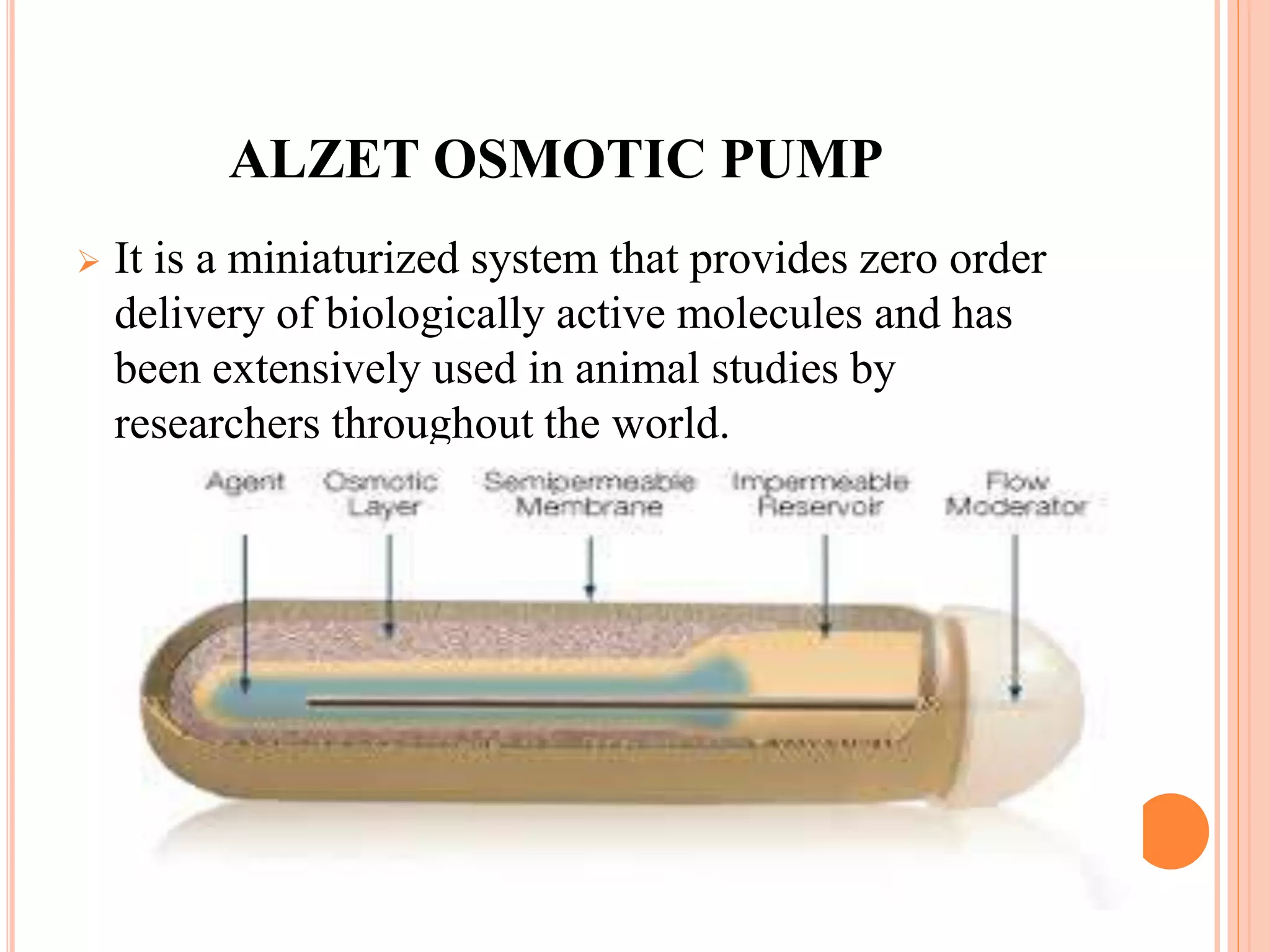
This document discusses implantable drug delivery systems. It describes how implantable pellets or capsules can continuously release drugs over long periods of time to treat conditions without frequent injections or hospital visits. Ideal properties of implants include biocompatibility and controlled drug release. Various types of implants are described, including biodegradable polymer matrices and osmotic pumps, which use osmotic pressure to precisely deliver drugs. Applications include cancer treatment and osteoporosis. Advantages are continuous dosing and patient compliance, while disadvantages include need for minor surgery and inability to easily stop therapy.