Imfa
•Download as PPTX, PDF•
1 like•3,722 views
1. Intermolecular forces of attraction include ionic, dipole-dipole, hydrogen bonding, and dispersion forces. These forces determine physical properties like boiling point. 2. Stronger intermolecular forces require more kinetic energy to overcome, resulting in higher boiling points. Polar substances have stronger intermolecular forces than nonpolar substances. 3. The boiling point of a liquid is reached when the vapor pressure of the escaping molecules equals the pressure of the air above the liquid. Stronger intermolecular forces make it harder for molecules to break away from the liquid, increasing the boiling point.
Report
Share
Report
Share
Recommended
Earth & Life Science Solar System (lesson 2)

The Solar System(Its Origin & Evolution) for Grade 11 Earth & Life Science (Core Subject)
Intermolecular Forces of attraction

Physical Science Grade 11 or 12 ABM Strand K-12 Senior High School (Curriculum Guided)
Earth and Life Science - Earth Subsystems

This is a powerpoint presentation that is about one of the Senior High School Core Subject: Earth and Life Science. It is composed of the theories that explains the Earth and its Subsystems (The Four Spheres).
Recommended
Earth & Life Science Solar System (lesson 2)

The Solar System(Its Origin & Evolution) for Grade 11 Earth & Life Science (Core Subject)
Intermolecular Forces of attraction

Physical Science Grade 11 or 12 ABM Strand K-12 Senior High School (Curriculum Guided)
Earth and Life Science - Earth Subsystems

This is a powerpoint presentation that is about one of the Senior High School Core Subject: Earth and Life Science. It is composed of the theories that explains the Earth and its Subsystems (The Four Spheres).
Synthesis of the New Elements in the Laboratory 

The power point presentation is intended for reporting purposes. Various slides were not defined well and needs to be explained by the reporter during the discussion. The slide started in explaining the objective of the reporting. Explain what a periodic table is. Present the synthetic elements and how they were made. The nuclear reactions presented were only depicted by images and thus, needed to be researched.
Unifying Themes of Life

• THEMES OF LIFE:
o BIOLOGICAL SYSTEMS
o THE CELLULAR BASIS OF LIFE
o STRUCTURE AND FUNCTION
o REPRODUCTION AND INHERITANCE
o ENVIRONMENTAL INTERACTIONS
o ENERGY AND LIFE
o REGULATION
o EVOLUTION AND DIVERSITY
o SCIENTIFIC INQUIRY
o SCIENCE, TECHNOLOGY AND SOCIETY
Introduction to Life Science and The Theories on the Origin of Life

I. Introduction to Life Science
II. The Concept of Life
III. Characteristics of Life
IV. Theories on the Origin of Life
V. Unifying Themes in the Study of Life
General Biology 1 - Lesson 1: Cell (structure,function, and theory)

K-12 Curriculum
Senior High School
General Biology 1
STEM 12
Chapter 2.1 Solutions and Their Properties

1.Types of Solution
2. Energy of Solution
3, Concentration of Solution
Planet Earth and its properties necessary to support life

Department of Education | Senior High School
Topic: Planet Earth and its properties necessary to support life.
Learning Competency:
Earth and Life Science: Recognize the uniqueness of Earth, being the only planet in the Solar System with properties necessary to support life.
Earth Science (for STEM): Describe the characteristics of Earth that are necessary to support life.
Please LIKE / FOLLOW and SHARE my other social media accounts.
Facebook: https://www.facebook.com/Simple-ABbieC-131584525051378/
-----------------------------------------------------------------------
Youtube:
http://tiny.cc/SimpleABbieC
-----------------------------------------------------------------------
Slideshare:
https://www.slideshare.net/AbbieMahinay
-----------------------------------------------------------------------
Blogger:
https://simpleabbiec.blogspot.com/?m=1
Early model of the Universe

The early model of the universe and the theories proposed by ancient philosophers.
Session no. 3.1. energy transformation atp – adp cycle and photosynthesis

atp-adp cycle, energy transformation
Grade 11 Earth & Life Science Lesson 1

Touring the Universe (An Introduction to Formation of the Universe)
I hope this lesson can shed light to SHS Grade 11 Science Teachers. My appeal to those who will download this ppt. please email me at marileahmendina08@gmail.com for my own references. I would be glad to hear from you.
More Related Content
What's hot
Synthesis of the New Elements in the Laboratory 

The power point presentation is intended for reporting purposes. Various slides were not defined well and needs to be explained by the reporter during the discussion. The slide started in explaining the objective of the reporting. Explain what a periodic table is. Present the synthetic elements and how they were made. The nuclear reactions presented were only depicted by images and thus, needed to be researched.
Unifying Themes of Life

• THEMES OF LIFE:
o BIOLOGICAL SYSTEMS
o THE CELLULAR BASIS OF LIFE
o STRUCTURE AND FUNCTION
o REPRODUCTION AND INHERITANCE
o ENVIRONMENTAL INTERACTIONS
o ENERGY AND LIFE
o REGULATION
o EVOLUTION AND DIVERSITY
o SCIENTIFIC INQUIRY
o SCIENCE, TECHNOLOGY AND SOCIETY
Introduction to Life Science and The Theories on the Origin of Life

I. Introduction to Life Science
II. The Concept of Life
III. Characteristics of Life
IV. Theories on the Origin of Life
V. Unifying Themes in the Study of Life
General Biology 1 - Lesson 1: Cell (structure,function, and theory)

K-12 Curriculum
Senior High School
General Biology 1
STEM 12
Chapter 2.1 Solutions and Their Properties

1.Types of Solution
2. Energy of Solution
3, Concentration of Solution
Planet Earth and its properties necessary to support life

Department of Education | Senior High School
Topic: Planet Earth and its properties necessary to support life.
Learning Competency:
Earth and Life Science: Recognize the uniqueness of Earth, being the only planet in the Solar System with properties necessary to support life.
Earth Science (for STEM): Describe the characteristics of Earth that are necessary to support life.
Please LIKE / FOLLOW and SHARE my other social media accounts.
Facebook: https://www.facebook.com/Simple-ABbieC-131584525051378/
-----------------------------------------------------------------------
Youtube:
http://tiny.cc/SimpleABbieC
-----------------------------------------------------------------------
Slideshare:
https://www.slideshare.net/AbbieMahinay
-----------------------------------------------------------------------
Blogger:
https://simpleabbiec.blogspot.com/?m=1
Early model of the Universe

The early model of the universe and the theories proposed by ancient philosophers.
Session no. 3.1. energy transformation atp – adp cycle and photosynthesis

atp-adp cycle, energy transformation
Grade 11 Earth & Life Science Lesson 1

Touring the Universe (An Introduction to Formation of the Universe)
I hope this lesson can shed light to SHS Grade 11 Science Teachers. My appeal to those who will download this ppt. please email me at marileahmendina08@gmail.com for my own references. I would be glad to hear from you.
What's hot (20)
Introduction to Life Science and The Theories on the Origin of Life

Introduction to Life Science and The Theories on the Origin of Life
General Biology 1 - Lesson 1: Cell (structure,function, and theory)

General Biology 1 - Lesson 1: Cell (structure,function, and theory)
Planet Earth and its properties necessary to support life

Planet Earth and its properties necessary to support life
Session no. 3.1. energy transformation atp – adp cycle and photosynthesis

Session no. 3.1. energy transformation atp – adp cycle and photosynthesis
Earth and Life Science - Earth Subsystems Grade 11

Earth and Life Science - Earth Subsystems Grade 11
Similar to Imfa
Lecture 17b- Water Curve Calcs

The second lecture for chapter 17 (honors and prep chemistry) covers the water curve, specific heat, and the enthalpy of phase changes.
Latent heat

When energy is absorbed as heat by a solid or liquid, the temperature of the object does not necessarily rise.
The thermal energy may cause the mass to change from one phase, or state, to another.
The amount of energy per unit mass that must be transferred as heat when a mass undergoes a phase change is called the heat of transformation, L.
Properties of Solids & Liquids

Discusses macroscopic and microscopic properties of solids and liquids.
**More good stuff available at:
www.wsautter.com
and
http://www.youtube.com/results?search_query=wnsautter&aq=f
Similar to Imfa (20)
More from Kristel Manuyag
More from Kristel Manuyag (17)
Recently uploaded
Generative AI Deep Dive: Advancing from Proof of Concept to Production

Join Maher Hanafi, VP of Engineering at Betterworks, in this new session where he'll share a practical framework to transform Gen AI prototypes into impactful products! He'll delve into the complexities of data collection and management, model selection and optimization, and ensuring security, scalability, and responsible use.
Why You Should Replace Windows 11 with Nitrux Linux 3.5.0 for enhanced perfor...

The choice of an operating system plays a pivotal role in shaping our computing experience. For decades, Microsoft's Windows has dominated the market, offering a familiar and widely adopted platform for personal and professional use. However, as technological advancements continue to push the boundaries of innovation, alternative operating systems have emerged, challenging the status quo and offering users a fresh perspective on computing.
One such alternative that has garnered significant attention and acclaim is Nitrux Linux 3.5.0, a sleek, powerful, and user-friendly Linux distribution that promises to redefine the way we interact with our devices. With its focus on performance, security, and customization, Nitrux Linux presents a compelling case for those seeking to break free from the constraints of proprietary software and embrace the freedom and flexibility of open-source computing.
Alt. GDG Cloud Southlake #33: Boule & Rebala: Effective AppSec in SDLC using ...

Effective Application Security in Software Delivery lifecycle using Deployment Firewall and DBOM
The modern software delivery process (or the CI/CD process) includes many tools, distributed teams, open-source code, and cloud platforms. Constant focus on speed to release software to market, along with the traditional slow and manual security checks has caused gaps in continuous security as an important piece in the software supply chain. Today organizations feel more susceptible to external and internal cyber threats due to the vast attack surface in their applications supply chain and the lack of end-to-end governance and risk management.
The software team must secure its software delivery process to avoid vulnerability and security breaches. This needs to be achieved with existing tool chains and without extensive rework of the delivery processes. This talk will present strategies and techniques for providing visibility into the true risk of the existing vulnerabilities, preventing the introduction of security issues in the software, resolving vulnerabilities in production environments quickly, and capturing the deployment bill of materials (DBOM).
Speakers:
Bob Boule
Robert Boule is a technology enthusiast with PASSION for technology and making things work along with a knack for helping others understand how things work. He comes with around 20 years of solution engineering experience in application security, software continuous delivery, and SaaS platforms. He is known for his dynamic presentations in CI/CD and application security integrated in software delivery lifecycle.
Gopinath Rebala
Gopinath Rebala is the CTO of OpsMx, where he has overall responsibility for the machine learning and data processing architectures for Secure Software Delivery. Gopi also has a strong connection with our customers, leading design and architecture for strategic implementations. Gopi is a frequent speaker and well-known leader in continuous delivery and integrating security into software delivery.
A tale of scale & speed: How the US Navy is enabling software delivery from l...

Rapid and secure feature delivery is a goal across every application team and every branch of the DoD. The Navy’s DevSecOps platform, Party Barge, has achieved:
- Reduction in onboarding time from 5 weeks to 1 day
- Improved developer experience and productivity through actionable findings and reduction of false positives
- Maintenance of superior security standards and inherent policy enforcement with Authorization to Operate (ATO)
Development teams can ship efficiently and ensure applications are cyber ready for Navy Authorizing Officials (AOs). In this webinar, Sigma Defense and Anchore will give attendees a look behind the scenes and demo secure pipeline automation and security artifacts that speed up application ATO and time to production.
We will cover:
- How to remove silos in DevSecOps
- How to build efficient development pipeline roles and component templates
- How to deliver security artifacts that matter for ATO’s (SBOMs, vulnerability reports, and policy evidence)
- How to streamline operations with automated policy checks on container images
LF Energy Webinar: Electrical Grid Modelling and Simulation Through PowSyBl -...

Do you want to learn how to model and simulate an electrical network from scratch in under an hour?
Then welcome to this PowSyBl workshop, hosted by Rte, the French Transmission System Operator (TSO)!
During the webinar, you will discover the PowSyBl ecosystem as well as handle and study an electrical network through an interactive Python notebook.
PowSyBl is an open source project hosted by LF Energy, which offers a comprehensive set of features for electrical grid modelling and simulation. Among other advanced features, PowSyBl provides:
- A fully editable and extendable library for grid component modelling;
- Visualization tools to display your network;
- Grid simulation tools, such as power flows, security analyses (with or without remedial actions) and sensitivity analyses;
The framework is mostly written in Java, with a Python binding so that Python developers can access PowSyBl functionalities as well.
What you will learn during the webinar:
- For beginners: discover PowSyBl's functionalities through a quick general presentation and the notebook, without needing any expert coding skills;
- For advanced developers: master the skills to efficiently apply PowSyBl functionalities to your real-world scenarios.
GDG Cloud Southlake #33: Boule & Rebala: Effective AppSec in SDLC using Deplo...

Effective Application Security in Software Delivery lifecycle using Deployment Firewall and DBOM
The modern software delivery process (or the CI/CD process) includes many tools, distributed teams, open-source code, and cloud platforms. Constant focus on speed to release software to market, along with the traditional slow and manual security checks has caused gaps in continuous security as an important piece in the software supply chain. Today organizations feel more susceptible to external and internal cyber threats due to the vast attack surface in their applications supply chain and the lack of end-to-end governance and risk management.
The software team must secure its software delivery process to avoid vulnerability and security breaches. This needs to be achieved with existing tool chains and without extensive rework of the delivery processes. This talk will present strategies and techniques for providing visibility into the true risk of the existing vulnerabilities, preventing the introduction of security issues in the software, resolving vulnerabilities in production environments quickly, and capturing the deployment bill of materials (DBOM).
Speakers:
Bob Boule
Robert Boule is a technology enthusiast with PASSION for technology and making things work along with a knack for helping others understand how things work. He comes with around 20 years of solution engineering experience in application security, software continuous delivery, and SaaS platforms. He is known for his dynamic presentations in CI/CD and application security integrated in software delivery lifecycle.
Gopinath Rebala
Gopinath Rebala is the CTO of OpsMx, where he has overall responsibility for the machine learning and data processing architectures for Secure Software Delivery. Gopi also has a strong connection with our customers, leading design and architecture for strategic implementations. Gopi is a frequent speaker and well-known leader in continuous delivery and integrating security into software delivery.
Smart TV Buyer Insights Survey 2024 by 91mobiles.pdf

91mobiles recently conducted a Smart TV Buyer Insights Survey in which we asked over 3,000 respondents about the TV they own, aspects they look at on a new TV, and their TV buying preferences.
GraphSummit Singapore | The Future of Agility: Supercharging Digital Transfor...

Leonard Jayamohan, Partner & Generative AI Lead, Deloitte
This keynote will reveal how Deloitte leverages Neo4j’s graph power for groundbreaking digital twin solutions, achieving a staggering 100x performance boost. Discover the essential role knowledge graphs play in successful generative AI implementations. Plus, get an exclusive look at an innovative Neo4j + Generative AI solution Deloitte is developing in-house.
GraphRAG is All You need? LLM & Knowledge Graph

Guy Korland, CEO and Co-founder of FalkorDB, will review two articles on the integration of language models with knowledge graphs.
1. Unifying Large Language Models and Knowledge Graphs: A Roadmap.
https://arxiv.org/abs/2306.08302
2. Microsoft Research's GraphRAG paper and a review paper on various uses of knowledge graphs:
https://www.microsoft.com/en-us/research/blog/graphrag-unlocking-llm-discovery-on-narrative-private-data/
Essentials of Automations: The Art of Triggers and Actions in FME

In this second installment of our Essentials of Automations webinar series, we’ll explore the landscape of triggers and actions, guiding you through the nuances of authoring and adapting workspaces for seamless automations. Gain an understanding of the full spectrum of triggers and actions available in FME, empowering you to enhance your workspaces for efficient automation.
We’ll kick things off by showcasing the most commonly used event-based triggers, introducing you to various automation workflows like manual triggers, schedules, directory watchers, and more. Plus, see how these elements play out in real scenarios.
Whether you’re tweaking your current setup or building from the ground up, this session will arm you with the tools and insights needed to transform your FME usage into a powerhouse of productivity. Join us to discover effective strategies that simplify complex processes, enhancing your productivity and transforming your data management practices with FME. Let’s turn complexity into clarity and make your workspaces work wonders!
Securing your Kubernetes cluster_ a step-by-step guide to success !

Today, after several years of existence, an extremely active community and an ultra-dynamic ecosystem, Kubernetes has established itself as the de facto standard in container orchestration. Thanks to a wide range of managed services, it has never been so easy to set up a ready-to-use Kubernetes cluster.
However, this ease of use means that the subject of security in Kubernetes is often left for later, or even neglected. This exposes companies to significant risks.
In this talk, I'll show you step-by-step how to secure your Kubernetes cluster for greater peace of mind and reliability.
SAP Sapphire 2024 - ASUG301 building better apps with SAP Fiori.pdf

Building better applications for business users with SAP Fiori.
• What is SAP Fiori and why it matters to you
• How a better user experience drives measurable business benefits
• How to get started with SAP Fiori today
• How SAP Fiori elements accelerates application development
• How SAP Build Code includes SAP Fiori tools and other generative artificial intelligence capabilities
• How SAP Fiori paves the way for using AI in SAP apps
GraphSummit Singapore | The Art of the Possible with Graph - Q2 2024

Neha Bajwa, Vice President of Product Marketing, Neo4j
Join us as we explore breakthrough innovations enabled by interconnected data and AI. Discover firsthand how organizations use relationships in data to uncover contextual insights and solve our most pressing challenges – from optimizing supply chains, detecting fraud, and improving customer experiences to accelerating drug discoveries.
GraphSummit Singapore | Enhancing Changi Airport Group's Passenger Experience...

Dr. Sean Tan, Head of Data Science, Changi Airport Group
Discover how Changi Airport Group (CAG) leverages graph technologies and generative AI to revolutionize their search capabilities. This session delves into the unique search needs of CAG’s diverse passengers and customers, showcasing how graph data structures enhance the accuracy and relevance of AI-generated search results, mitigating the risk of “hallucinations” and improving the overall customer journey.
Transcript: Selling digital books in 2024: Insights from industry leaders - T...

The publishing industry has been selling digital audiobooks and ebooks for over a decade and has found its groove. What’s changed? What has stayed the same? Where do we go from here? Join a group of leading sales peers from across the industry for a conversation about the lessons learned since the popularization of digital books, best practices, digital book supply chain management, and more.
Link to video recording: https://bnctechforum.ca/sessions/selling-digital-books-in-2024-insights-from-industry-leaders/
Presented by BookNet Canada on May 28, 2024, with support from the Department of Canadian Heritage.
National Security Agency - NSA mobile device best practices

Threats to mobile devices are more prevalent and increasing in scope and complexity. Users of mobile devices desire to take full advantage of the features
available on those devices, but many of the features provide convenience and capability but sacrifice security. This best practices guide outlines steps the users can take to better protect personal devices and information.
Elizabeth Buie - Older adults: Are we really designing for our future selves?

Elizabeth Buie - Older adults: Are we really designing for our future selves?
Recently uploaded (20)
Generative AI Deep Dive: Advancing from Proof of Concept to Production

Generative AI Deep Dive: Advancing from Proof of Concept to Production
Why You Should Replace Windows 11 with Nitrux Linux 3.5.0 for enhanced perfor...

Why You Should Replace Windows 11 with Nitrux Linux 3.5.0 for enhanced perfor...
Alt. GDG Cloud Southlake #33: Boule & Rebala: Effective AppSec in SDLC using ...

Alt. GDG Cloud Southlake #33: Boule & Rebala: Effective AppSec in SDLC using ...
A tale of scale & speed: How the US Navy is enabling software delivery from l...

A tale of scale & speed: How the US Navy is enabling software delivery from l...
LF Energy Webinar: Electrical Grid Modelling and Simulation Through PowSyBl -...

LF Energy Webinar: Electrical Grid Modelling and Simulation Through PowSyBl -...
GDG Cloud Southlake #33: Boule & Rebala: Effective AppSec in SDLC using Deplo...

GDG Cloud Southlake #33: Boule & Rebala: Effective AppSec in SDLC using Deplo...
Smart TV Buyer Insights Survey 2024 by 91mobiles.pdf

Smart TV Buyer Insights Survey 2024 by 91mobiles.pdf
GraphSummit Singapore | The Future of Agility: Supercharging Digital Transfor...

GraphSummit Singapore | The Future of Agility: Supercharging Digital Transfor...
Essentials of Automations: The Art of Triggers and Actions in FME

Essentials of Automations: The Art of Triggers and Actions in FME
Securing your Kubernetes cluster_ a step-by-step guide to success !

Securing your Kubernetes cluster_ a step-by-step guide to success !
SAP Sapphire 2024 - ASUG301 building better apps with SAP Fiori.pdf

SAP Sapphire 2024 - ASUG301 building better apps with SAP Fiori.pdf
GraphSummit Singapore | The Art of the Possible with Graph - Q2 2024

GraphSummit Singapore | The Art of the Possible with Graph - Q2 2024
GraphSummit Singapore | Enhancing Changi Airport Group's Passenger Experience...

GraphSummit Singapore | Enhancing Changi Airport Group's Passenger Experience...
Transcript: Selling digital books in 2024: Insights from industry leaders - T...

Transcript: Selling digital books in 2024: Insights from industry leaders - T...
National Security Agency - NSA mobile device best practices

National Security Agency - NSA mobile device best practices
FIDO Alliance Osaka Seminar: Passkeys and the Road Ahead.pdf

FIDO Alliance Osaka Seminar: Passkeys and the Road Ahead.pdf
Elizabeth Buie - Older adults: Are we really designing for our future selves?

Elizabeth Buie - Older adults: Are we really designing for our future selves?
Imfa
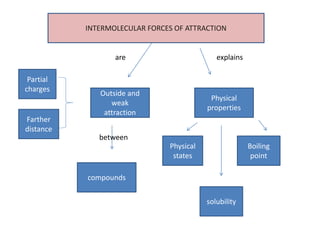
- 1. INTERMOLECULAR FORCES OF ATTRACTION Outside and weak attraction Physical properties Physical states Boiling point compounds Partial charges Farther distance solubility are between explains
- 2. KIND OF IMFA COMPOUNDS THAT FORMED THEM electrostatic Ionic & ionic Ion dipole Ionic & polar H bond Polar H terminal & lone pair of small Electronegative atom Dipole dipole Polar & polar Ion induced dipole Ionic & non polar Dipole induced diipole Polar & non polar dispersion Non polar & non polar Decreasing strenght
- 3. electrostatic Ionic with ionic NaCl NaCl electrostatic
- 5. Ion dipole Ion with polar Na + H2O iondipole
- 6. H bond Polar H terminal with lone pairs of small electronegative atoms H2O H2O H-bond
- 7. Dipole-dipole Polar with polar CH3 C = O CH3 CH3 CH3 C=O dipole-dipole
- 8. Ion-induced dipole Ion with non polar O = O Na + ion induced dipole O = O Cl - e'movement e'movement
- 9. Dipole induced diploe Dipole with non polar O = O H2O dipoleinduceddipole
- 10. Dispersion Non polar with non polar O = O O = O dispersion
- 11. IMFA and Solubility 1. Dissolved substance (solute) must separate (IMFA breaking- endothermic heat) 2. Dissolving substance (solvent) must separate (IMFA breaking- endothermic heat) 3. Solute and solvent must mix (IMFA forming- exothermic heat) For dissolving to happen, 3 processes must occur
- 12. IMFA between solute and solvent > IMFA among solute and or IMFA among solvent _ • Summarized as : LIKE DISSOLVES LIKE ; • polar solvent dissolves polar solute • Non polar solvent dissolves non polar solute
- 13. IMFA and Physical State
- 14. PHYSICAL STATE IMFA KINETIC ENERGY MOLECULES TOGETHER MOLECULES APART Interplay between draws separates
- 15. Kinds Solid IMFA >>> KE Gas IMFA <<< KE liquid IMFA is continuously being formed and continuously being broken
- 16. PHYSICAL STATES Solid Gas Liquid IMFA >>> KE KE >>> IMFA IMFA is continously being formed And continously being broken
- 18. MELTING FREEZINGEVAPORATION CONDENSATION S to L L to SG to LL to G Temp > IMFA Temp >>> IMFA Temp < IMFA Temp <<<IMFA ENDO EXO Sorrounding cold Sorrounding Warm IS IS IS IS WHENWHEN WHEN WHEN makes makes is is
- 19. ENERGY CURVE DURING PHASE CHANGE (OF WATER)
- 20. TE MP E NE R GY -4 0 s l l - g s - l g 80 cal/g 540 cal/g 100 25 Heat of Fusion- amount of energy needed to melt 1 gram of a substance at its melting point H fusion water = 80 cal/g Q= mass X H fusion Heat of Vaporization – amount of energy needed to evaporate 1 gram of a substance at its boiling point H vap water = 540 cal/g Q = mass X H vap Heat of Freezing = energy released to change 1 gram of liquid to solid Heat of Condensation= energy released to convert 1 gram of gas to liquid SPECIFIC HEATS Energy involved to change the temperature of 1 gram of a substance, 10Celsius
- 21. LATENT HEATS • Heat of Fusion- amount of energy needed to melt 1 gram of a substance at its melting point • H fusion water = 80 cal/g • Q= mass X H fusion
- 22. LATENT HEATS • Heat of Freezing = energy released to change 1 gram of liquid to solid • • Heat of Fusion (endo) = Heat of Freezing (exo)
- 23. LATENT HEATS • Heat of Vaporization – amount of energy needed to evaporate 1 gram of a substance at its boiling point • H vap water = 540 cal/g • Q = mass X H vap
- 24. LATENT HEATS • Heat of Condensation= energy released to convert 1 gram of gas to liquid • • Heat of Vaporization (endo) = Heat of Condensation(exo)
- 25. SPECIFIC HEATS • Energy involved to change the temperature of 1 gram of a substance, 10Celsius • For water: Sp. Heat = 1 cal/g-0C
- 26. UNIQUE PROPERTIES OF LIQUIDS 1. Surface Tension 2. Capillarity 3. Viscosity •
- 27. IMFA & Boiling Point Because of the KE energy of evaporating liquids, they break their IMFA and result in some of them going into the gaseous state and exert a pressure called vapor pressure. Under a given temperature each liquid has its own vapor pressure. When the vapor pressure of escaping liquid molecules become equal to the pressure of the air above it. The temperature at that point is called the boiling point of the liquid. The stronger the IMFA, the harder to break, the longer time the liquid to evaporate the higher the boiling point Vapor pressure Air pressure • Vapor pressure = air pressure : Boiling point
- 28. IMFA & Boiling Point • IMFA increases, boiling point increases • MWt increases, IMFA increases, boiling point increases • Branching increases, IMFA decreases, boiling point decreases
