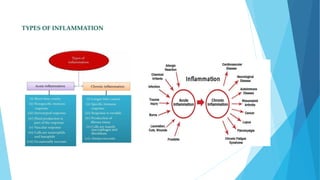
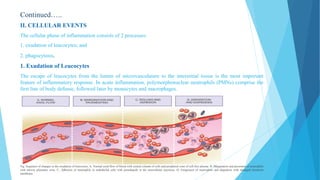
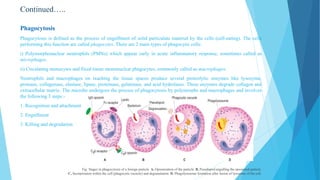
Inflammation is the body's local response to injury, triggered by various agents such as bacteria, toxins, and physical trauma, characterized by the four cardinal signs: redness, swelling, heat, and pain. It can be acute, lasting less than two weeks, or chronic, where inflammation persists. Wound healing involves phases of hemostasis, inflammation, proliferation, and remodeling, regulated by various growth factors and the extracellular matrix.