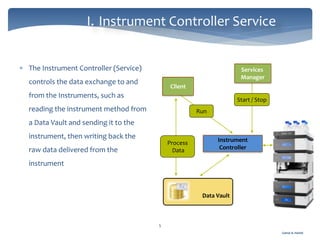
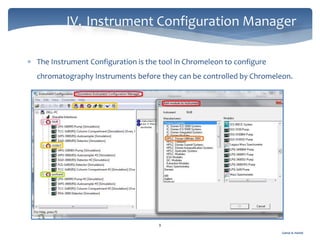
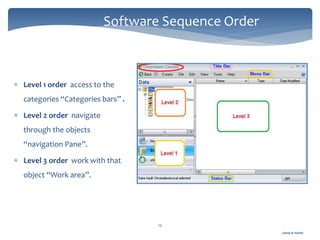
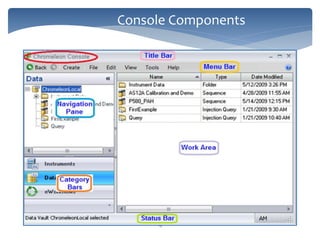
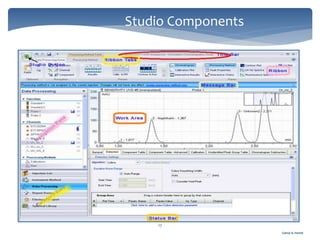
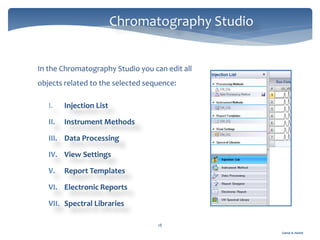
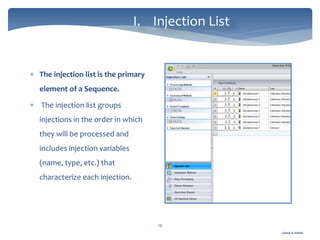
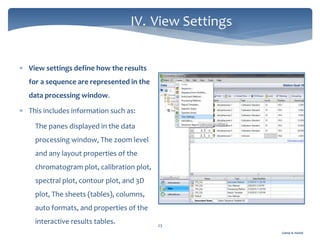
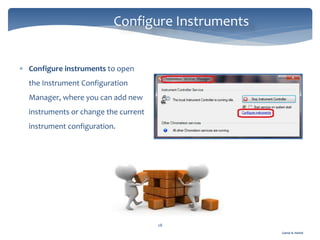
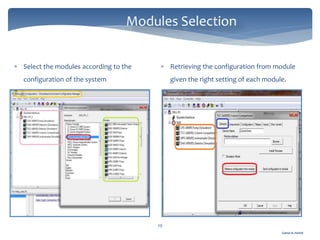
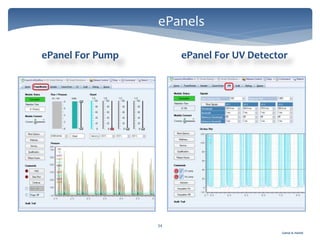
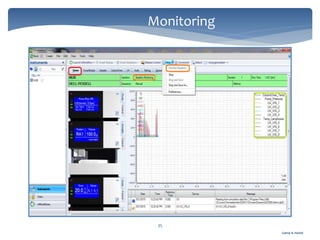
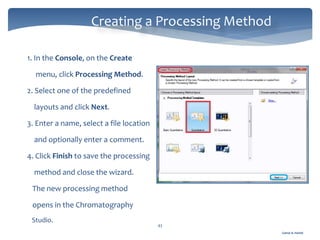
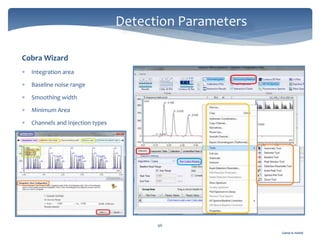
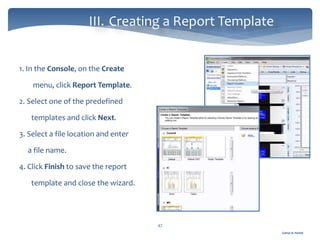
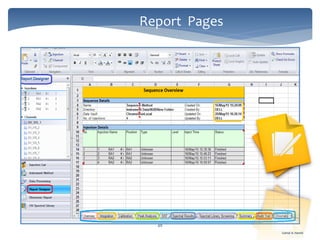
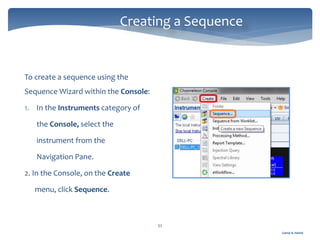
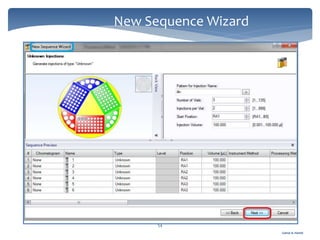
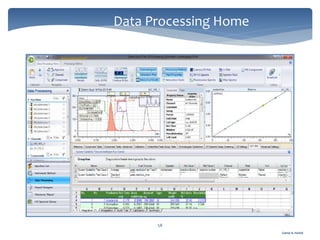
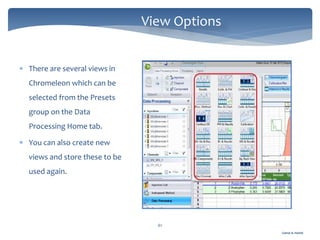
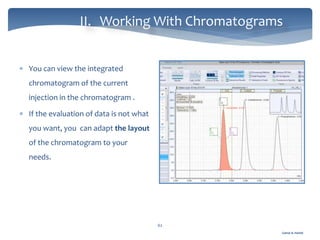
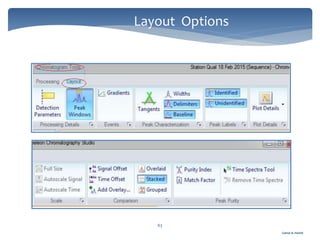
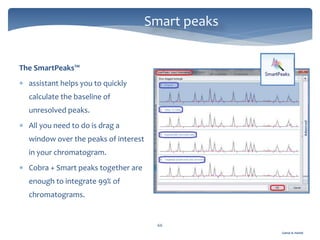
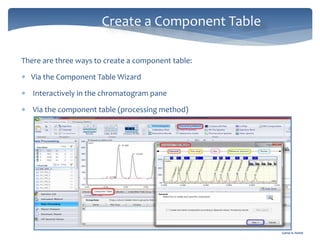
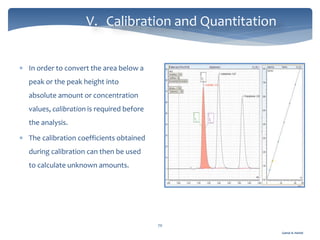
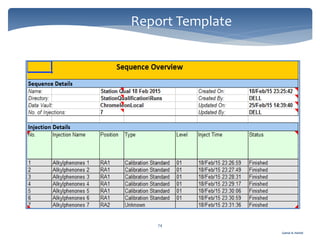
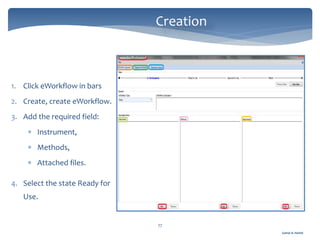
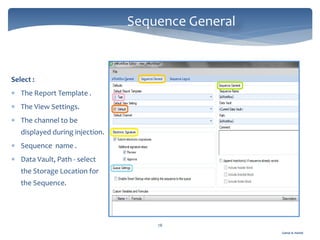
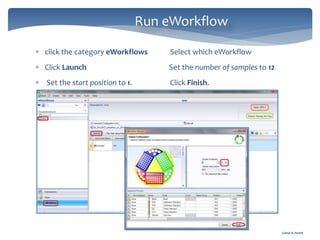
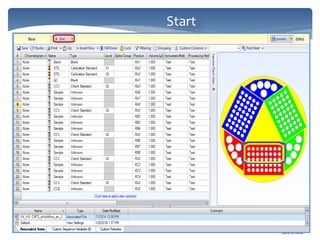
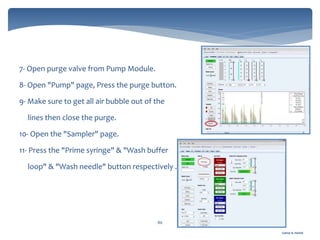
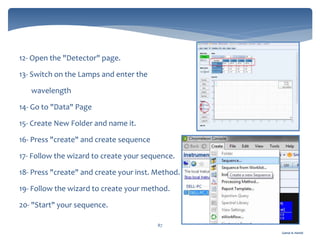
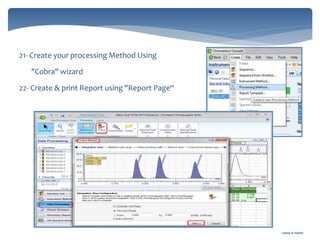
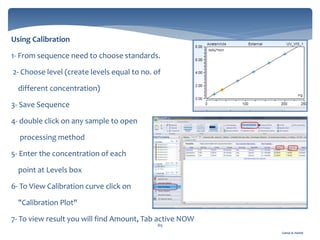
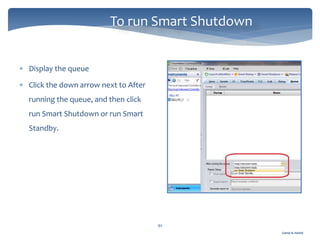
The document discusses Chromeleon, a chromatography data system. It describes the main Chromeleon software components, including the instrument controller service, client, services manager, instrument configuration manager, and administration console. It also covers the Chromeleon client, which contains the Chromeleon console and chromatography studio. The simple steps of an analysis using Chromeleon are outlined as starting Chromeleon, starting the instrument, creating a sequence, acquiring data, processing data, and reviewing/reporting results.