The document summarizes key aspects of hemostasis including:
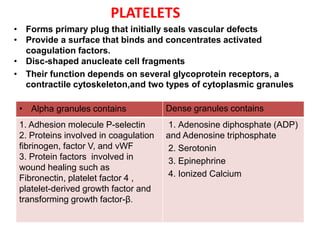
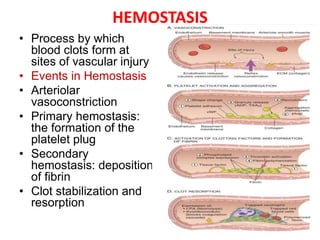
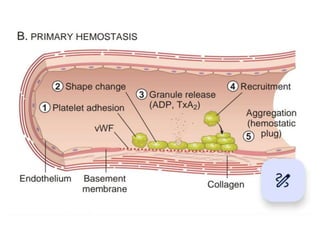
1. Platelet aggregation forms a primary plug at sites of vascular injury through interactions with von Willebrand factor, collagen, and fibrinogen bridges between platelets. Inherited deficiencies of von Willebrand factor or glycoprotein IIb/IIIa result in bleeding disorders.
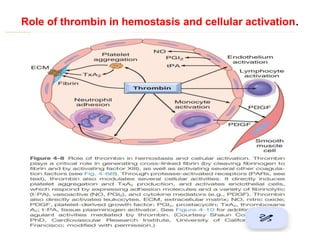
2. The coagulation cascade is a series of enzymatic reactions that results in thrombin generation and fibrin deposition to form a clot. Thrombin both converts fibrinogen to fibrin and activates platelets.
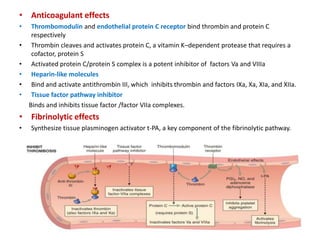
3. Counterregulatory mechanisms like the endothelial barrier and fibrinolytic system activate proteins like tissue plasminogen activator and plasmin to limit clotting