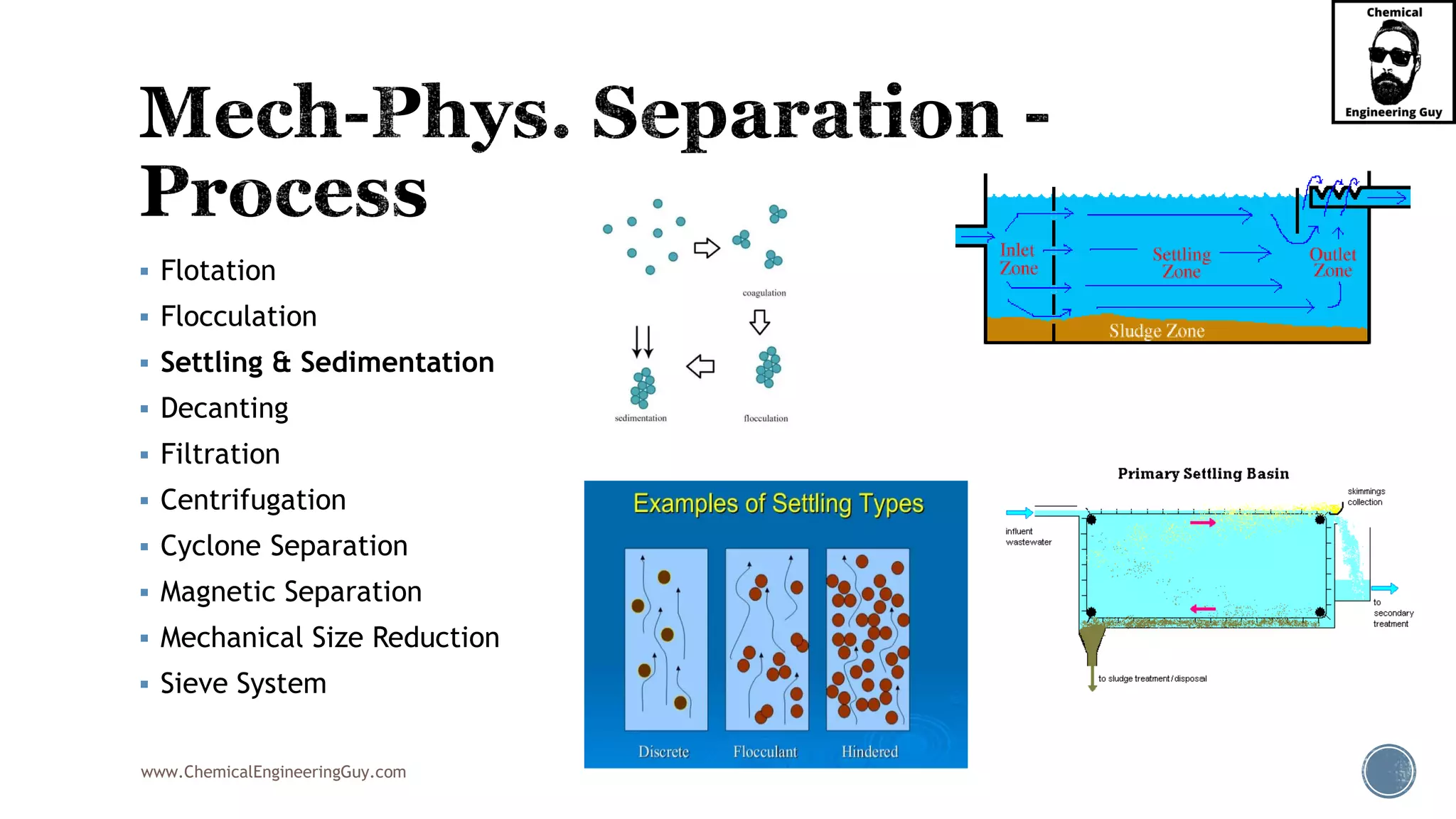
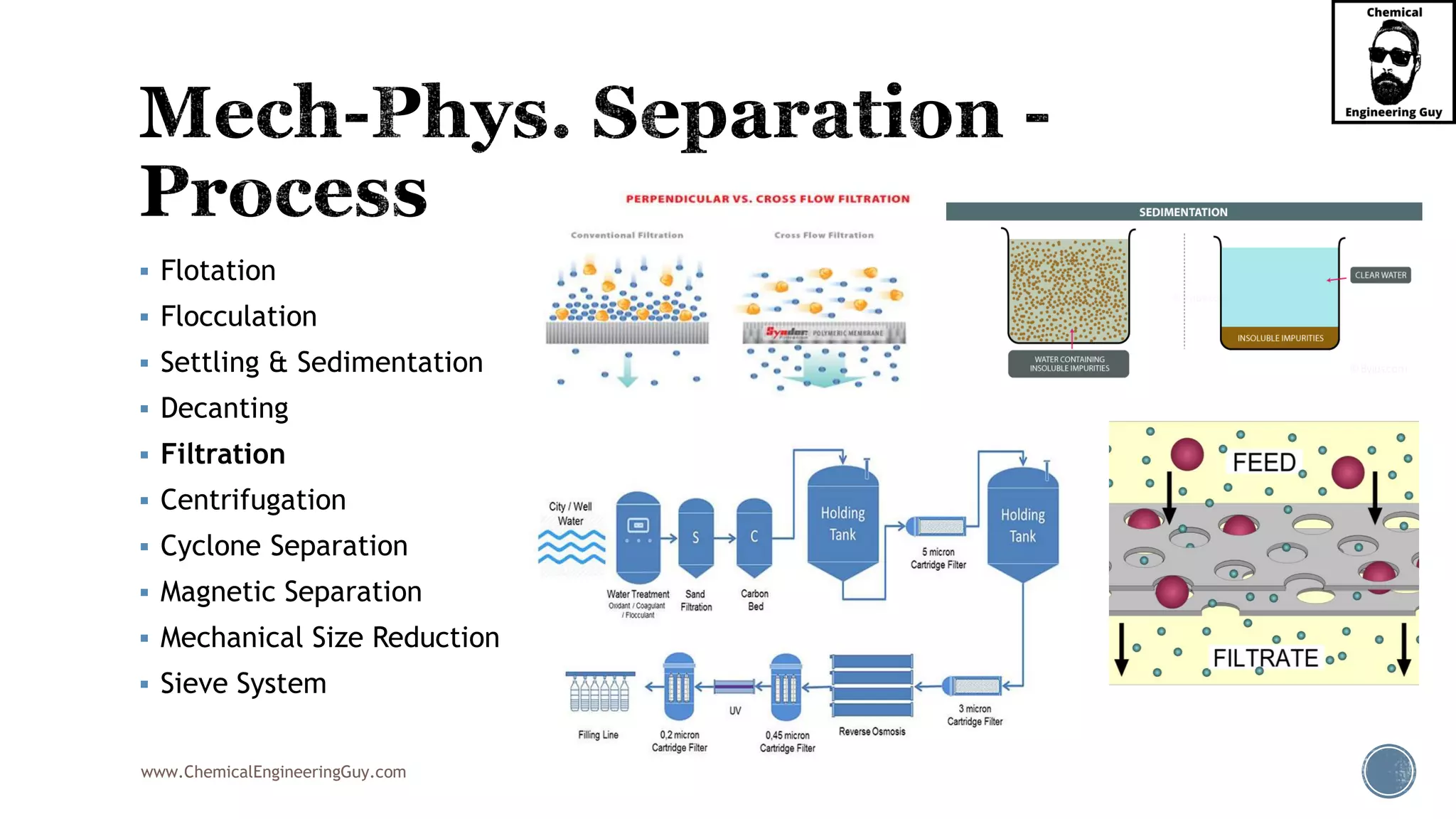
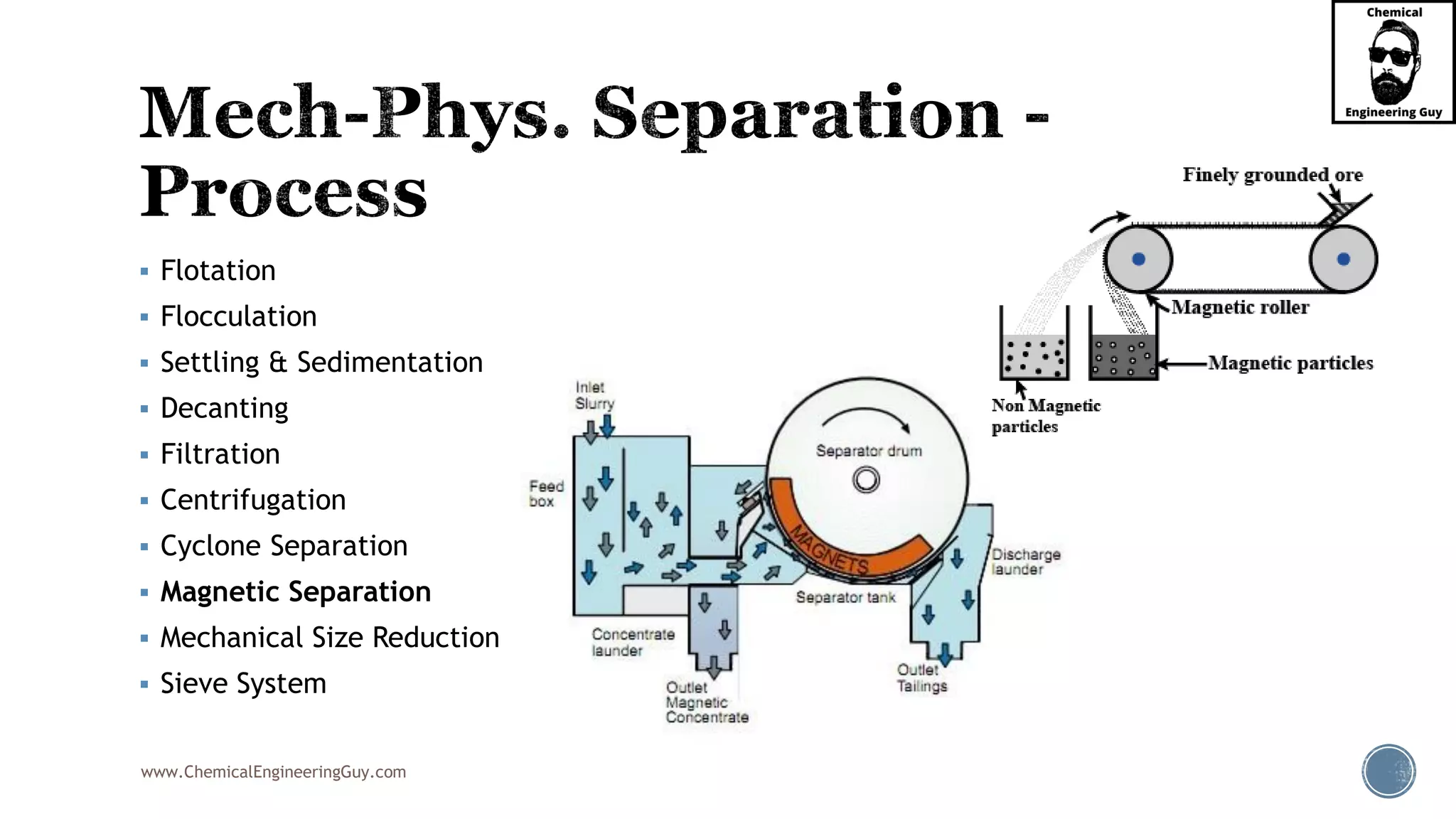
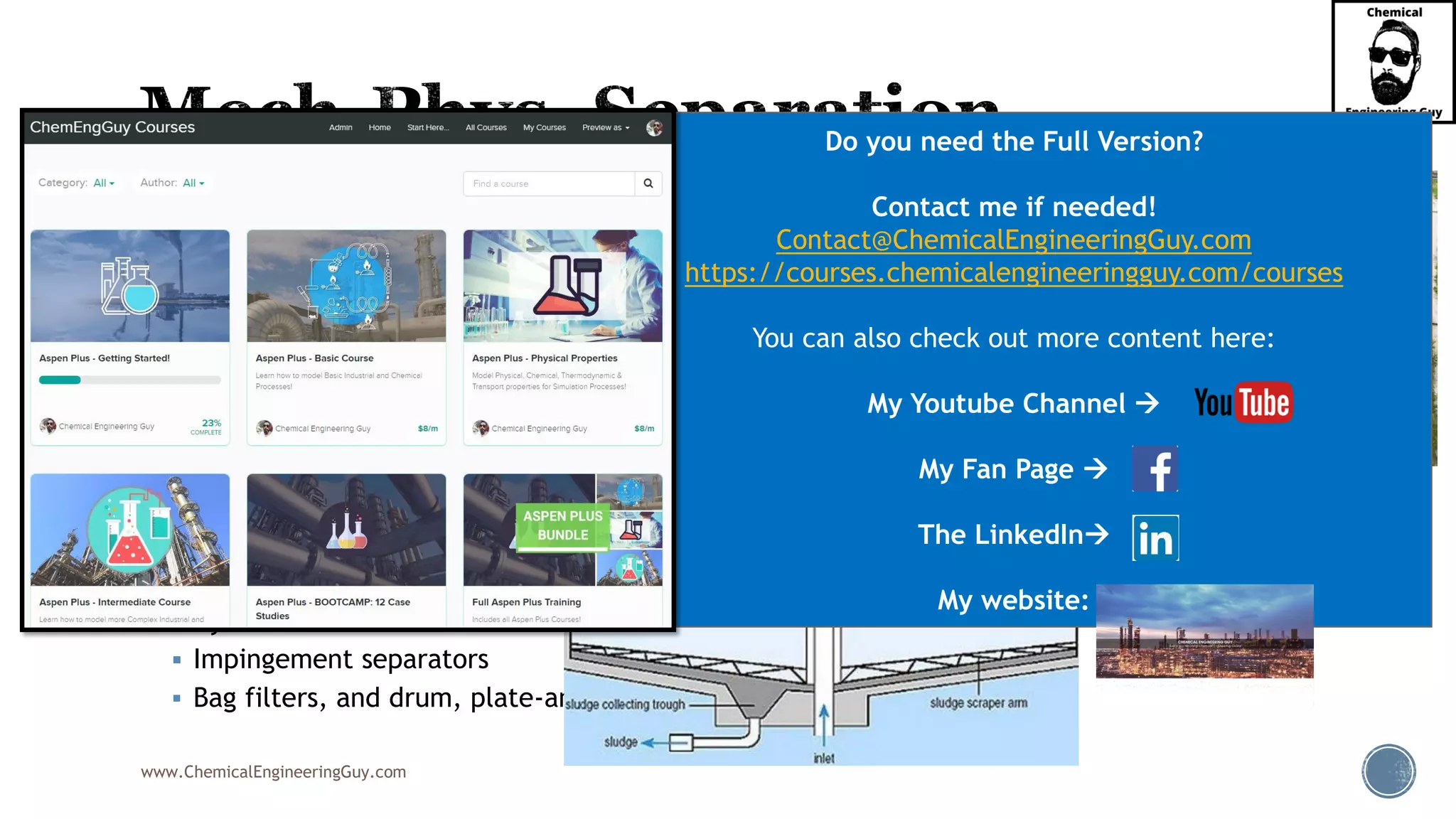
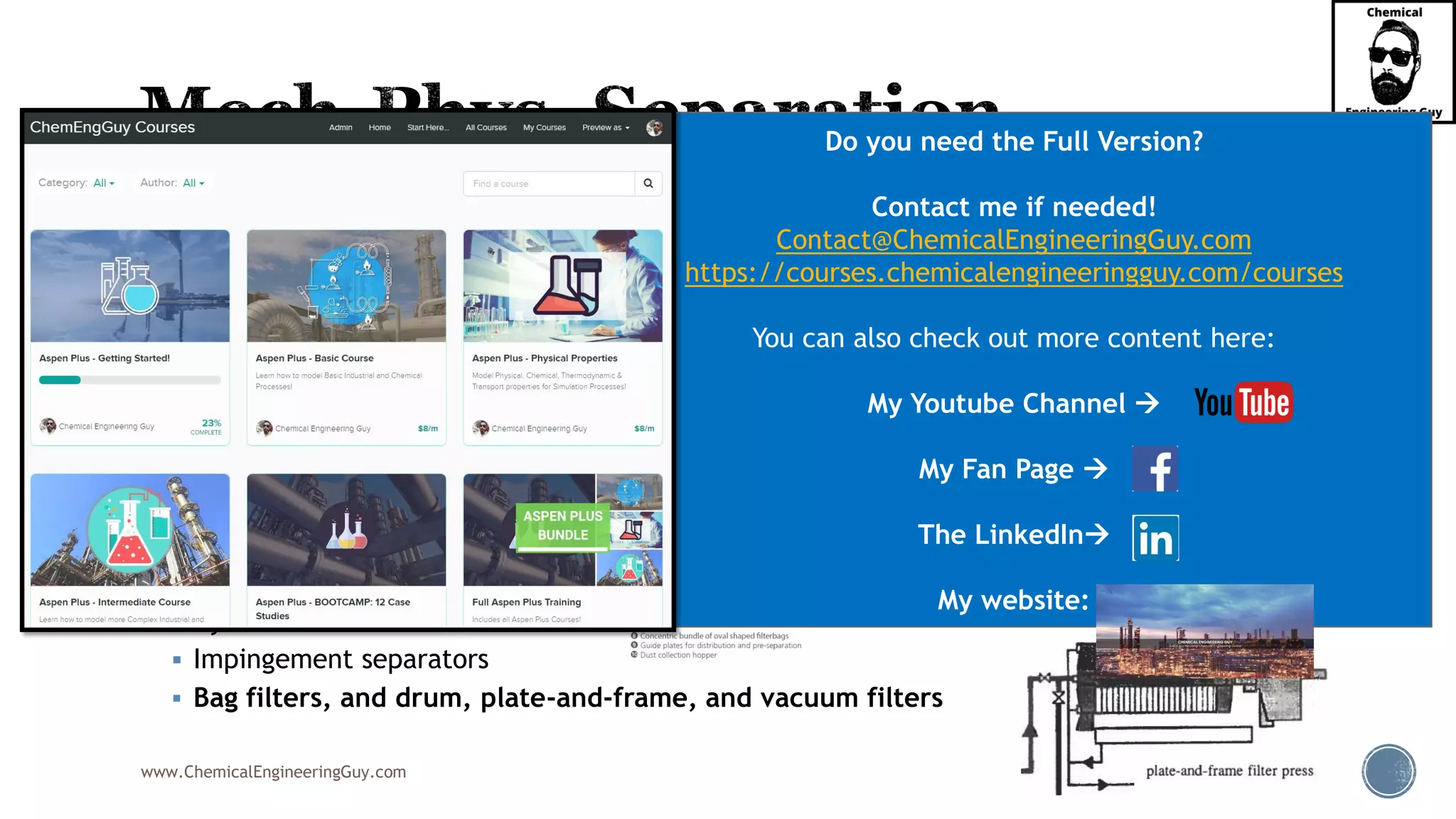
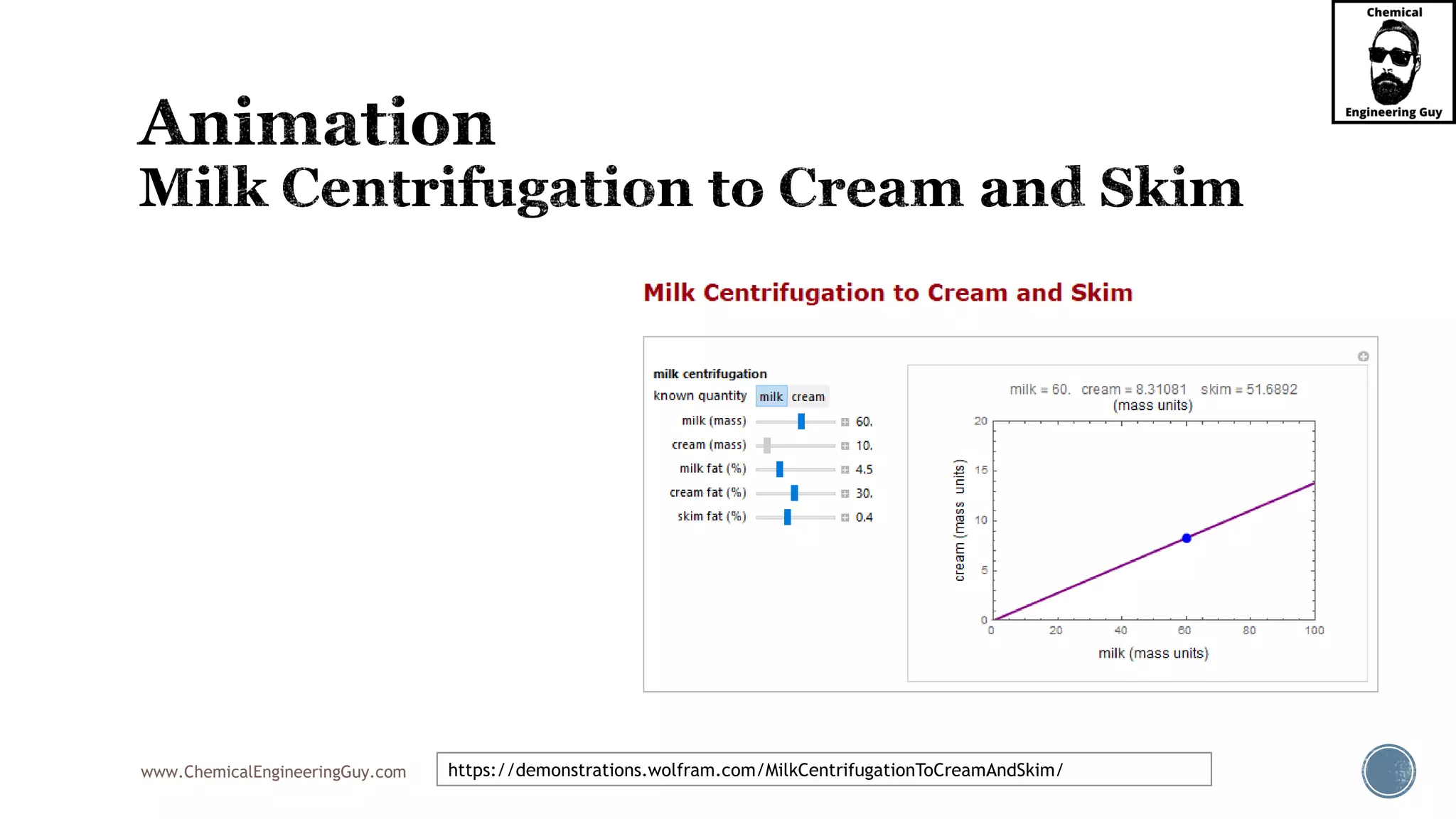
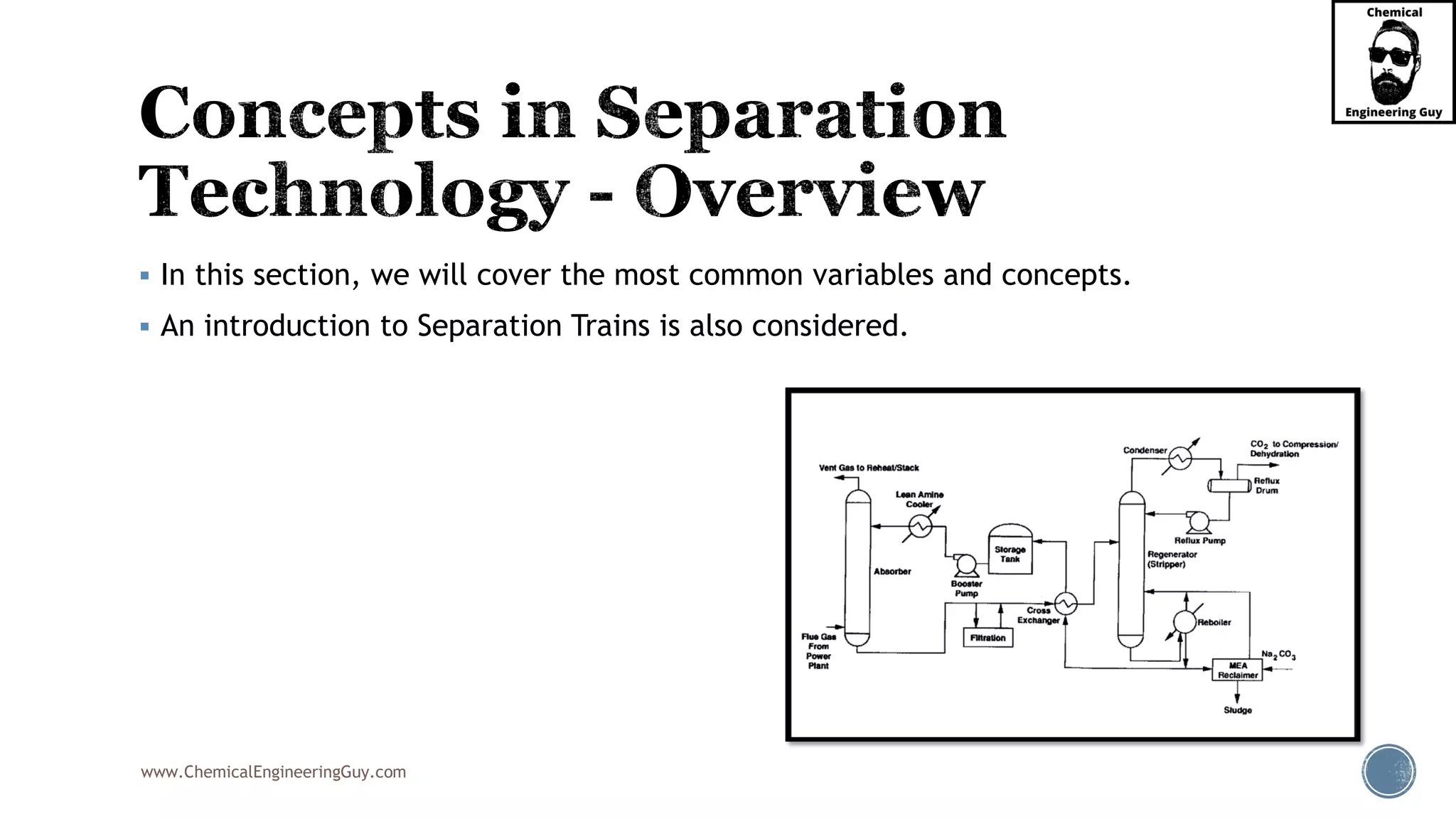
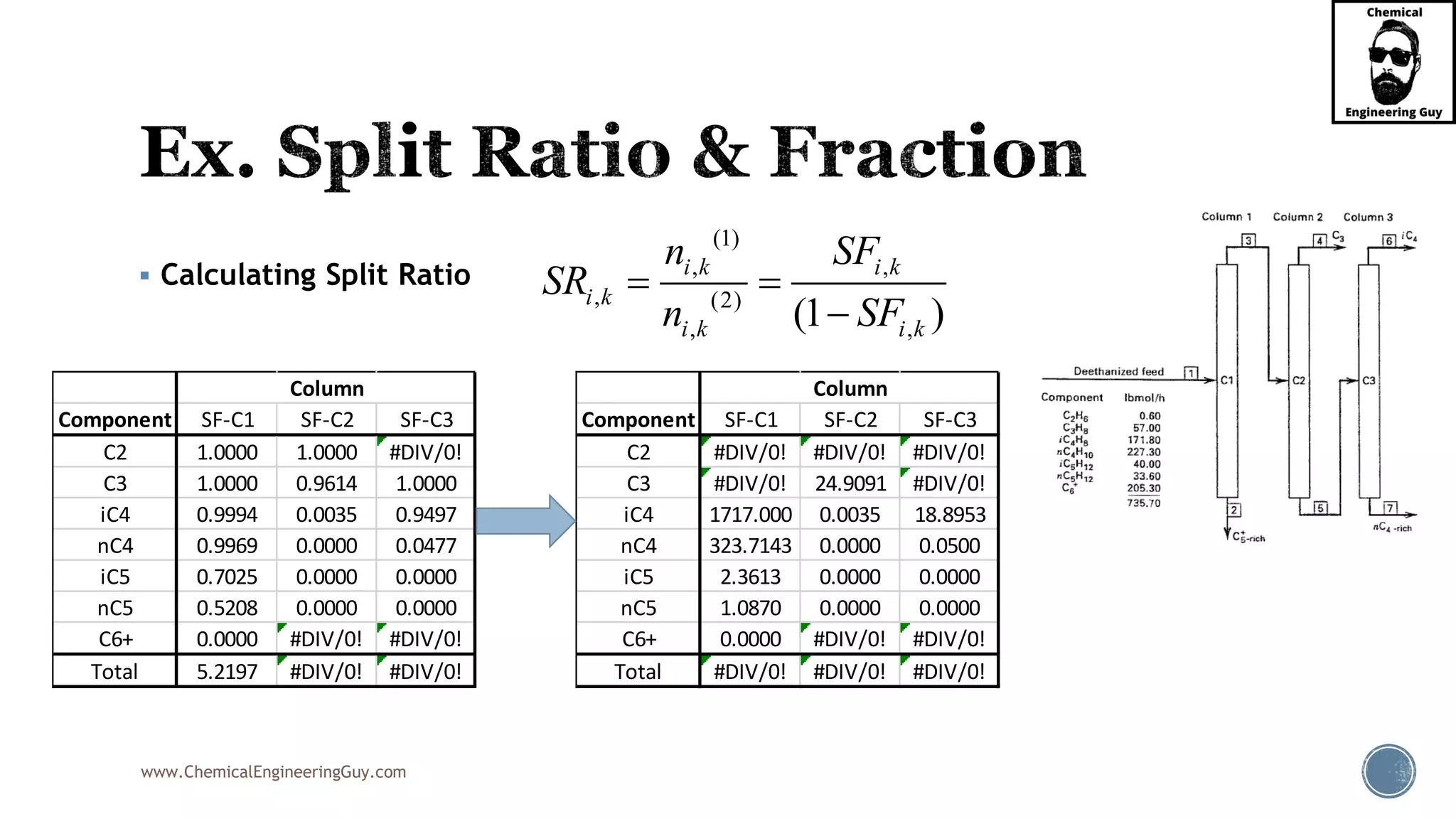
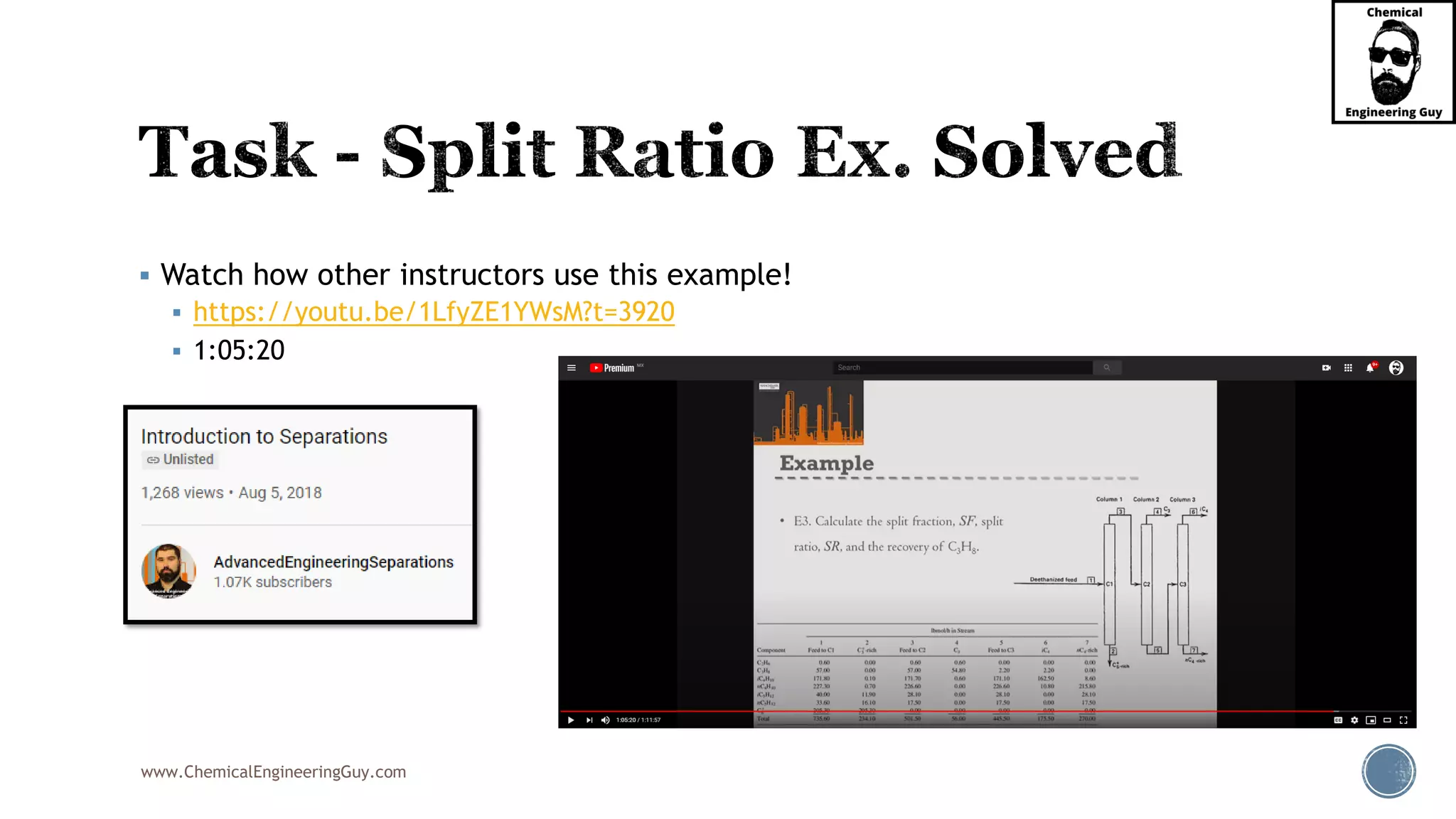
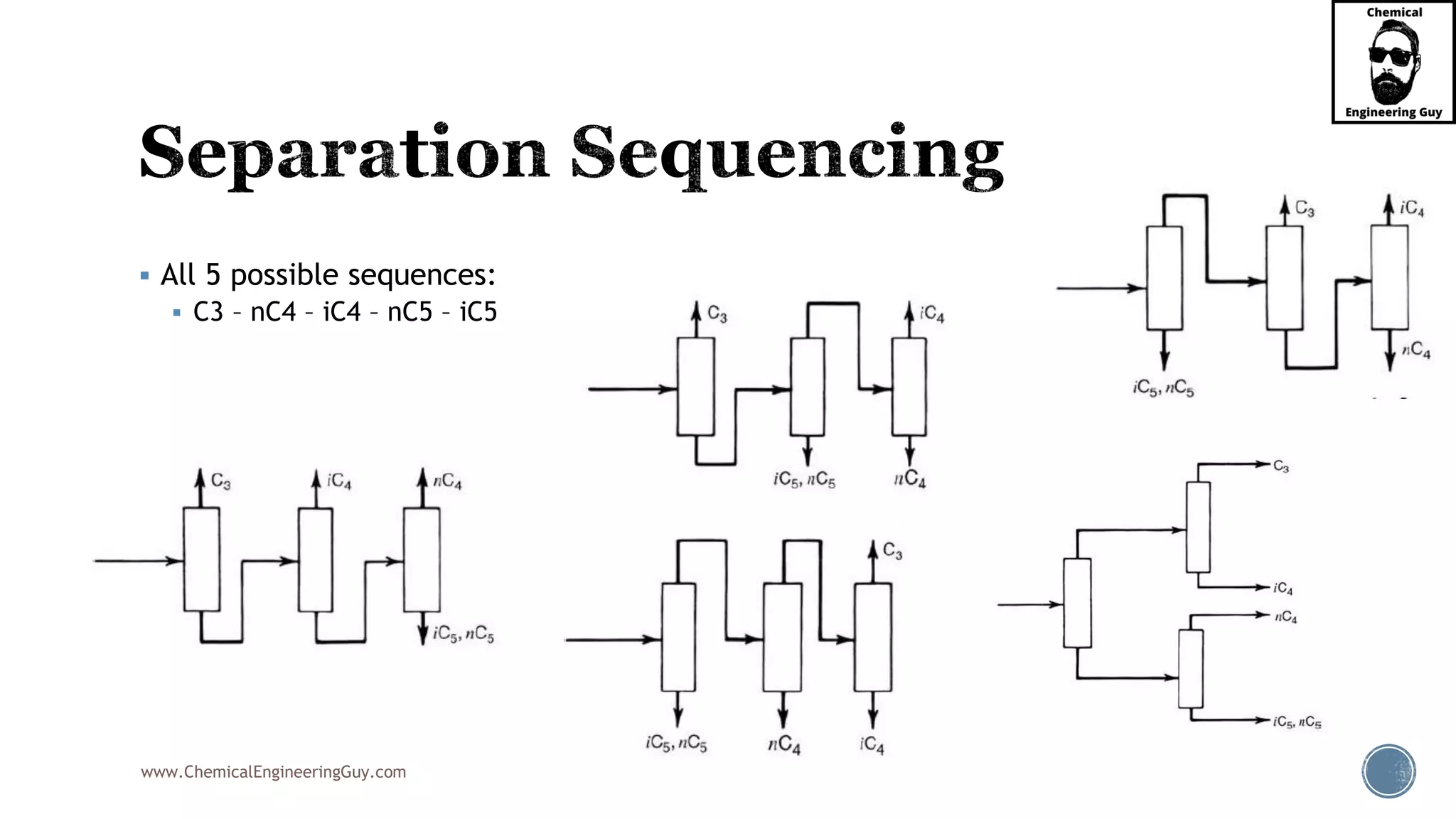
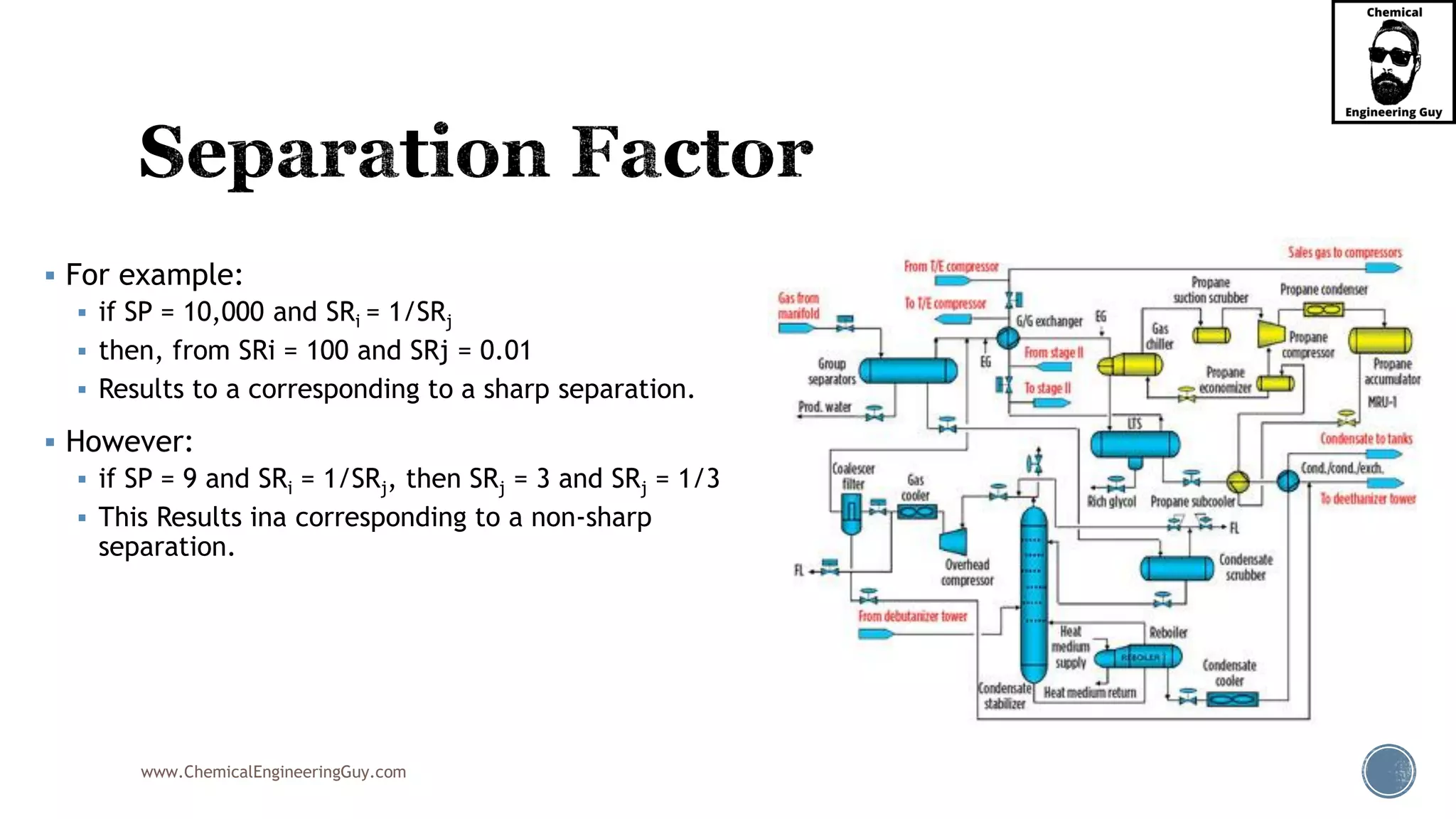
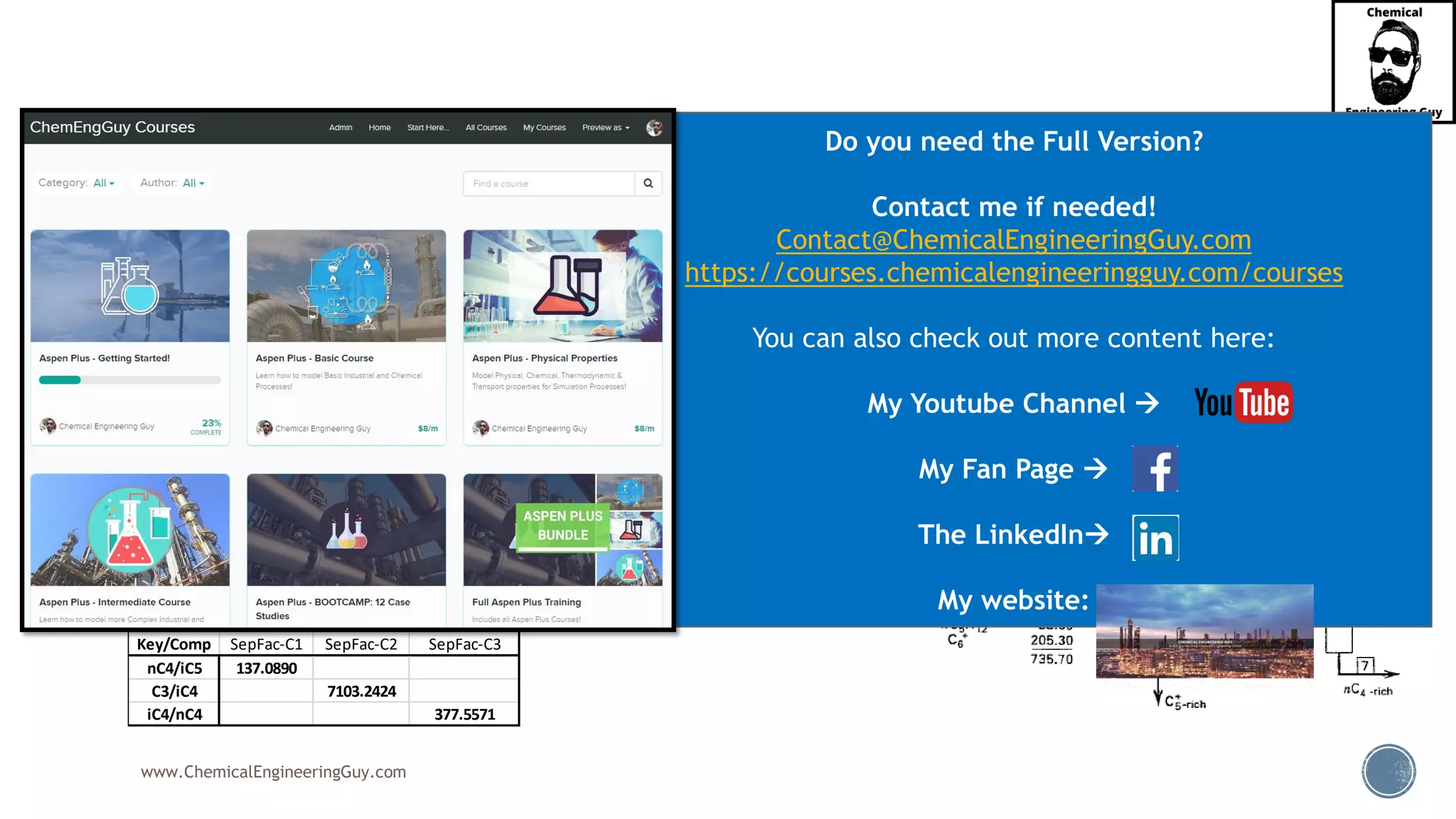
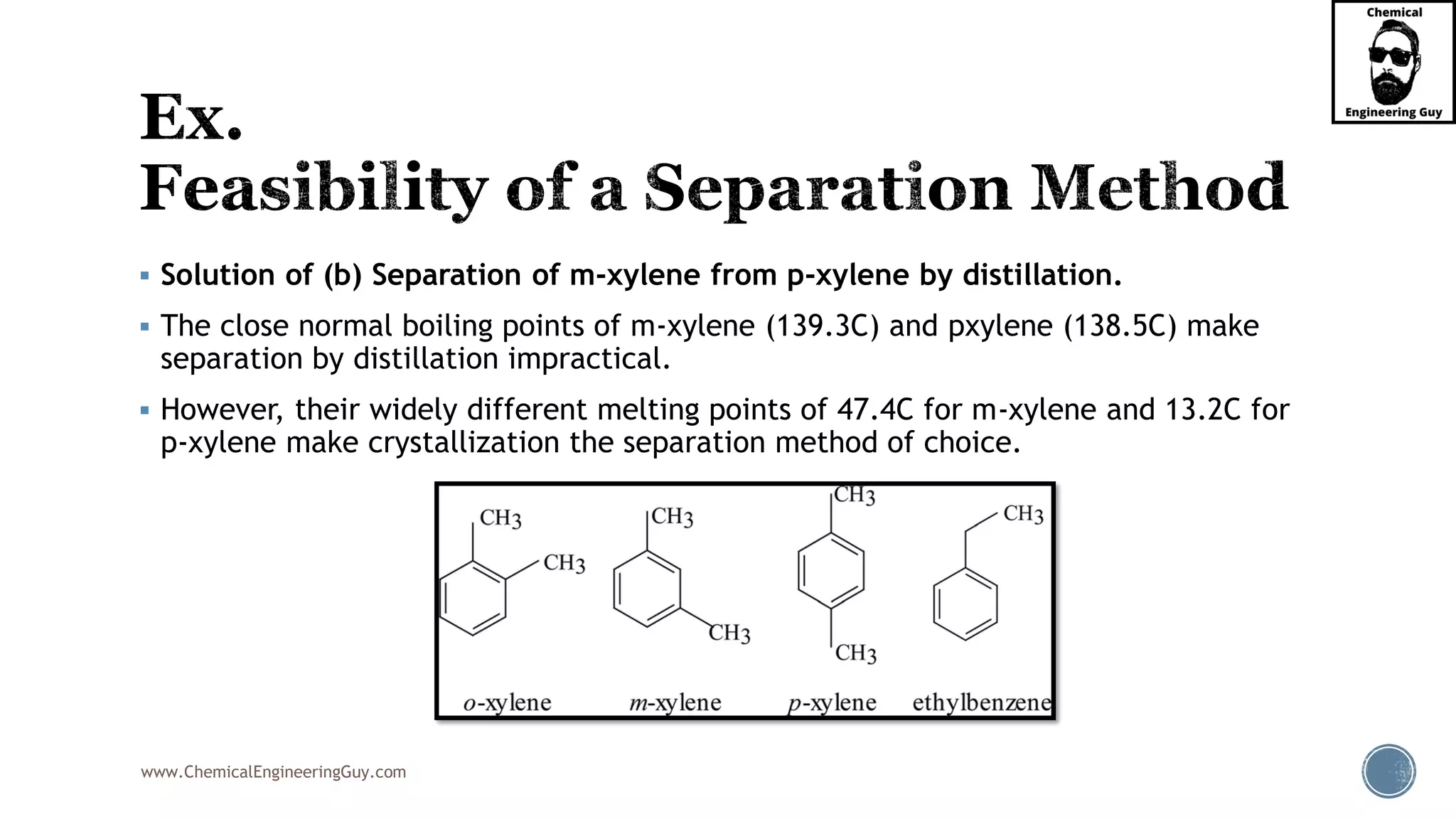
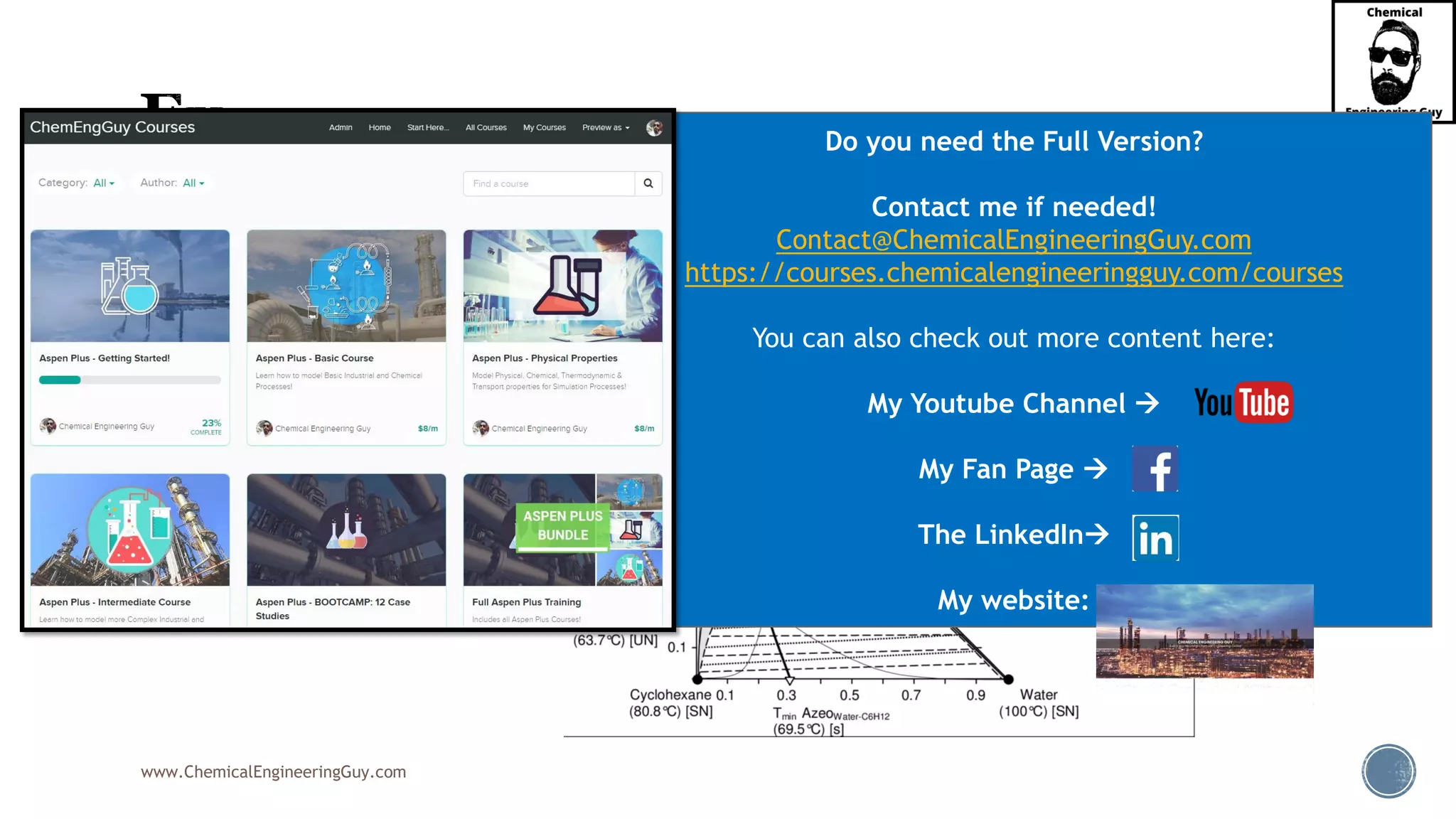
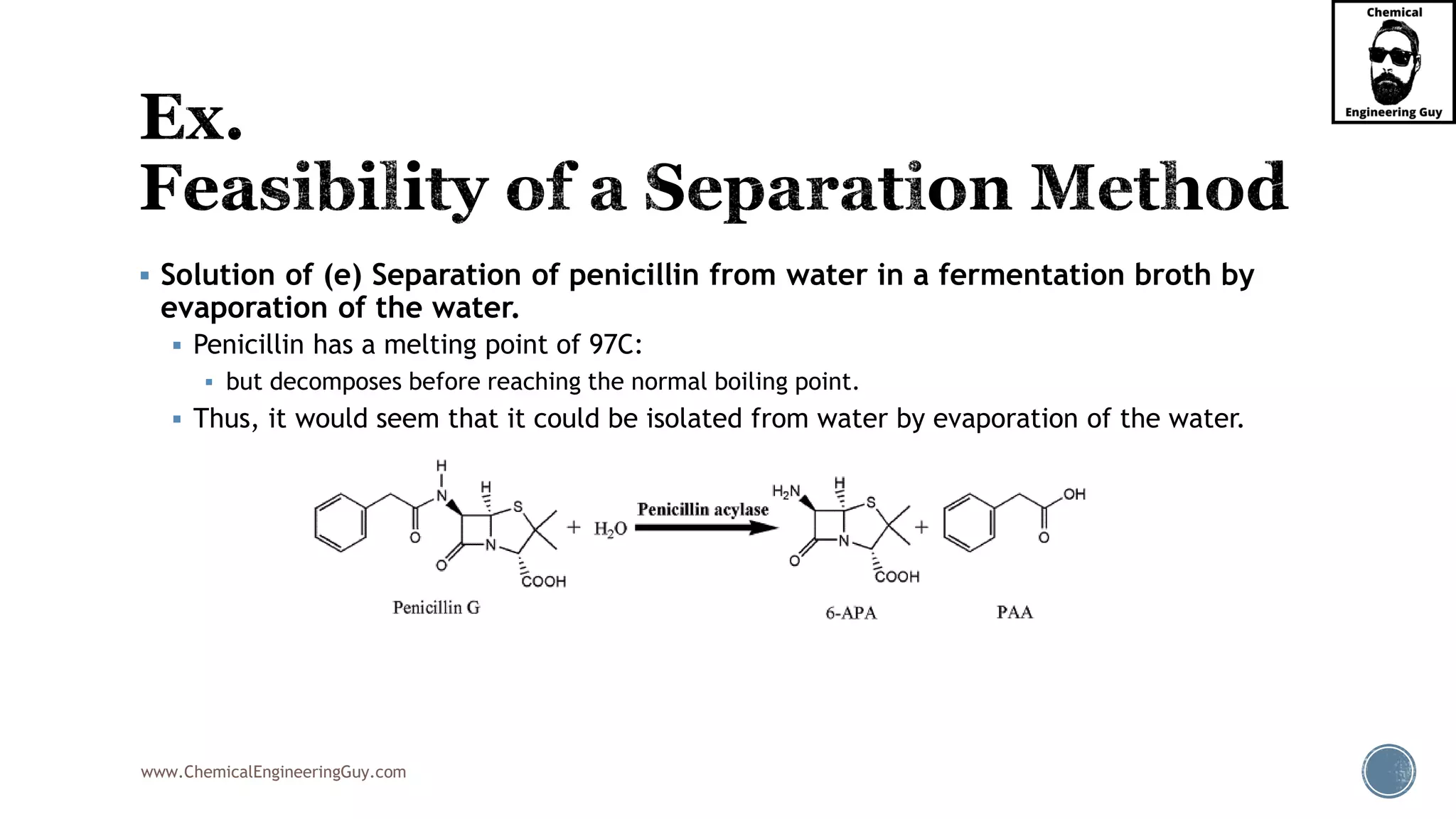
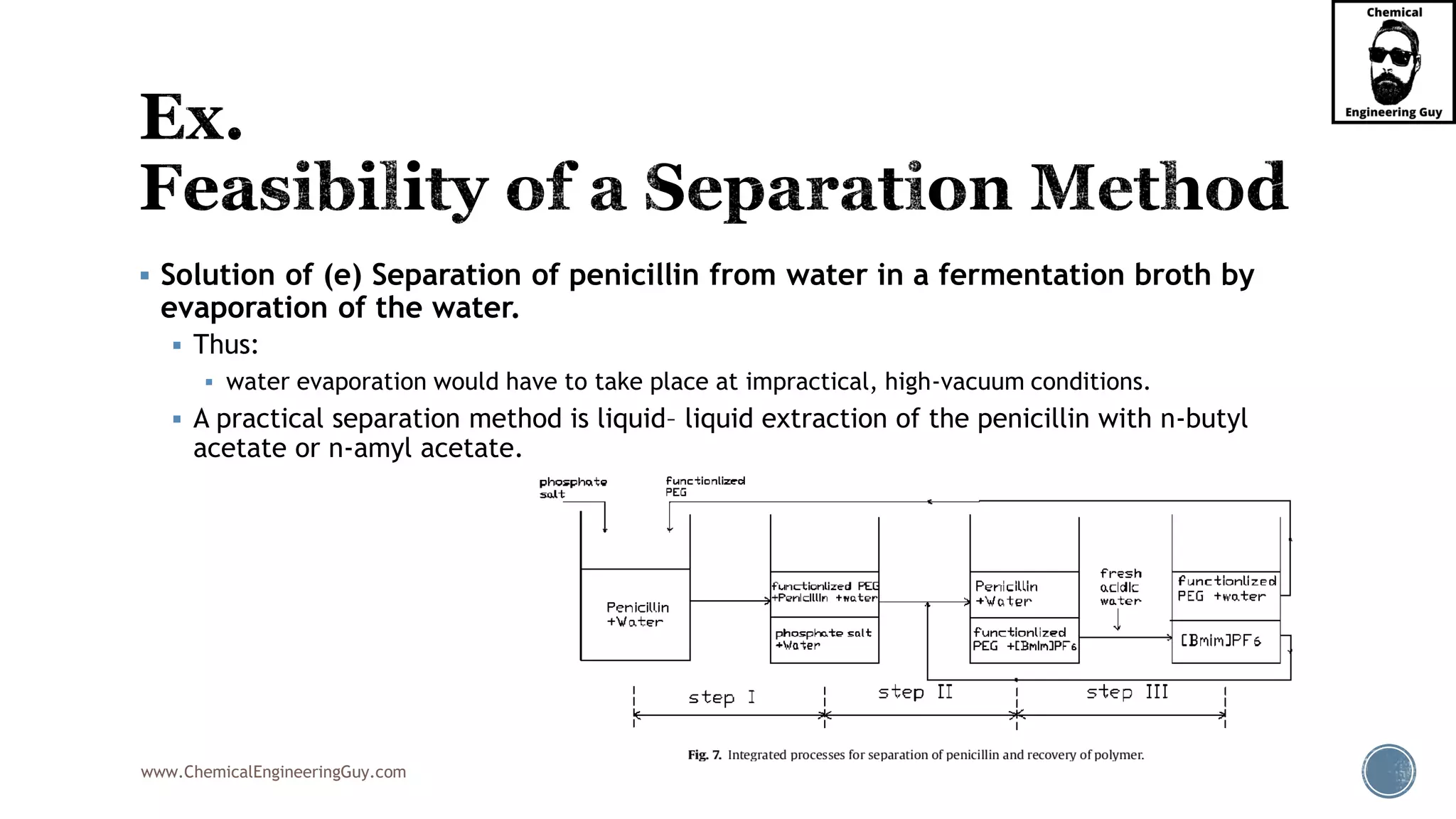
The document outlines various mechanical separation processes such as flotation, flocculation, and centrifugation, along with the equipment commonly used in these processes. It emphasizes the importance of understanding particle sizes and force balances in separation operations, as well as calculating key metrics like split ratios and component recovery. The author provides insights into the principles governing separation methods and stresses the need for effective design in chemical plants to meet specified product purity and recovery targets.