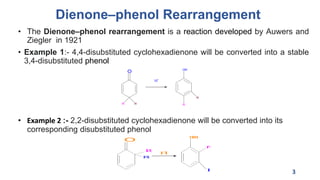
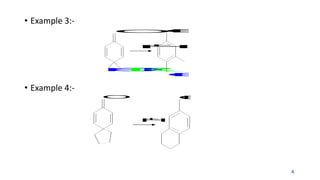
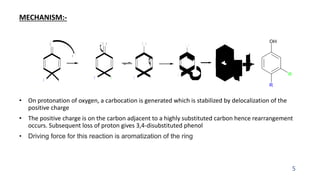
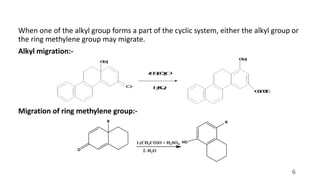
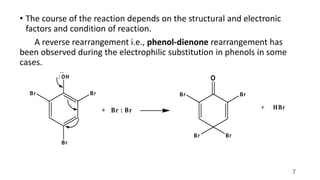
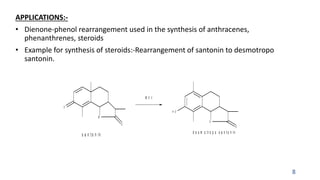
The document summarizes the Dienone-Phenol rearrangement reaction. It begins by classifying rearrangement reactions based on the migrating group or atom. It then provides examples of the Dienone-Phenol rearrangement where disubstituted cyclohexadienones rearrange to the corresponding disubstituted phenols. The mechanism involves the generation of a carbocation through protonation, followed by rearrangement and loss of a proton to form the phenol product. The rearrangement is driven by aromatization. It notes applications in synthesizing anthracenes, phenanthrenes, and steroids like santonin. References on reaction mechanisms and rearrangements are also provided.