IB Chemistry on Structural Isomers and Benzene Structure
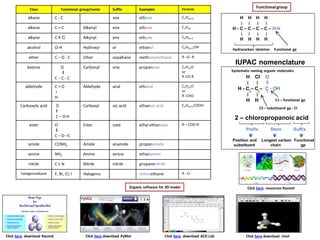
- 1. Class Functional group/name Suffix Examples Formula alkane C - C ane ethane CnH2n+2 alkene C = C Alkenyl ene ethene CnH2n alkyne C ≡ C Alkynyl yne ethyne CnH2n-2 alcohol O-H Hydroxyl ol ethanol CnH2n+1OH ether C – O - C Ether oxyalkane methoxymethane R –O -R ketone O ‖ C – C - C Carbonyl one propanone CnH2nO or R-CO-R aldehyde C = O ׀ H Aldehyde anal ethanal CnH2nO or R -CHO Carboxylic acid O ‖ C – O H Carboxyl oic acid ethanoic acid CnH2n+1COOH ester O ‖ C – O –C Ester oate ethyl ethanoate R – COO-R amide CONH2 Amide anamide propanamide amine NH2 Amine amine ethanamine nitrile C ≡ N Nitrile nitrile propanenitrile halogenoalkane F, Br, CI, I Halogeno chloroethane R - CI Functionalgroup H H H H ׀ ׀ ׀ ׀ H - C – C – C – C – O-H ׀ ׀ ׀ ׀ H H H H Hydrocarbon skeleton Functional gp IUPAC nomenclature Systematic naming organic molecules H CI O ׀ ׀ ‖ H - C – C – C - OH ׀ ׀ H H C1 – functional gp C2 – substituent gp - CI 2 – chloropropanoic acid Prefix Stem Suffix Position and substituent Longest carbon chain Functional gp Organic software for 3D model Click here resources Rasmol Click here download Rasmol Click here download PyMol Click here download ACD Lab Click here download Jmol 3 2 1
- 2. H CH3 H H H ׀ ׀ ׀ ׀ ׀ H - C - C – C – C – C – H ׀ ׀ ׀ ׀ ׀ H H H H H H H H H ׀ ׀ ׀ ׀ H - C – C – C – C – O-H ׀ ׀ ׀ ׀ H H H H Hydrocarbon skeleton Functional gp Hydrocarbon Aliphatic Aromatic Saturated Unsaturated benzene alkylbenzene H H ׀ ׀ H - C – C – H ׀ ׀ H H H H ׀ ׀ C = C ׀ ׀ H H Alkane Cycloalkane Compound Ethane Ethanoic acid Empirical formula CH3 CH2O Molecular formula C2H6 C2H4O2 Full SF Condensed SF CH3CH3 CH3COOH Stereochemical formula (3D) H H ׀ ׀ H - C – C – H ׀ ׀ H H H O ׀ ‖ H - C - C - OH ׀ H Organic Chemistry Cycloalkene Alkene IUPAC nomenclature Ring form Functionalgroup Structural formula 2 – chloropropanoic acid no aromatic ring Systematic naming organic molecules Prefix Stem Suffix Position and substituent Longest carbon chain Functional gp H CI O ׀ ׀ ‖ H - C – C – C - OH ׀ ׀ H H C1 – functional gp C2 – substituent gp - CI CH3(CH2)3CH3 H H H H H ׀ ׀ ׀ ׀ ׀ H - C - C – C – C – C – H ׀ ׀ ׀ ׀ ׀ H H H H H Use of parenthesis Repeat CH2 x 3 CH3CH(CH3)(CH2)2CH3 Repeat CH2 x 2CH3 branch H CH3 H CH3 H ׀ ׀ ׀ ׀ ׀ H - C - C – C – C – C – H ׀ ׀ ׀ ׀ ׀ H H H H H CH3CH(CH3)CH2CH(CH3)CH3 CH3 branch 3 2 1 benzene ring inside
- 3. IsomerismMolecules with same molecular formula but diff arrangement of atom Two types of Isomerism Positional Chain Isomer Functional Gp Isomer C – C – C – C – OH C4H10O1 StructuralIsomerism • Same molecular formula • Diff structural formula • Diff arrangement of atom Diff hydrocarbonchain skeleton • Same molecular formula • Same structuralformula • Diff spatial arrangement ofatom Stereoisomerism Hydrocarbon Chain Isomer Diff functional gp position Diff functional gp C – C – C – OH ׀ CH3 C – C – C –C ׀ OH C – C – C – C ׀ OH C – C – C – C ׀ OH C – C – C – O – C Optical IsomerGeometric Isomer Click here khan organic videos. Compound Ethane Ethanoic acid Empirical formula CH3 CH2O Molecular formula C2H6 C2H4O2 Full SF Condensed SF CH3CH3 CH3COOH Stereochemical formula (3D) Isomer Physical property Chemical property Structural isomer - Hydrocarbon chain - Functional gp position - Functional gp Different Different Different Similar Similar Different Geometrical isomer Different Similar Optical isomer Similar Similar H H ׀ ׀ H - C – C – H ׀ ׀ H H H O ׀ ‖ H - C - C - OH ׀ H Structural formula – arrangement atoms in molecule (2/3D) H H ׀ ׀ H - C – C – H ׀ ׀ H H CH3CH3 ethane Display full SF Condensed SF Ball/stick model Spacefilling Click here chemical search.
- 4. Diff functional gp position C4H9OH StructuralIsomerism • Same molecular formula • Diff structural formula • Diff arrangement of atom Diff hydrocarbon chain skeleton Diff functional gp position Diff functional gp CH3-CH2-CH2-CH3 ׀ OH Butan–1-ol CH3-CH-CH2-OH ׀ CH3 2-Methylpropan-1-ol CH3 ׀ CH3-C-OH ׀ CH3 CH3-CH2-CH-CH3 ׀ OH 2-Methylpropan-2-ol Butan-2-ol CH3-CH2-CH2-O-CH3 Methoxypropane CH3-CH2-O-CH2-CH3 Ethoxyethane 7 structural isomers CH3-CH-O-CH3 ׀ CH3 CH3-CH2-CH2=CH2 C4H8 CH3-CH=CH-CH3 CH3-C=CH2 ׀ CH3 CH2 – CH2 ׀ ׀ CH2 - CH2 C4H9Br CH3-CH2-CH2-CH2 ׀ Br CH3-CH2-CH-CH3 ׀ Br CH3 ׀ CH3-C-Br ׀ CH3 CH3- CH-CH2 ׀ ׀ CH3 Br Click here for organic notes C6H14 CH3-CH2-CH2-CH2-CH2-CH3 CH3-CH-CH2-CH2-CH3 ׀ CH3 CH3-CH2-CH-CH2-CH3 ׀ CH3 CH3 ׀ CH3-C-CH2-CH3 ׀ CH3 CH3- CH- CH-CH3 ׀ ׀ CH3 CH3 Diff hydrocarbon chain skeleton Diff functional gp position Diff functional gp Diff hydrocarbon chain skeleton Click here virtual organic text 4 structural isomers 4 structural isomers 5 structural isomers Diff hydrocarbon chain skeleton
- 5. StructuralIsomerism Click here for organic notes Click here virtual organic text C3H6O H ׀ CH3-CH2-C=O O ‖ CH3-C-CH3 OH ׀ CH3-CH=C-H OH ׀ CH2=CH-CH2 CH3-O-CH=CH2 C4H8O CH3-CH2-CH2-C-H ‖ O CH3 -CH2 -C-CH3 ‖ O CH3-CH - C-H ׀ ‖ CH3 O CH2=CH-CH2-CH2-OH CH3-CH=CH-CH2-OH CH3-CH2-CH=CH-OHCH3-CH2-O-CH=CH2CH3-CH=CH-O-CH3 C5H10 CH2=CH-CH2-CH2-CH3 CH3-CH=CH-CH2-CH3CH2=C-CH2-CH3 ׀ CH3 CH2=CH-CH-CH3 ׀ CH3 CH3-CH=C-CH3 ׀ CH3 CH2-CH-CH2-CH3 CH2 CH3-CH-CH-CH3 CH2 CH3 ׀ CH2– C-CH3 CH2 CH2 CH2 CH2 וּ ׀ ׀ CH2 –CH2 CH2 –CH-CH3 ׀ ׀ CH2– CH2 Aldehyde Ketone Alkene/Alcohol Alkene/Alcohol Alkene/Ether 5 structural isomers 8 structural isomers 10 structural isomers Cyclo – ring structure • Same molecular formula • Diff structural formula • Diff arrangement of atom
- 6. C5H11Br StructuralIsomerism Click here for organic notes Click here virtual organic text CH3-CH2-CH2-CH2-CH2-Br CH3-CH2-CH2-CH-CH3 ׀ Br CH3-CH2-CH-CH2-CH3 ׀ Br CH3-CH-CH2-CH2-Br ׀ CH3 CH3-CH2-CH-CH2-Br ׀ CH3 CH3 ׀ CH3–CH–CH-CH3 ׀ Br CH3 ׀ CH3-CH2- C-CH3 ׀ Br CH3 ׀ CH3-C-CH2-Br ׀ CH3 CH3- CH- CH-Br ׀ ׀ CH3 CH3 CH3 CH3 ׀ ׀ CH3- C - CH2 ׀ Br CH3 ׀ CH3-CH2- C-Br ׀ CH3 C3H6O2 CH3-CH2-C-OH ‖ O CH3 - C- O-CH3 ‖ O H- C-O-CH2-CH3 ‖ O CH2- CH = CH ׀ ׀ OH OH CH=C-CH3 Ι Ι OH OH HO-C=CH-CH3 ׀ OH HO-CH-CH=CH2 ׀ OH OH ׀ CH3-CH–C-H ‖ O CH2- C = CH2 ׀ ׀ OH OH OH ׀ CH2-CH2-CH ‖ O CH3-O-CH2-CH ‖ O CH2 = C-O-CH3 ׀ OH CH2= CH-O-CH2 ׀ OH CH=CH-O-CH3 ׀ OH 2-bromo-3-methylbutane 2-bromo-2-methylbutane 8 structural isomers Alcohol / Alkene Carboxylic acid Ester Aldehyde / Alcohol Alcohol / Alkene / Ether 14 structural isomers • Same molecular formula • Diff structural formula • Diff arrangement of atom
- 7. H H H H H ׀ ׀ ׀ ׀ ׀ H - C – C – C - C – C – F ׀ ׀ ׀ ׀ ׀ H CH3 H H H H CH3 ׀ ׀ CI – C – C – C ׀ ׀ H CH3 H CH3 H ׀ ׀ ׀ H - C – C – C – Br ׀ ׀ ׀ H CH3 H H CH3 H H ׀ ׀ ׀ ׀ H - C – C – C - C – H ׀ ׀ ׀ ׀ I H H H H C ׀ ׀ C – C – C – C - C ׀ CI C CH3 H ׀ ׀ ׀ H - C – C – C – H ׀ ׀ ׀ CI H H CH3 ׀ CI – C – C – C ׀ H H H CI ׀ ׀ ׀ H - C – C - C – H ׀ ׀ ׀ H CH3 H Halogenoalkane and Nomenclature H ׀ CH3 – C – CI ׀ H Types of halogenoalkane Primary 1 0 NO alkyl /1 alkyl/R gp bond to C attach to halogen H CH3 H ׀ ׀ ׀ H - C – C – C – CI ׀ ׀ ׀ H CH3 H H ׀ H - C – CI ׀ H Secondary 2 0 2 alkyl/R gp bond to C attach to halogen H ׀ CH3 – C – CI ׀ CH3 H H H ׀ ׀ ׀ H - C – C – C – H ׀ ׀ ׀ H CI H H ׀ R – C – CI ׀ R Tertiary 3 0 3 alkyl/R gp bond to C attach to halogen CH3 ׀ CH3 – C – CI ׀ CH3 H CH3 H ׀ ׀ ׀ H - C – C – C – H ׀ ׀ ׀ H CI H R ׀ R – C – CI ׀ R H H H ׀ ׀ ׀ H - C – C – C – H ׀ ׀ ׀ H CI H H CH3 H ׀ ׀ ׀ H - C – C – C – C ׀ ׀ ׀ H CI H CH3 ׀ CH3 – C – CI ׀ CH3 H CH3 H ׀ ׀ ׀ H - C – C – C – H ׀ ׀ ׀ H Br H C ׀ CI – C – C – C ׀ C – C Br CH3 ׀ ׀ C – C – C – C – C ׀ ׀ CH3 CH3 C CI C ׀ ׀ ׀ C – C – C – C – C ׀ C Primary 1 o Secondary2 o Tertiary 3 o H H H ׀ ׀ ׀ H - C – C - C – H ׀ ׀ ׀ H H F H H CH3 H H ׀ ׀ ׀ ׀ ׀ H – C – C - C – C – C - H ׀ ׀ ׀ ׀ ׀ H CI CH3 H H H ׀ CH3 – C – CI ׀ CH3
- 8. C ׀ HO – C – C – C ׀ C – C H CH3 ׀ ׀ HO – C – C – C ׀ ׀ H CH3 H H H H H ׀ ׀ ׀ ׀ ׀ H - C – C – C - C – C – OH ׀ ׀ ׀ ׀ ׀ H CH3 H H H H CH3 H ׀ ׀ ׀ H - C – C – C – OH ׀ ׀ ׀ H CH3 H H CH3 H H ׀ ׀ ׀ ׀ H - C – C – C - C – H ׀ ׀ ׀ ׀ OH H H H H C ׀ ׀ C – C – C – C - C ׀ OH C CH3 ׀ ׀ H - C – C – C – H ׀ ׀ ׀ OH H H CH3 ׀ HO – C – C – C ׀ H H H OH ׀ ׀ ׀ H - C – C - C – H ׀ ׀ ׀ H CH3 H Alcohol and Nomenclature H ׀ CH3 – C – OH ׀ H Types of alcohol Primary 1 0 NO alkyl /1 alkyl/R gp bond to C attach to OH H CH3 H ׀ ׀ ׀ H - C – C – C – OH ׀ ׀ ׀ H CH3 H H ׀ H - C – OH ׀ H Secondary 2 0 2 alkyl/R gp bond to C attach to OH H ׀ CH3 – C – OH ׀ CH3 H H H ׀ ׀ ׀ H - C – C – C – H ׀ ׀ ׀ H OH H H ׀ R – C – OH ׀ R Tertiary 3 0 3 alkyl/R gp bond to C attach to OH CH3 ׀ CH3 – C – OH ׀ CH3 H CH3 H ׀ ׀ ׀ H - C – C – C – H ׀ ׀ ׀ H OH H R ׀ R – C – OH ׀ R H H H ׀ ׀ ׀ H - C – C – C – H ׀ ׀ ׀ H OH H H CH3 H ׀ ׀ ׀ H - C – C – C – C ׀ ׀ ׀ H OH H CH3 ׀ CH3 – C – OH ׀ CH3 H CH3 H ׀ ׀ ׀ H - C – C – C – H ׀ ׀ ׀ H OH H OH CH3 ׀ ׀ C – C – C – C – C ׀ ׀ CH3 CH3 C OH C ׀ ׀ ׀ C – C – C – C – C ׀ C Primary 1 o Secondary2 o Tertiary 3 o H H H ׀ ׀ ׀ H - C – C - C – H ׀ ׀ ׀ H H OH H H CH3 H H ׀ ׀ ׀ ׀ ׀ H – C – C - C – C – C - H ׀ ׀ ׀ ׀ ׀ H OH CH3 H H H ׀ CH3 – C – OH ׀ CH3
- 9. H H H H H ׀ ׀ ׀ ׀ ׀ H - C – C – C - C – C –NH2 ׀ ׀ ׀ ׀ ׀ H CH3 H H H H CH3 ׀ ׀ NH2 – C – C – C ׀ ׀ H CH3 H CH3 H ׀ ׀ ׀ H - C – C – C – NH2 ׀ ׀ ׀ H CH3 H H CH3 H H ׀ ׀ ׀ ׀ H - C – C – C - C – H ׀ ׀ ׀ ׀ NH2 H H H H C ׀ ׀ C – C – C – C - C ׀ NH2 C CH3 H ׀ ׀ ׀ H - C – C – C – H ׀ ׀ ׀ NH2 H H C ׀ NH2 – C – C – C ׀ H H H NH2 ׀ ׀ ׀ H - C – C - C – H ׀ ׀ ׀ H CH3 H Amines and Nomenclature H ׀ CH3 – C – NH2 ׀ H Types of amines Primary 1 0 NO alkyl /1 alkyl/R gp bond to C attach to nitrogen H CH3 H ׀ ׀ ׀ H - C – C – C – NH2 ׀ ׀ ׀ H CH3 H H ׀ H - C – NH2 ׀ H Secondary 2 0 2 alkyl/R gp bond to C attach to nitrogen H ׀ CH3 – C – NH2 ׀ CH3 H H H ׀ ׀ ׀ H - C – C – C – H ׀ ׀ ׀ H NH2 H H ׀ R – C – NH2 ׀ R Tertiary 3 0 3 alkyl/R gp bond to C attach to nitrogen CH3 ׀ CH3 – C – NH2 ׀ CH3 H CH3 H ׀ ׀ ׀ H - C – C – C – H ׀ ׀ ׀ H NH2 H R ׀ R – C – NH2 ׀ R H H H ׀ ׀ ׀ H - C – C – C – H ׀ ׀ ׀ H NH2 H H CH3 H ׀ ׀ ׀ H - C – C – C – C ׀ ׀ ׀ H NH2 H CH3 ׀ CH3 – C – NH2 ׀ CH3 H CH3 H ׀ ׀ ׀ H - C – C – C – H ׀ ׀ ׀ H NH2 H C ׀ NH2 – C – C – C ׀ C – C NH2 CH3 ׀ ׀ C – C – C – C – C ׀ ׀ CH3 CH3 C NH2 C ׀ ׀ ׀ C – C – C – C – C ׀ C Primary 1 o Secondary2 o Tertiary 3 o H H H ׀ ׀ ׀ H - C – C - C – H ׀ ׀ ׀ H H NH2 H H CH3 H H ׀ ׀ ׀ ׀ ׀ H – C – C - C – C – C - H ׀ ׀ ׀ ׀ ׀ H NH2 CH3 H H H ׀ CH3 – C – NH2 ׀ CH3
- 10. Delocalizationof electrons Resonance • Describing delocalizationof electrons within a molecule/polyatomic ion where bonding cannot be express by ONE single Lewis structure • Delocalization of π bond – π electrons spread over more than 2 nuclei • π electrons are shared/spread– more stable Resonance structuresbenzene Benzene 6HC6 resonance structure 1 resonance structure 2 Resonance hybrid • All bonds C6H6 are identical in length/ strength • Hybrid of 2 resonance structures • No C-C (single) or C=C (double) bonds found • Only C ----- C bond • Intermediate character bet single/double bond • Bond Order = • Unhybridised p orbital • Delocalization electrons above below plane • sp2 hybridization on carbon center 1.5 Click here to view Delocalized electrons Kekulé structure Cyclohexa- 1,3,5 triene χ ✓ double/single bonds bet them Benzene Hexagonal, planar Resonance Hybrid more stable than any of the resonance structure ✓ Click here to view Kekule ✓ or
- 11. Resonance/Delocalization Energy ΔH cyclohexene = -120 kJmol-1 ΔH cyclohexa1,3,5 triene = - 360 kJmol-1 ΔH Benzene = - 208kJmol-1 Enthalpy change hydrogenation χ - Benzene lower in energy by 152 kJ - More stable due to delocalization of π electrons - 152 kJ - 152 C-C Single bond C = C Double bond C = C Benzene Bond length/pm 154 134 140 Bond enthalpy/kJmol-1 346 614 507 Evidence for Benzene structure 1 2 Click here evidence against Kekule • X ray hit benzene crystal • Interact with electron (electron density map) • X ray diffraction produced • Bond length measured X ray crystallography NO single/double bond detected ✓ ✓ 3 No addition rxn in benzene C=C Addition rxn Substitution rxn NO double bond -240 …… -360 - 208 ΔH cyclohexa 1,3 diene = -240 kJmol-1 + ✓ ✓ + Br - Br →✓
