This document summarizes a seminar on the cascade of inflammation. It discusses the signs of inflammation, inflammatory cells and mediators, types of inflammation including acute and chronic, and the mechanisms and cellular events of acute inflammation. Specifically, it outlines the vascular events of acute inflammation including changes in blood flow and vascular permeability, as well as the cellular events of leucocyte exudation and phagocytosis.







![I. VASCULAR EVENTS
Alteration in the microvasculature (arterioles, capillaries and venules) is
the earliest response to tissue injury. These alterations include:
haemodynamic changes and changes in vascular permeability.
I.A] Haemodynamic Changes
The earliest features of inflammatory response result from changes in
the vascular flow and calibre of small blood vessels in the injured
tissue. The sequence of these changes is as under:](https://image.slidesharecdn.com/inflammation-230813185929-ff02826e/85/INFLAMMATION-pptx-10-320.jpg)



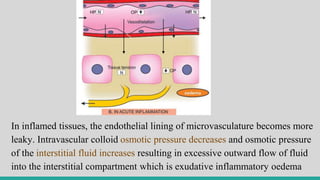
![I.B] Altered Vascular Permeability
Pathogenesis
Normally the fluid balance is maintained
by two opposing sets of forces:
i) Forces that cause outward movement of
fluid from microcirculation: These are
intravascular hydrostatic pressure and
colloid osmotic pressure of interstitial
fluid.
ii) Forces that cause inward movement of
interstitial fluid into circulation: These are
intravascular colloid osmotic pressure and
hydrostatic pressure of interstitial fluid.](https://image.slidesharecdn.com/inflammation-230813185929-ff02826e/85/INFLAMMATION-pptx-14-320.jpg)
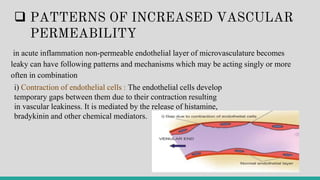
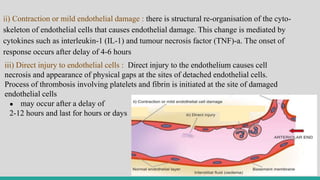
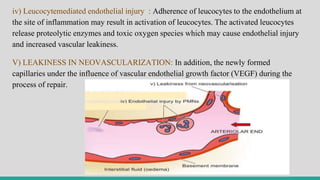
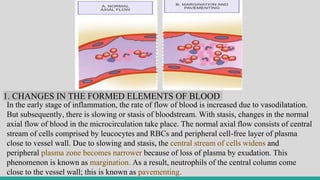
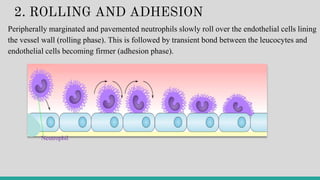
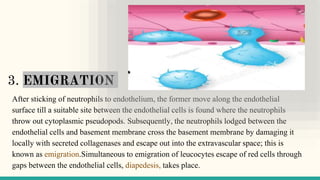
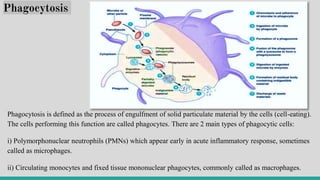
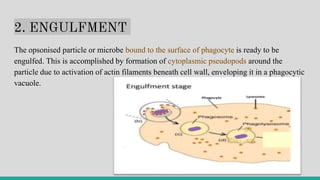
![II. CELLULAR EVENTS
The cellular phase of inflammation consists of 2 processes:
a. exudation of leucocytes; and
b. phagocytosis.
II.a] Exudation of Leucocytes
In acute inflammation, polymorphonuclear neutrophils (PMNs)
comprise the first line of body defense, followed later by monocytes
and macrophages.](https://image.slidesharecdn.com/inflammation-230813185929-ff02826e/85/INFLAMMATION-pptx-19-320.jpg)